 |
 |
 |
| |
Virologic and CD4 Responses to DTG Generally Steady With Advancing Age
|
| |
| |
Efficacy and safety of dolutegravir in treatment-naive people living with HIV-1 stratified by age: meta-analysis of 48-week results from ARIA, FLAMINGO, SINGLE, and SPRING-2
HIV Drug Therapy/Glasgow 2020, October 5-8, 2020
Mark Mascolini
In a 4-study analysis of antiretroviral-naive people starting dolutegravir (DTG) in a randomized trial, people between 50 and 65 years old had virologic and CD4 responses similar to people under 50 [1]. The virologic response rate to DTG was a little lower in people 65 or older than in younger people, but that result is hard to interpret because of the few 65+ people in this analysis.
Antiretroviral response data on people 50 or older remain limited, and data on people 65 or older are even more scarce. To offer a clearer picture of DTG response rates and other outcomes in older versus younger people with HIV, ViiV Healthcare researchers and collaborators pooled results of four trials that randomized antiretroviral-naive people to DTG or another regimen: ARIA, FLAMINGO, SINGLE, and SPRING-2 (ClinicalTrials.gov identifiers NCT01910402, NCT01449929, NCT01263015, and NCT01227824.) Respective comparator antiretrovirals in those trials were atazanavir/ritonavir, darunavir/ritonavir, efavirenz, and raltegravir. The trials combined DTG with either abacavir/lamivudine or tenofovir disoproxil fumarate/emtricitabine.
The pooled analysis included 2634 participants divided into three age groups: under 50 (1157 on DTG, 1158 on non-DTG), 50 to 64 (148 on DTG, 148 on non-DTG), and 65 or older (10 on DTG and 13 on non-DTG). Proportions in the DTG and non-DTG arms with a pretreatment viral load above 100,000 copies were 28% and 28% in people under 50, 34% and 29% in people 50 to 64, and 60% and 38% in people 65 or older. Proportions in the DTG and non-DTG groups with starting CD4 counts of 350 or higher were 52% and 54% in people under 50, 46% and 55% in people 50 to 64, and 20% and 54% in people 65 or older.
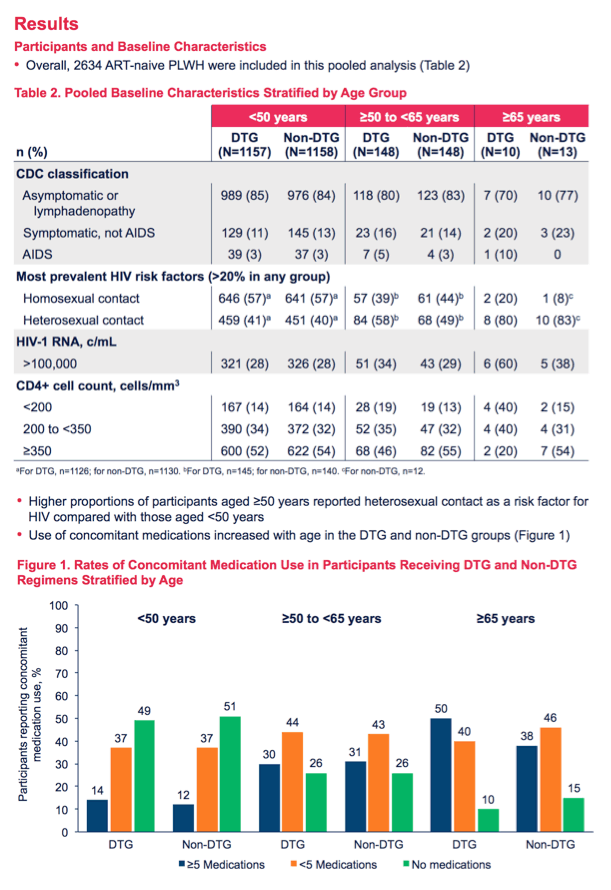
Among people under 50 years old, 14% randomized to DTG and 12% randomized to non-DTG regimens took 5 or more non-HIV medications. In people 50 to 64, those rates jumped to 30% in the DTG group and 31% in the non-DTG group. And in people 65 or older, the rates rose to 50% in the DTG group and 38% in the non-DTG group.
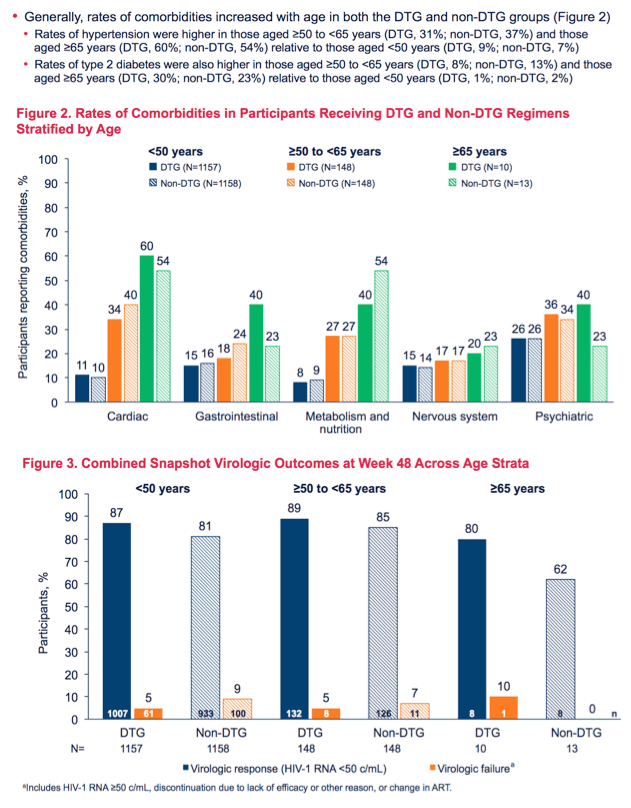
Comorbidity rates also climbed with age across the three age group. For example, hypertension was more frequent in the 50-64 group (31% on DTG, 37% on non-DTG) and in the 65+ group (60% on DTG, 54% on non-DTG), than in the under-50 group (9% on DTG, 7% on non-DTG). For type 2 diabetes, rates were 1% on DTG and 2% on non-DTG in the under-50 group, 8% on DTG and 13% on non-DTG in the 50-64 group, and 30% on DTG and 23% on non-DTG in the 65-or-older group.
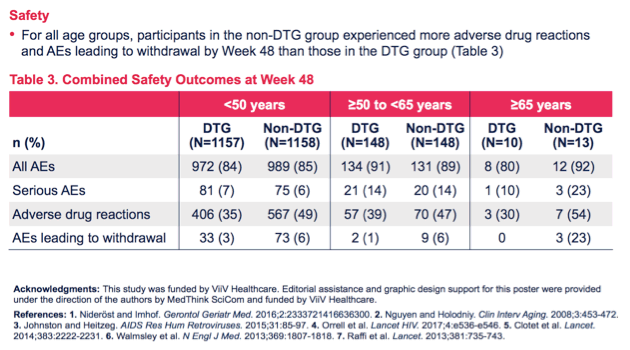
Across the three age strata, 48-week virologic response rates were similar with DTG and non-DTG regimens in people under 50 years old (87% and 81%) and in those 50 to 64 (89% and 85%). The 48-week virologic response rate was higher with DTG than non-DTG regimens in people 65 or older (80% versus 62%), but in that age group only 10 people took DTG and 13 took non-DTG regimens.
Average CD4 gains through 48 weeks were robust with all study regimens across the three age groups: under 50 years old, 250.7 on DTG and 230.0 on non-DTG; 50 to 64 years, 249.6 on DTG and 235.9 on non-DTG; 65 or older, 234.1 on DTG and 112.6 on non-DTG combinations.
Adverse event rates were generally similar with DTG and non-DTG regimens in the under-50 group (serious adverse events 7% on DTG and 6% on non-DTG), in the 50-to-64 group (14% in both treatment arms), and in the 65-and-older group (10% on DTG [1 person] versus 23% [3 people] on non-DTG combinations. Adverse drug reaction rates ranged from 30% to 54% across the age groups with only small differences between age groups or between treatment arms.
Reference
1. Spinelli F, Prakash M, Slater J, et al. Efficacy and safety of dolutegravir in treatment-naive people living with HIV-1 stratified by age: meta-analysis of 48-week results from ARIA, FLAMINGO, SINGLE, and SPRING-2. HIV Drug Therapy/Glasgow 2020, October 5-8, 2020. Abstract P010.


|
| |
|
 |
 |
|
|