 |
 |
 |
| |
HBV Reactivation Rare in HBV Patients Who Used Immune Checkpoint Inhibitors
|
| |
| |
EASL 2020, Digital International Liver Congress, August 27-29, 2020
Mark Mascolini
In a territory-wide Hong Kong study, people with chronic or past HBV infection who took immune checkpoint inhibitors rarely had HBV reactivation [1]. No hepatitis B surface antigen (HBsAg)-positive people attained HBsAg seroclearance after immunotherapy.
Monoclonal antibody immune checkpoint inhibitors have improved survival of people with diverse advanced or metastatic malignancies. These agents exert their effect by disrupting immune tolerance, but the relationship between toxicity and response remains incompletely understood.
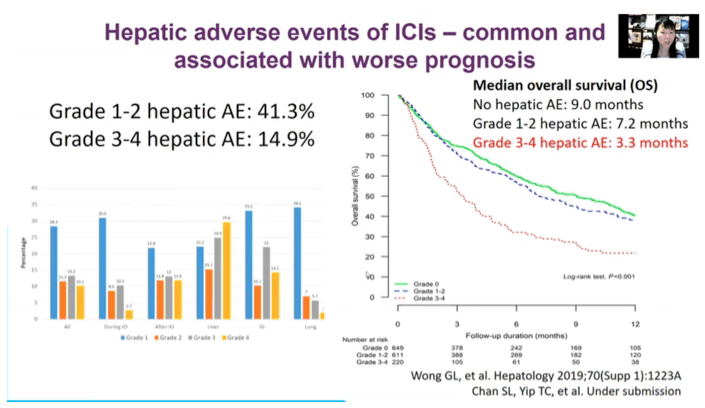
Recent work suggests that new checkpoint inhibitors-programmed death receptor 1 (PD-1) inhibitors and programmed cell death ligand 1 (PD-L1) inhibitors-may have a role in treating chronic HBV infection. HBV-specific T cells in the liver express PD-1, and chronic HBV infection drives PD-L1 expression in hepatocytes. Researchers at the Chinese University of Hong Kong and collaborators who conducted this study in people with HBV note that hepatic adverse events are common with immune checkpoint inhibitors and are linked to worse overall survival.
To learn more about the potential impact of these agents in people with chronic hepatitis B or past HBV infection, the Chinese University team mounted a retrospective observational cohort study across the territory of Hong Kong. Using the Clinical Data Analysis and Reporting System (CDARS), they identified consecutive adult Chinese patients who took at least one dose of a checkpoint inhibitor from July 2014 through October 2018. Participants had (1) chronic HBV infection, defined as being HBsAg-positive for at least 6 months or (2) past HBV infection, defined as being HBsAg-negative but positive for antibody to hepatitis B core antigen (anti-HBc-positive). The analysis excluded people with hepatitis C or D virus or HIV coinfection as well as people missing HBsAg, anti-HBc, or alanine aminotransferase (ALT) results.
The analysis included 395 HBV patients-237 who were HBsAg-positive (current infection) and 158 who were HBsAg-negative but positive for anti-HBc (past infection). Participants with chronic HBV were younger than those with past HBV (average 57.9 vs 62.9 years), more likely to be men (84.4% vs 69.6%), and had higher ALT (average 0.8 vs 0.6 at the upper limit of normal [ULN]). HBV DNA averaged 1.9 log10 IU/mL in the chronic HBV group and 0.06 log10 IU/mL in the past HBV group.
Almost everyone in the chronic HBV group (97%) took some nucleos(t)ide analog, compared with 28% in the past HBV group. Treatment usually included entecavir (89% and 27%).
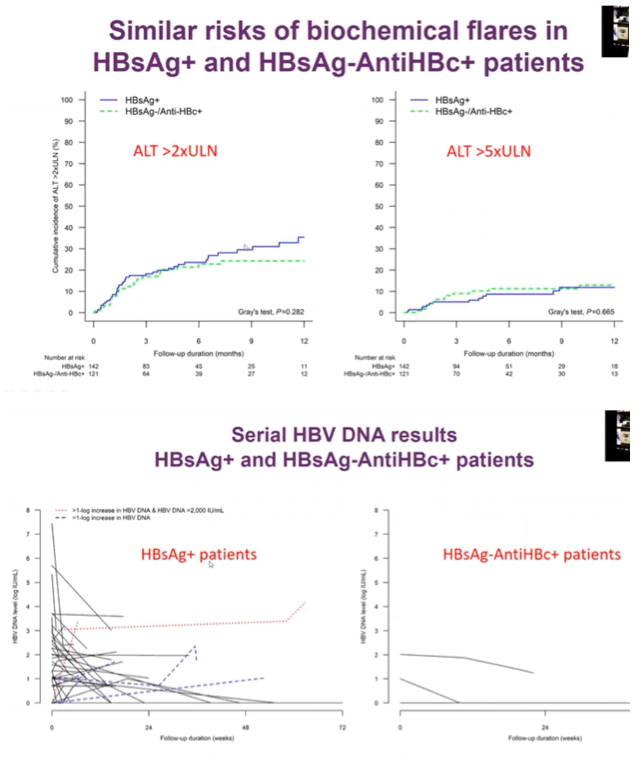
People with chronic HBV had marginally more biochemical and virologic flares than the past-HBV group. When the researchers defined HBV reactivation as ALT more than 2 times ULN, 31.0% of the HBsAg-positive group and 22.3% of the anti-HBc-positive group had a flare (P = 0.282). Smaller proportions had reactivation defined as ALT more than 5 times the upper limit of normal (12.7% vs 11.6%, P = 0.665). When the research team defined reactivation virologically as HBV DNA above 2000 IU/mL, only 2 HBsAg-positive people (1.7%) and no anti-HBC-positive people had a flare (P = 0.506). When they defined reactivation as more than a 10-fold jump in HBV DNA IU/mL, only 6 HBsAg-positive people (4.1%) and no anti-HBC-positive people had a flare (P = 0.085).
The investigators found that no HBsAg-positive people cleared HBsAg (seroconversion) and no anti-HBc-positive person had HBsAg seroreversion during follow-up.
The Hong Kong team concluded that HBV reactivation is rare in people who received immunotherapy for chronic or past HBV infection. They stressed that none of 237 HBsAg-positive people had HBsAg seroclearance after immunotherapy.
Reference
1. Wong GLH, Yip TCF, Wong VWS, et al. Immune checkpoint inhibitors and hepatitis B viral kinetics--a territory-wide cohort study. EASL 2020, Digital International Liver Congress, August 27-29, 2020. Abstract AS092.



|
| |
|
 |
 |
|
|