 |
 |
 |
| |
Rapid DTG/3TC Safe and Usually Effective in US Test-and-Treat Trial
|
| |
| |
HIV Drug Therapy/Glasgow 2020, October 5-8, 2020
Mark Mascolini
Starting dolutegravir/lamivudine (DTG/3TC) promptly after HIV diagnosis proved safe and usually effective through 24 weeks, according to results of the US STAT trial [1]. Eight participants had to modify the DTG/3TC regimen when testing showed HBV infection or the M184V mutation, or when a rash arose. People with higher viral loads when entering the trial tended to have a worse virologic response rate, but numbers in those subgroups are small.
US and other antiretroviral guidelines recommend DTG/3TC for first-line therapy, except in people with HBV infection,* with a viral load above 500,000 copies, or without resistance test results. Those exclusions would prohibit DTG/3TC in a test-and-treat protocol that calls for treatment before results of the needed tests become available. To address the feasibility of DTG/3TC use in test-and-treat scenarios, US researchers planned the STAT study, a multicenter, open-label, single-arm trial [2].
Participants were at least 18 years old and diagnosed with HIV within 14 days of entering the trial, without lab results available at that point. The DTG/3TC regimen could be altered if test results later indicated HBV infection, genotypic resistance to DTG or 3TC, or creatinine clearance below 30 mL/min. Anyone with a regimen adjustment stayed in the study.
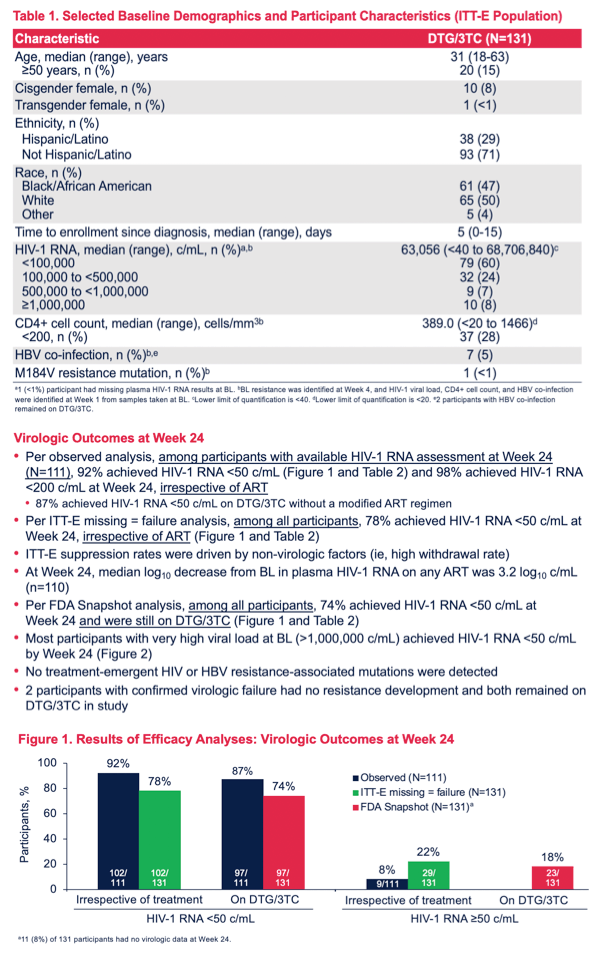
Primary study outcomes were proportions of participants with a viral load below 50 copies at 24 weeks in (1) an intention-to-treat-exposed analysis, with missing data meaning failure (proportion of all participants below 50 copies at week 24, regardless of regimen), (2) observed analysis (proportion of participants below 50 copies at week 24 in those with a viral load at that point, regardless of regimen), and (3) FDA Snapshot analysis (proportion of all participants below 50 copies at week 24 still taking DTG/3TC).
STAT enrolled 131 people at 16 sites, including 10 (8%) cisgender females, 1 transgender female, 38 (29%) Hispanics, 61 (47%) blacks, and 65 (50%) whites. Median age stood at 31 years (range 18 to 63), and 20 people (15%) were 50 or older. A median of 5 days passed between HIV diagnosis and enrollment (range 0 to 15). Median viral load measured 63,056 copies, 24% had a viral load between 100,000 and 500,000 copies, 7% had between 500,000 and 1 million copies, and 8% had 1 million copies or more. Median CD4 count stood at 389 (range less than 20 to 1466).
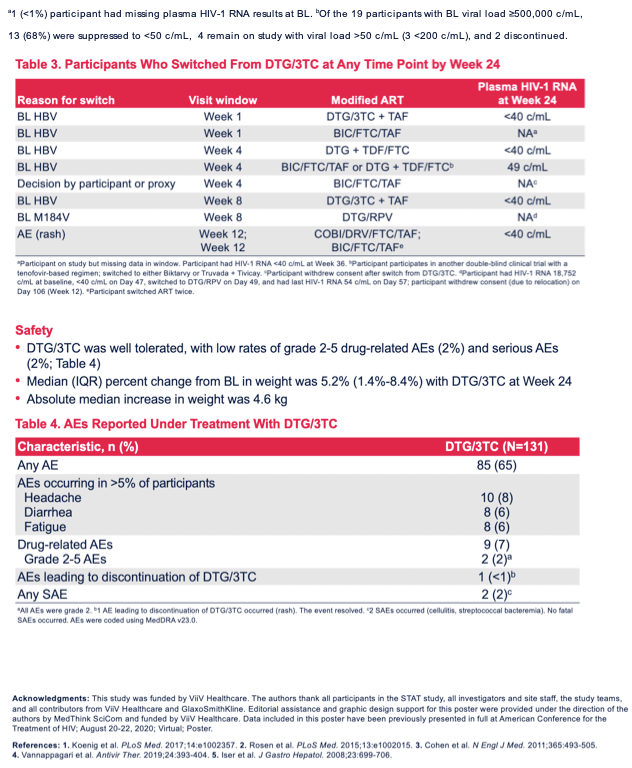
After trial entry, testing revealed that 7 participants (5%) had HBV infection and 1 had the M184V mutation, which makes HIV resistant to 3TC. Eight people modified their DTG/3TC regimen, 5 because of HBV infection, 1 because of the M184V resistance mutation, 1 because of rash, and 1 because of withdrawing from the trial.
In the intention-to-treat missing-equals-failure analysis, 102 of 131 people (78%) had a week-24 viral load below 50 copies. In the observed analysis, 102 of 111 people (92%) had a sub-50-copy load at week 24. In the FDA Snapshot analysis, 97 of 131 people (74%) had a load below 50 copies at week 24. Everyone who changed their regimen and had a viral load test at week 24 had a load below 50 copies. No resistance mutations emerged in either of the 2 people with confirmed virologic failure, who remained on DTG/3TC throughout the study.
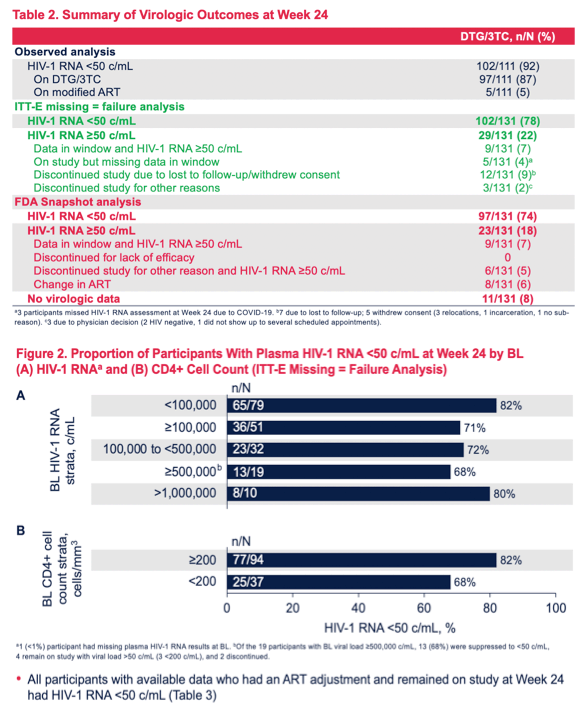
Proportions of participants with a 24-week load under 50 copies trended lower with higher pretreatment viral loads, but numbers of people in these subgroups are small:
-- Pretreatment below 100,000 copies: 65 of 79 (82%) under 50 copies
-- Pretreatment above 100,000 copies: 36 of 51 (71%) under 50 copies
-- Pretreatment 100,000 to 500,000 copies: 23 of 32 (72%) under 50 copies
-- Pretreatment above 500,000 copies: 13 of 19 (68%) under 50 copies
-- Pretreatment above 1 million copies: 8 of 10 (80%) under 50 copies
Nine of 131 people (6.9%) had drug-related adverse events, only 2 of them (1.5%) grade 2 to 5. One person had an adverse event (rash) that led to stopping DTG/3TC. There were 2 serious adverse events (cellulitis and streptococcal bacteremia). Median weight rose 5.2%, reflecting an absolute median gain of 4.6 kg (10 pounds).
The STAT investigators think their findings "demonstrate the feasibility and safety of using DTG/3TC as a first-line regimen in a test-and-treat (rapid ART) setting." They believe their results show that treatment can be adjusted in time if testing uncovers pretreatment resistance or HBV infection after DTG/3TC begins.
*Using 3TC as the only anti-HBV agent in people with HBV infection can lead to rapid emergence of resistant virus.
References
1. Rolle CP, Berhe M, Singh T, et al. Feasibility, efficacy, and safety of using dolutegravir/lamivudine (DTG/3TC) as a first-line regimen in a test-and-treat setting for newly diagnosed PLWH: the STAT study. HIV Drug Therapy/Glasgow 2020, October 5-8, 2020. Abstract P020.
2. ClinicalTrials.gov. Rapid test and treat dolutegravir plus lamivudine study in newly diagnosed human immunodeficiency virus (HIV)-1 infected adults. ClinicalTrials.gov identifier NCT03945981.

|
| |
|
 |
 |
|
|