 |
 |
 |
| |
ViiV Healthcare to present long-term safety and efficacy data for 2-drug regimen Dovato (dolutegravir/lamivudine) alongside other key research advances at the HIV Glasgow 2020 congress
|
| |
| |
For media and investors only
Issued: London, UK 30 September 2020
• Data presented will reinforce the potential to shift the treatment paradigm to 2-drug regimens (2DRs) for people living with HIV
ViiV Healthcare, the global specialist HIV company majority owned by GSK, with Pfizer Inc. and Shionogi Limited as shareholders will be presenting 25 abstracts during the HIV Glasgow 2020 congress, being held virtually 5-8 October. Research will be presented from across its diverse portfolio of late-stage pipeline and licensed HIV treatments as well as findings from one of the largest, global, HIV patient-reported outcomes studies to date.
Key data presentations will include:
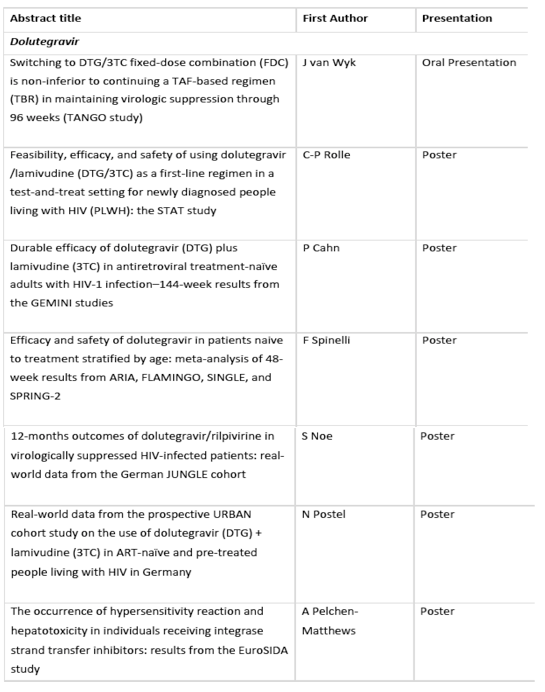
• GEMINI 144-week and TANGO 96-week pivotal trial data that build upon the existing efficacy and safety profile of 2-drug regimen dolutegravir plus lamivudine in treatment naïve adults with HIV-1 and Dovato (dolutegravir/lamivudine) in virologically suppressed adults with HIV-1[1],[2]
• STAT 24-week data which evaluated Dovato for rapid initiation of treatment after diagnosis in adults with HIV-1[3]
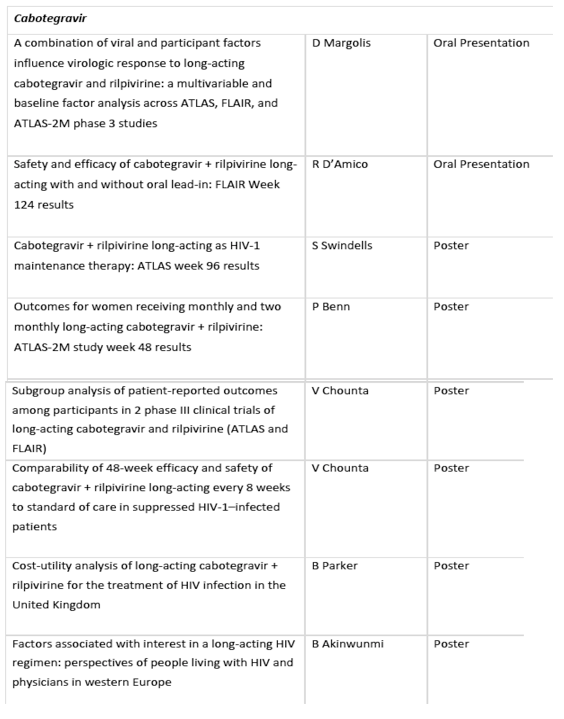
• FLAIR 124-week findings for long-acting investigational cabotegravir and rilpivirine that further define its efficacy and safety profile, an analysis from the Phase 3 clinical development programme on factors influencing viral response to long-acting cabotegravir and rilpivirine, as well as an analysis of ATLAS-2M 48-week data of key outcomes in women, an often-under-represented population in clinical trials[4],[5],[6]
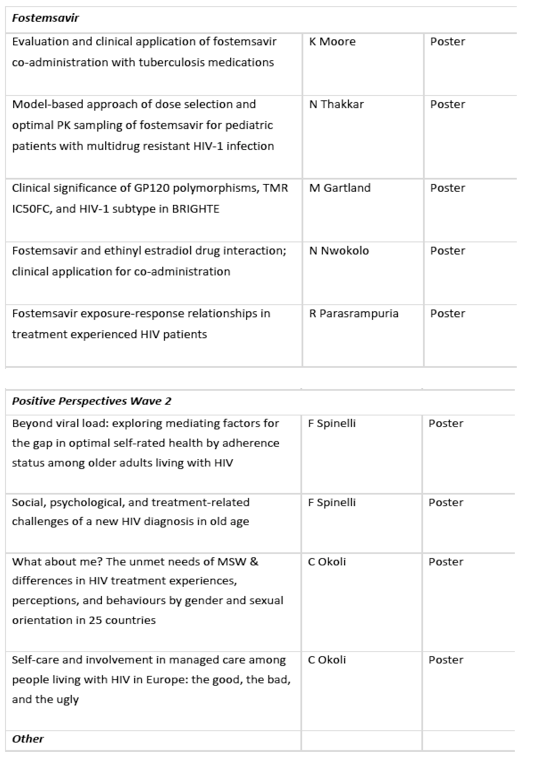
• Positive Perspectives Wave 2 data exploring gaps in achieving optimal self-rated health; challenges of a new HIV diagnosis in old age; self-care and involvement in HIV care[7],[8],[9],[10]
ViiV Healthcare will also be hosting a virtual media event (9 October 2020, 13:00-14:00 BST) for registered journalists, looking at how the introduction of 2DRs and their growing evidence base can impact the HIV treatment paradigm. The session will reflect on clinical findings presented at HIV Glasgow and how they will potentially impact people living with HIV who are taking medications over a lifetime. Speakers include renowned HIV specialist Pedro Cahn M.D., who has been at the forefront of 2DR development in HIV. To register for the event, email: info@viivmediaevent.co.uk.
Kimberly Smith, Head of Research & Development, ViiV Healthcare, said: "The advances we will be presenting from across the portfolio reflect our continued commitment to addressing the challenges of HIV and delivering therapies that really make a difference in people's lives. Dovato has already delivered impressive safety and efficacy results for both treatment-naïve and treatment-experienced adults living with HIV. The new long-term results presented at Glasgow will go even further to characterise the durability of and resistance profile for this treatment. We're excited to participate in the congress and share our latest findings with the community."
Key abstracts to be presented by ViiV Healthcare and study partners at HIV Glasgow 2020:
Week 144 findings from the GEMINI 1 & 2 studies evaluating dolutegravir plus lamivudine in treatment-naïve HIV-1 adults: The GEMINI studies were set up to see if treatment-naïve adults with HIV-1 could get the same level of viral suppression by taking two drugs instead of three.[11],[12] These three-year data will build upon 96-week results, which demonstrated the non-inferior efficacy of dolutegravir plus lamivudine compared to the 3-drug regimen off dolutegravir plus tenofovir disoproxil fumarate/emtricitabine (TDF/FTC), with no treatment-emergent resistance.[13]
Week 96 data from the TANGO study evaluating Dovato in treatment-experienced HIV-1 adults: The TANGO study was established to determine if virally suppressed people living with HIV can reduce the number of medicines in their HIV treatment regimen, while maintaining viral suppression.[14] The two-year data will provide further indication of Dovato's efficacy, safety and tolerability in this group of participants, following 48-week data which showed non-inferior efficacy for Dovato compared to continuation of a tenofovir alafenamide fumarate (TAF)-containing regimen of at least three drugs, with no confirmed virologic withdrawals and no resistance in the Dovato arm of the study.[15]
Week 24 findings from the STAT study assessing dolutegravir plus lamivudine in a Test and Treat setting: The Phase IIIb STAT study evaluates the feasibility, efficacy and safety of Dovato in a rapid Test and Treat model of care increasingly seen in clinical practice, with treatment initiated within 14 days of diagnosis before baseline HBV co-infection status, renal function and resistance test results were available.[3]
Week 124 findings from Phase 3 FLAIR study of investigational cabotegravir and rilpivirine long-acting: The FLAIR study was designed to assess non-inferiority of the once-monthly regimen of long-acting cabotegravir and rilpivirine compared to daily, oral Triumeq (abacavir/dolutegravir/lamivudine).[16] Data to be presented at HIV Glasgow 2020 will provide insights into the tolerability, safety and efficacy of long-acting cabotegravir and rilpivirine maintenance therapy for people living with HIV-1 infection who switched at Week 100 from the Triumeq arm to the regimen of cabotegravir and rilpivirine.[4]
Multivariable and baseline factor analyses across ATLAS, FLAIR, and ATLAS-2M influencing viral response to investigational long-acting cabotegravir and rilpirivine: The Phase 3 development programme of long-acting cabotegravir and rilpivirine was designed to demonstrate its efficacy and safety when dosed once-monthly in ATLAS and FLAIR, and every two months in ATLAS-2M. Findings to be presented at HIV Glasgow 2020 will provide further insights from the clinical development programme into the participant and viral factors that influence virologic outcomes to long-acting cabotegravir and rilpivirine.[5]
A 48-week subgroup analysis in women from the ATLAS-2M study of investigational long-acting cabotegravir and rilpivirine administered every two months compared to once monthly: The ATLAS-2M study was designed to assess the non-inferiority and safety of long-acting cabotegravir and rilpivirine administered every two months compared to once-monthly administration.[17] This subgroup analysis will compare the outcomes of women who participated in the ATLAS-2M study, which previously demonstrated in 48-week data that long-acting cabotegravir and rilpivirine administered every two months was non-inferior and as safe as its once-monthly dosing.[18] This analysis of 280 women across 13 countries will further characterize this investigational treatment in key populations who are often underrepresented in clinical trials and is reflective of ViiV Healthcare's commitment to all people living with HIV.[6]
Positive Perspectives Wave 2 study results: The data will reveal differences in perceptions of HIV care based on age, gender and sexual orientation with a special focus on Europe in one of the posters. The Positive Perspectives study describes the experiences and concerns of more than 2,300 people living with HIV from 25 countries. At HIV Glasgow 2020, the data presented will show how these perceptions of HIV care can impact their health - from worries about treatment side effects to confiding in healthcare providers and self-care.[7],[8],[9],[10]
The full list of ViiV Healthcare data to be presented at HIV Glasgow is outlined below:

About Dovato (dolutegravir/lamivudine)
Dovato is a once-daily, single-pill, 2-drug regimen (2DR) that combines the integrase strand transfer inhibitor (INI) dolutegravir (Tivicay, 50 mg) with the NRTI lamivudine (Epivir, 300 mg).[19]
Dovato (dolutegravir 50 mg/ lamivudine 300 mg tablets) is authorised in the EU for the treatment of HIV-1 infection in adults and adolescents above 12 years of age weighing at least 40 kg, with no known or suspected resistance to the INI class, or lamivudine. In the US, Dovato is indicated as a complete regimen to treat HIV-1 infection in adults with no ARV treatment history or to replace the current ARV regimen in those who are virologically suppressed (HIV-1 RNA <50 copies/mL) on a stable ARV regimen with no history of treatment failure and no known resistance to any component of Dovato.[20]
Like a dolutegravir-based three-drug regimen, Dovato uses two drugs to inhibit the viral cycle at two different sites. INIs, like dolutegravir, inhibit HIV replication by preventing the viral DNA from integrating into the genetic material of human immune cells (T-cells). This step is essential in the HIV replication cycle and is also responsible for establishing chronic infection. Lamivudine is an NRTI that works by interfering with the conversion of viral ribonucleic acid (RNA) into deoxyribonucleic acid (DNA) which in turn stops the virus from multiplying.[19]
Link to full press release
https://www.gsk.com/en-gb/media/press-releases/viiv-healthcare-to-present-long-term-safety-and-efficacy-data-for-2-drug-regimen-dovato-dolutegravirlamivudine-alongside-other-key-research-advances-at-the-hiv-glasgow-2020-congress/
|
| |
|
 |
 |
|
|