 |
 |
 |
| |
Proton Beam Radiotherapy Noninferior to Radiofrequency Ablation for HCC
|
| |
| |
Proton beam radiotherapy versus radiofrequency ablation treatment in patients with recurrent hepatocellular carcinoma: a randomized controlled phase 3 non-inferiority APROH trial.
EASL 2020, Digital International Liver Congress, August 27-29, 2020.
Mark Mascolini
Proton beam radiotherapy (PBT) proved noninferior to radiofrequency ablation (RFA) in local progression-free survival of people with small recurrent or residual hepatocellular carcinoma (HCC), according to results of the 144-person randomized APROH trial in South Korea [1]. PBT was generally tolerable and safe.
RFA remains the standard of care for people with Barcelona Clinic Liver Cancer (BCLC) stage 0 and stage A HCC that cannot be treated surgically. A US 76-person phase 2 trial found PBT "safe and effective local-regional therapy for inoperable HCC" [2]. But no randomized trial had assessed PBT until researchers in South Korea mounted this investigator-initiated phase 3 noninferiority trial comparing PBT with RFA in patients with recurrent or residual HCC (rHCC).
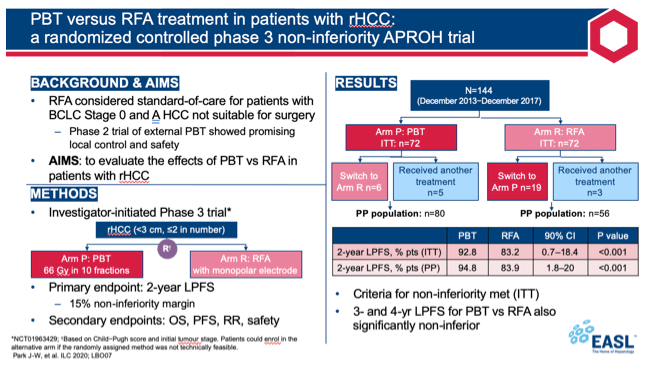
The APROH team randomly assigned patients with rHCC (1 or 2 tumors smaller than 3 cm) to PBT or RFA according to Child-Pugh score and initial tumor stage. If the random assignment proved technically infeasible, patients could enroll in the other arm. Participants could have no vascular invasion and no extrahepatic metastases. They had to have an ECOG performance status of 0 to 2 and a Child-Pugh score of A or B7 without uncontrolled ascites.
PBT patients received 66 gray equivalent in 10 fractions, while RFA patients received RFA with a monopolar electrode. The trial's primary endpoint was 2-year local progression-free survival with a noninferiority margin of 15%.
From December 2013 through December 2017, researchers enrolled 72 people in the PBT arm and 72 in the RFA arm. These 144 people made up the intention-to-treat population. Six people in the PBT arm switched to the RFA arm, and 5 received another treatment. Nineteen patients in the RFA arm switched to the PBT arm, and 3 received another treatment. As a result the per-protocol population consisted of 80 people in the PBT arm and 56 in the RFA arm.
Baseline characteristics were well matched between the two study arms. About 82% of participants were men and median age stood around 61. Median follow-up lasted 51.6 months in the PBT arm and 50.7 months in the RFA arm.
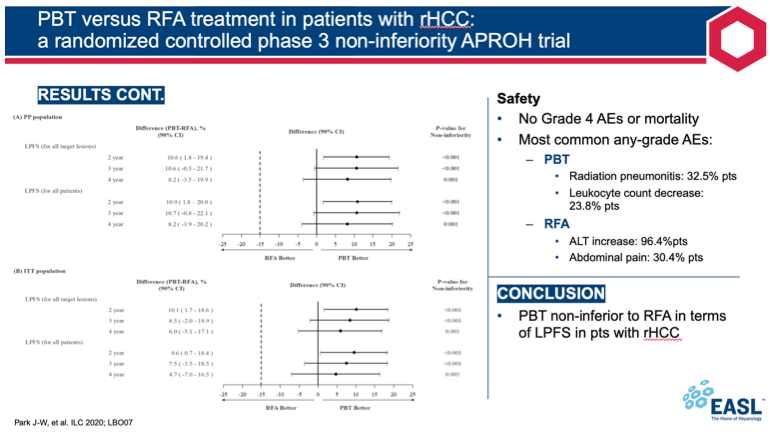
When the last patient completed the trial in January 2020, the 2-year local progression-free survival rate in the intention-to-treat population was greater in the PBT arm than the RFA arm (92.8% vs 83.2%, P < 0.001 for noninferiority). That result met criteria indicating that PBT is noninferior to RFA for local progression-free survival. Results for 2-year local progression-free survival were similar in the per-protocol population, 94.8% with PBT vs 83.9% with RFA (P < 0.001 for noninferiority).
Three- and 4-year local progression-free survival rates in the intention-to-treat population were also noninferior with PBT versus RFA (85.8% vs 78.3%, P < 0.001 for noninferiority; 83.0% vs 78.3%, P = 0.003 for noninferiority).
The most frequent adverse events of any grade in the PBT arm were radiation pneumonitis (32.5%) and decreased leukocyte count (23.8%). In the RFA arm, the most frequent adverse events were increased alanine aminotransferase (96.4%) and abdominal pain (30.4%). There were no grade 4 adverse events and no deaths.
The APROH investigators conclude that this first phase 3 randomized controlled trial of PBT confirms its statistical noninferiority to RFA in local progression-free survival among patients with recurrent or residual HCC and with 1 to 2 tumors less than 3 cm in size. The researchers believe the results "suggest that PBT may be a promising treatment option for small HCC."
References
1. Park JW, Kim TH, Koh YH, et al. Proton beam radiotherapy versus radiofrequency ablation treatment in patients with recurrent hepatocellular carcinoma: a randomized controlled phase 3 non-inferiority APROH trial. EASL 2020, Digital International Liver Congress, August 27-29, 2020. Abstract LBO07.
2. Bush DA, Kayali Z, Grove R, Slater JD. The safety and efficacy of high-dose proton beam radiotherapy for hepatocellular carcinoma: a phase 2 prospective trial. Cancer. 2011 Jul 1;117(13):3053-9. doi: 10.1002/cncr.25809.
|
| |
|
 |
 |
|
|