 |
 |
 |
| |
Direct-acting antiviral treatment in patients infected with a nonepidemic
hepatitis C genotype in the Netherlands: results from a
nationwide cohort study
|
| |
| |
EASL 2020 Aug 27-29 virtual
Reported by Jules Levin
Download the PDF here
Download the PDF here
C. Isfordink1,2, T. van de Laar3,4, S. Rebers1, E. Wessels5, R. Molenkamp6, M. Knoester7, B. Baak4, C. van Nieuwkoop8, B. van Hoek5, S. Brakenhoff6, H. Blokzijl7, J. Arends2, M. van der Valk1, J. Schinkel1, on behalf of the HepNed study group
1Amsterdam UMC, Amsterdam, the Netherlands (NL); 2UMC Utrecht, Utrecht, NL. 3Sanquin Research, Amsterdam, NL; 4OLVG, Amsterdam, NL; 5Leiden UMC, Leiden, NL; 6Erasmus MC, Rotterdam, NL; 7UMC Groningen, Groningen, NL; 8Haga Teaching Hospital, The Hague, NL
----------------------------------
Efficacy of NS5A inhibitors against unusual and potentially difficult-to-treat HCV subtypes commonly found in sub Saharan Africa and South East Asia
Research Needs Editorial - Efficacy of NS5A inhibitors against unusual and potentially difficult-to-treat HCV subtypes commonly found in sub Saharan Africa and South East Asia About the absolute need to keep active research on the efficacy of direct-acting antiviral drugs against the hepatitis C virus
Suboptimal SVR rates in African patients with atypical genotype 1 subtypes: Implications for global elimination of hepatitis C. Childs et al. pdf attached
Treatment response
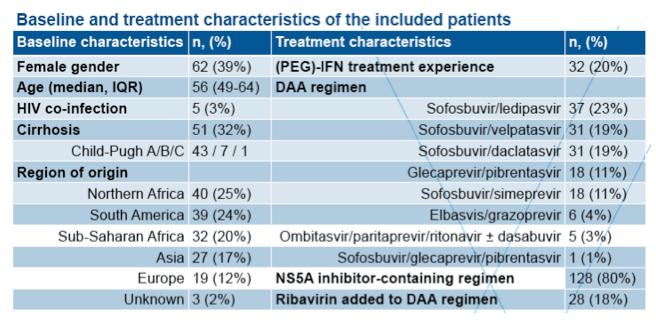
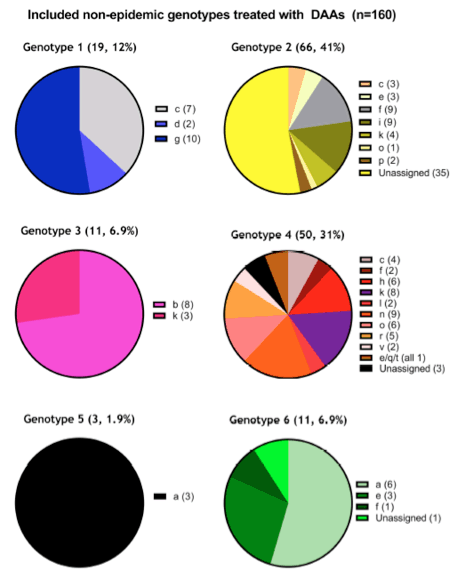
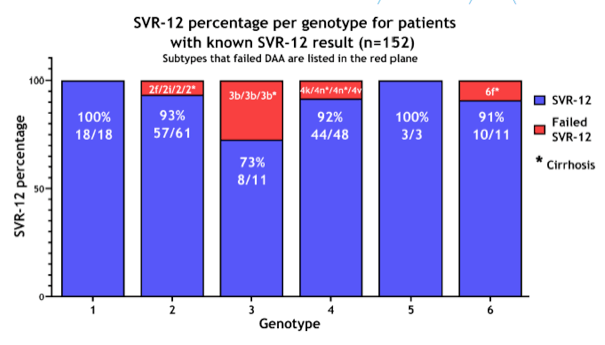
To date, 63 patients have completed DAA treatment and follow-up. Fifty-six of these patients have achieved an SVR, while 7 failed treatment, giving an overall SVR rate of 89%. As expected, response rates in genotypes 1a or 1b were uniformly high. Similarly, all patients infected with genotype 2, 3, 4 and 5 achieved an SVR. In those patients with unusual G1 and G4 African subtypes, treatment response according to genotype and choice of treatment regimen are shown in Table 2. An SVR was observed in only 21 of the 28 (75%) patients infected with unusual African genotype 1.
Five patients who failed initial treatment have thus far commenced retreatment: 3 of these patients have completed 16 weeks of glecaprevir and pibrentasvir, 2 of whom have achieved an SVR. One individual with cirrhosis and G1* HCV virus failed to achieve an SVR with 16 weeks of glecaprevir and pibrentasvir. Two other patients were retreated with sofosbuvir, velpatasvir and voxilaprevir and achieved an SVR.
Editorial - DAA failures in African patients with ''unusual'' HCV subtypes - Hey! Didn't you know there was another world? Jean-Michel Pawlotsky. Pdf attached



|
| |
|
 |
 |
|
|