 |
 |
 |
| |
Positive topline results from a 24-week, randomized,
double-blind, placebo-controlled, multicenter, phase 2 study of the FGF19 analogue aldafermin (NGM282) in patients with nonalcoholic steatohepatitis
|
| |
| |
EASL 2020 Aug 29 virtual
Stephen A. Harrison1,2, Guy Neff3, Cynthia D. Guy4, Mustafa Bashir5, Nadege Gunn2,
Angelo H. Paredes6, Ziad Younes7, Juan P. Frias8, James F. Trotter9, Anita Kohli10, Sam E. Moussa11, Kristin Nelson12, Mildred Gottwald12, William Chang12, Andrew Z. Yan12, Alex M. DePaoli12, Lei Ling12 and Hsiao D. Lieu12
1University of Oxford, 2Pinnacle Clinical Research, 3Covenant Research, 4Pathology, Duke University, 5Radiology and Medicine (Gastroenterology), Duke University, 6San Antonio Military Medical Center, 7Gastro One Research, TN, 8National Research Institute, CA, 9Texas Digestive Disease Consultants, TX, 10Arizona Liver Health, AZ, 11Adobe Clinical Research, AZ, 12NGM Biopharmaceuticals, CA


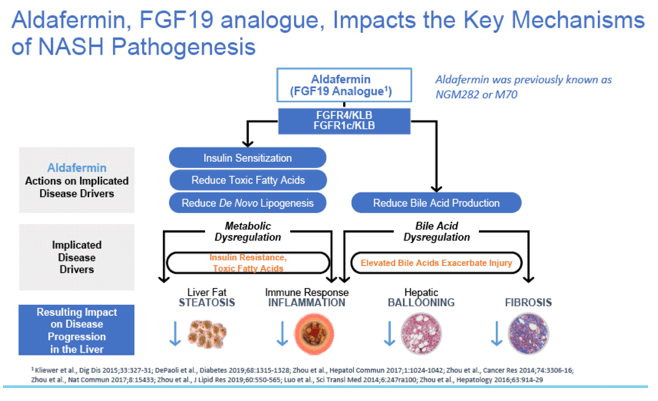
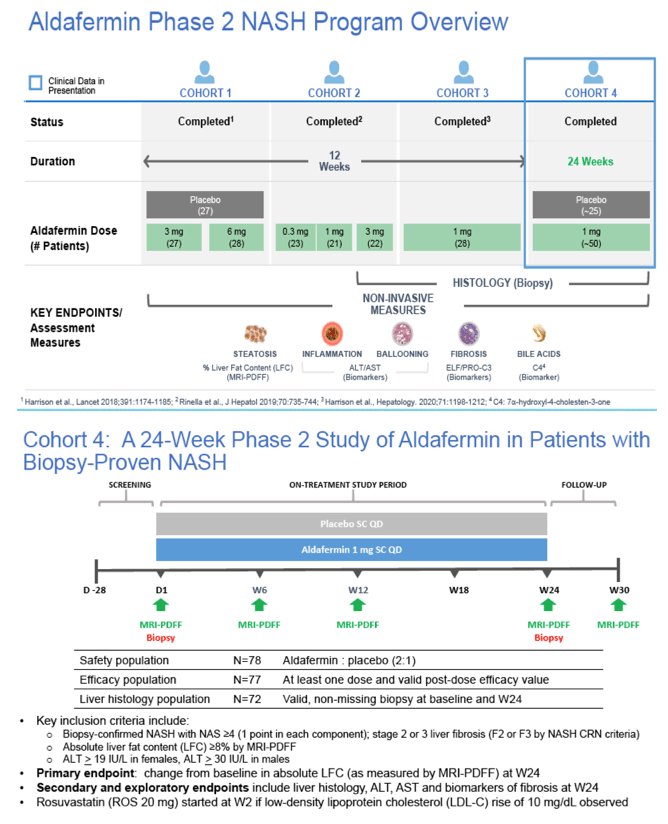
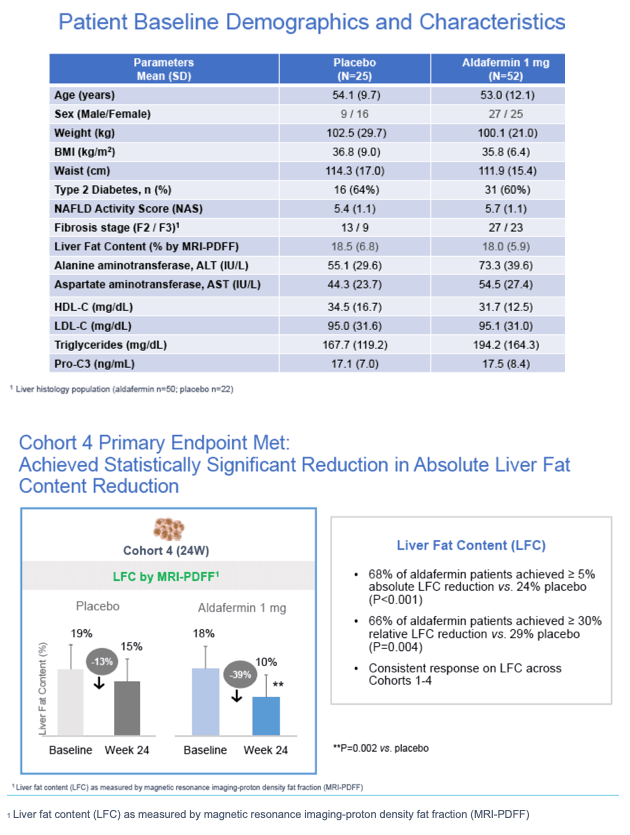
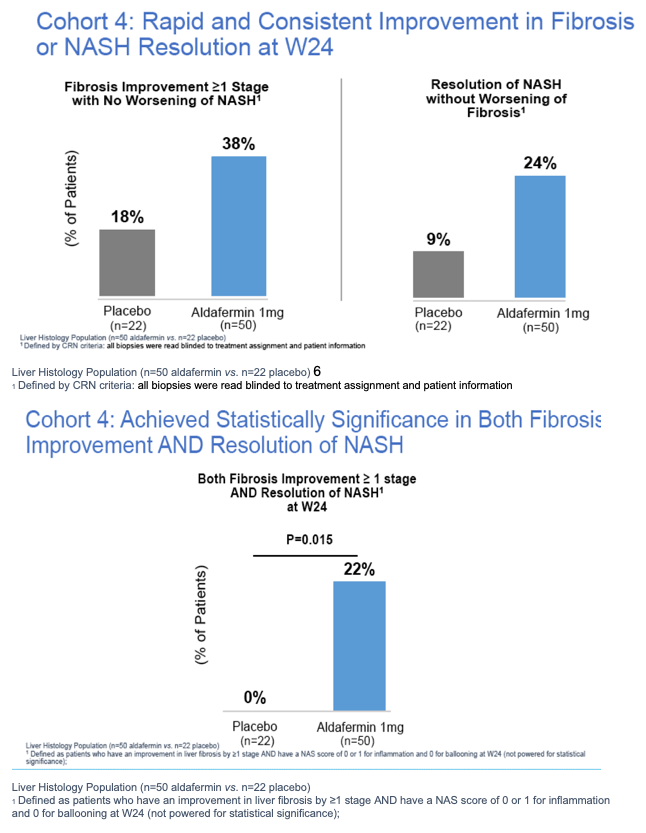
|
| |
|
 |
 |
|
|