 |
 |
 |
| |
Impact of adherence on viral suppression with bictegravir
and dolutegravir (DTG) containing triple therapy in clinical practice
|
| |
| |
Adherence Affects Response to Dolutegravir (but not Bictegravir) Regimens
HIV Drug Therapy/Glasgow 2020, October 5-8, 2020
Mark Mascolini
Analysis of more than 2200 US residents with HIV linked better antiretroviral adherence to better viral suppression when switching to a dolutegravir regimen, but response to a replacement bictegravir regimen remained consistent across adherence brackets [1]. Six months after the switch, viral suppression was high and virtually identical with dolutegravir regimens and the bictegravir regimen.
Paul Sax (Brigham and Women's Hospital, Harvard) and collaborators across the United States pointed out that single-tablet bictegravir plus emtricitabine/tenofovir alafenamide (BIC/FTC/TAF) and dolutegravir-based 3-drug regimens had comparable efficacy in clinical trials. They conducted this analysis of US patients in practice to see if adherence differed between bictegravir and dolutegravir regimens and whether adherence affected virologic response.
The analysis relied on data from the Trio Health HIV EMR and dispensing database. Researchers focused on people switching from a stable antiretroviral combination to single-tablet BIC/FTC/TAF, single-tablet dolutegravir/lamivudine/abacavir (DTG/3TC/ABC), or a dolutegravir multitablet regimen. Participants had to be at least 18 years old at HIV diagnosis, have a viral load below 200 copies when switching regimens, have at least 6 months of observation before the switch, and have viral load measured 6 months after the switch.
The investigators defined high adherence as proportion of days covered at least 80% or at least 95%. They used a negative binomial model adjusted for gender, race, and CD4 count at the switch to assess associations between high adherence and viral suppression 6 months after the switch.
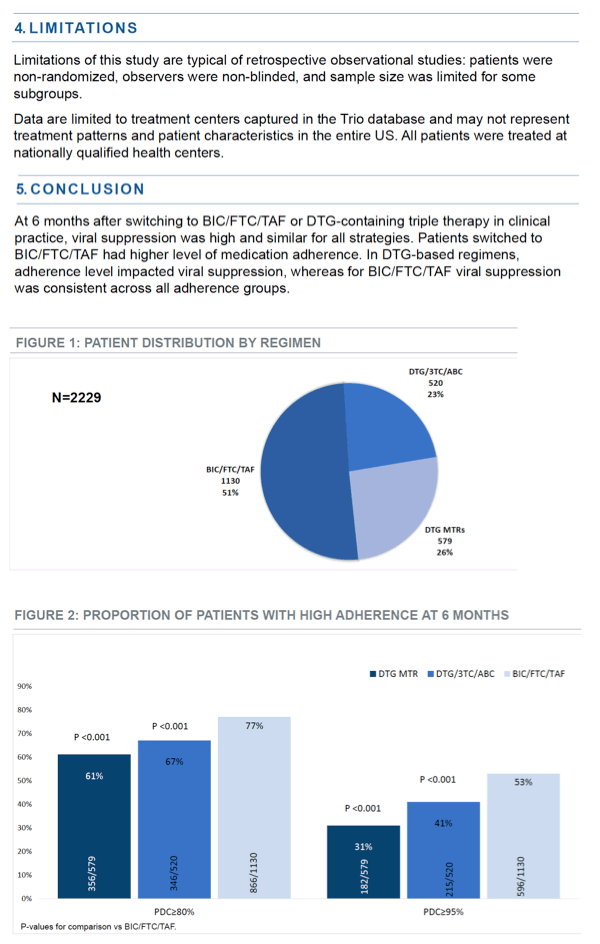
Among 2229 people eligible for the analysis, 1130 (51%) switched to BIC/FTC/TAF, 520 (23%) switched to single-tablet DTG/3TC/ABC, and 579 (26%) switched to dolutegravir multitablet regimens. Proportions of men in those three groups were 75%, 81%, and 69%; proportions of blacks were 25%, 33%, and 36%; and about 45% of all three groups were older than 50. About 5% across the three groups had a CD4 count below 200 at the switch.
Six months after the switch, proportions with at least 80% adherence were 77% with BIC/FTC/TAF, 67% with DTG/3TC/ABC, and 61% with a multitablet dolutegravir combination (P < 0.001 for either dolutegravir regimen vs BIC/FTC/TAF). Proportions with 95% or better adherence were 53% with BIC/FTC/TAF, 41% with DTG/3TC/ABC, and 31% with a multitablet dolutegravir combination (P < 0.001 for either dolutegravir regimen vs BIC/FTC/TAF).
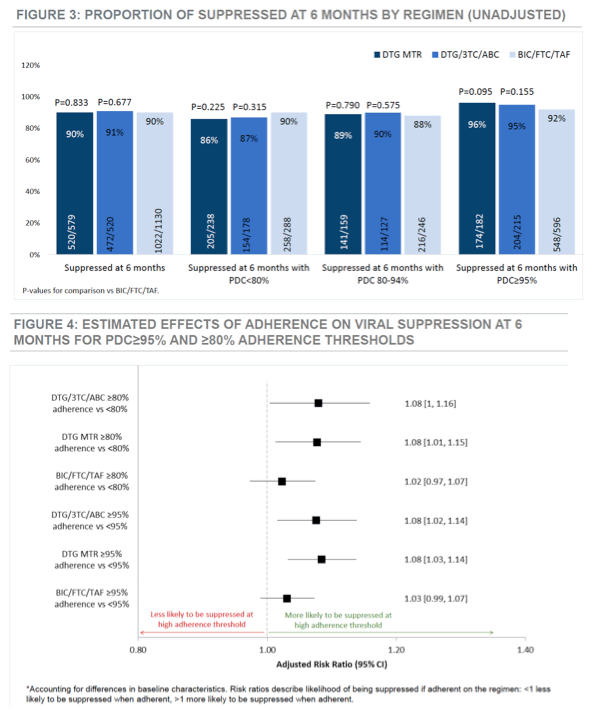
Proportions of participants with viral suppression 6 months after starting the new regimen were virtually identical with BIC/FTC/TAF (90%), DTG/3TC/ABC (91%), and a dolutegravir multitablet combination (90%). And rates of 6-month viral suppression did not differ much between these three groups regardless of adherence status: below 80% adherence, 80% to 94% adherence, or 95% or better adherence.
But good adherence, either above 80% or above 95%, independently predicted viral suppression at 6 months with either dolutegravir regimen, but not with the bictegravir regimen. In other words, better adherence meant a better chance of viral suppression with the dolutegravir regimens, but response did not depend on adherence with BIC/FTC/TAF.
The researchers cautioned that results may not apply to everyone in the United Sates, because data came only from centers reflected in the Trio database. But all centers were nationally qualified health centers.
References
1. Sax PE, Althoff KN, Eron JJ, et al. Impact of adherence on viral suppression with bictegravir and dolutegravir (DTG) containing triple therapy in clinical practice. HIV Drug Therapy/Glasgow 2020, October 5-8, 2020. Abstract P029.
2. Trio Health. Interactive MDX Research Platform. https://www.triohealth.com/mdx

Paul E. Sax consults for Gilead Sciences, ViiV Healthcare, Merck, Janssen. He received research grants from Gilead Sciences, Merck, and ViiV Healthcare. Keri N. Althoff previously served on a Medical Advisory Board for Gilead Sciences. Joseph J. Eron consults for Merck, ViiV Healthcare, Gilead Sciences, and Janssen. The University of North Carolina receives research funding from ViiV Healthcare, Gilead Sciences, and Janssen from which he receives support as an investigator. Janna Radtchenko is employed by Trio Health. Helena Diaz-Cuervo is employed by Gilead Sciences. Karam Mounzer advises for ViiV, Merck, Janssen, Gilead Sciences. He is on the speakers' bureau for ViiV, Merck, Janssen, Gilead Sciences, Clinical care options, and Simply speaking, Prime. Mounzer has received research grants fromViiV, Merck, Janssen, Gilead Sciences. He is on the advisory board for Epividian. Moti Rampogal is a consultant for ViiV Healthcare, Gilead Sciences, Janssen, and Merck and received research funding from these companies. He is a speaker for ViiV Healthcare, Gilead Sciences, and Janssen. Steven Santiago serves on the Medical Advisory Board for Gilead and is a Speaker for Gilead and Janssen. Richard A. Elion received grants from Gilead Sciences and Proteus, serveson the Advisory boards for Gilead Sciences and ViiV Healthcare, and is a speaker for Gilead Sciences and Janssen. Drs. Sax, Althoff, Elion, Santiago, and Eron serve on Trio Health's Scientific Advisory Board
|
| |
|
 |
 |
|
|