| |
|
High mortality rates for SARS-CoV-2 infection in patients with pre-existing chronic liver disease and cirrhosis: Preliminary results from an international registry
|
| |
| |
Download the PDF here
Sept 1 2020 Jnl of Hepatology
To the Editor:
Chronic liver disease (CLD) and cirrhosis are common conditions associated with immune dysregulation, leading to concerns that these patients are at increased risk of complications from COVID-19 resulting from infection with SARS-CoV-2. However, the effects of COVID-19 among patients with pre-existing liver disease are currently undefined.
We report the outcomes of the first 152 consecutive submissions of clinician-reported cases of laboratory-confirmed COVID-19 in patients with CLD to two international reporting registries (COVID-Hep.net and COVIDCirrhosis.org) between 25 March 2020 and 20 April 2020. Our combined database includes 103 patients with cirrhosis and 49 with non-cirrhotic CLD from 21 countries across 4 continents (59.9% male, median age 61 years, aetiology: 22.4% non-alcoholic fatty liver disease, 19.7% alcohol, 11.8% hepatitis B, 10.5% hepatitis C, 35.6% other/combination).
Contributors were encouraged to enter data at the end of the patient's disease course. For patients admitted to hospital, cases were only included in the analysis if a definitive outcome of death or discharge was reported. 95.2% of patients with cirrhosis were hospitalised with a median length of hospital stay until discharge or death of 10 days (IQR 5–14 days). Outcomes for patients with cirrhosis included admission to intensive care unit (ICU) in 23.3%, invasive ventilation in 17.5%, non-invasive ventilatory support in 18.6%, renal replacement therapy in 4.9% and death in 39.8%. Mortality far exceeded that reported in unselected populations, hospitalised patients with cirrhosis in the era preceding COVID-19, and in patients with cirrhosis admitted with influenza.
In patients not admitted to ICU, 59.5% had non-severe disease, 27.8% had severe disease but escalation of care was deemed inappropriate, and 3.7% were considered to require ICU but were not admitted due to lack of availability. Targeted antiviral therapy was used in 38.1% of total cases. The most frequently used treatments were chloroquine/hydroxychloroquine (23.0%), lopinovir/ritonavir (6.6%), tocilizumab (3.3%), and interferon-alpha (3.3%).
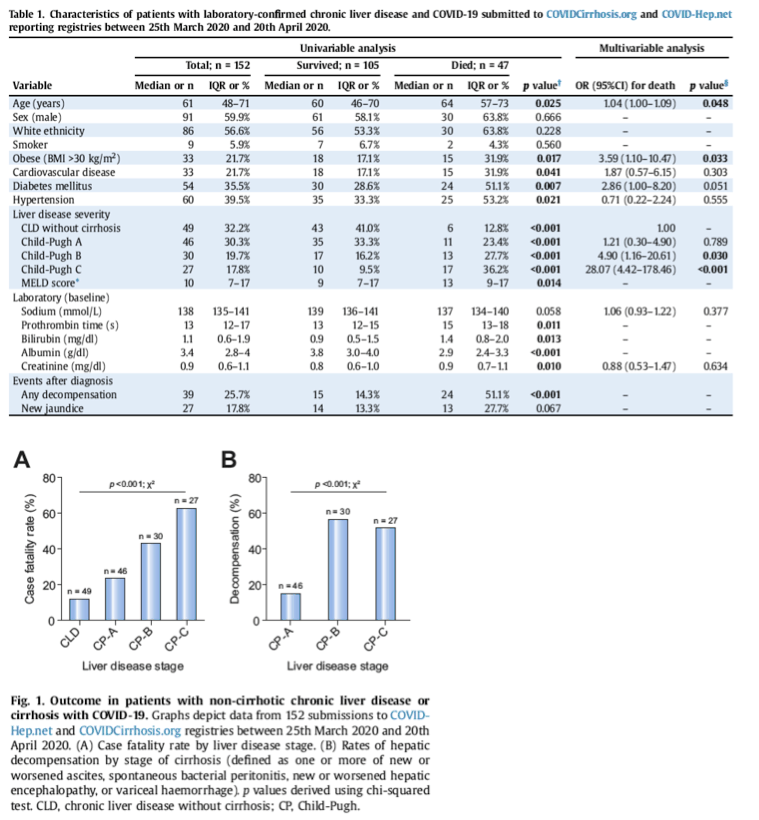
Cause of death in patients with cirrhosis was reported as COVID-19 lung disease in 78.7%, cardiac-related in 4.3%, and liver-related in 12.2%. Risk factors for poor COVID-19 outcomes in the general population, including advanced age, obesity, renal impairment, heart disease, and diabetes mellitus, were over-represented among those who died, although male sex and non-white ethnicity were not. Mortality correlated strongly with baseline Child-Pugh class and model for end-stage liver disease (MELD) score (Table 1).
Deaths occurred in 12.2% of patients with CLD without cirrhosis, 23.9% with Child-Pugh class A cirrhosis, 43.3% with Child-Pugh class B cirrhosis, and 63.0% with Child-Pugh class C cirrhosis (Fig. 1A). Child-Pugh class B and C cirrhosis remained associated with death after adjusting for baseline characteristics including comorbidities (Table 1). Child-Pugh class B and C cirrhosis remained significant predictors of mortality when analysis was restricted to those with cirrhosis.
Hepatic decompensation occurred in 36.9% and was associated with baseline Child-Pugh class (Fig. 1B). Decompensation events included worsening ascites (27.2%), spontaneous bacterial peritonitis (2.9%), hepatic encephalopathy (16.5%), and variceal haemorrhage (1%). Hepatic decompensation during COVID-19 was strongly associated with a subsequent risk of death: 63.2% of those with new decompensation died compared to 26.2% of those without new decompensation. Notably, 24.3% of those with new hepatic decompensation had no respiratory symptoms of COVID-19 at the time of diagnosis.
This large, multicentre, international cohort of patients with chronic liver disease and cirrhosis allows for in-depth assessment of the clinical factors associated with poor outcomes from COVID-19. Accepting that data from registries are subject to selection bias, preliminary findings suggest that baseline liver disease severity is strongly associated with COVID-19-related morbidity and mortality. Furthermore, many SARS-CoV-2-infected patients with cirrhosis experienced hepatic decompensation even in the absence of respiratory symptoms. These findings have important implications for clinicians regarding risk stratification and prognostication for patients with cirrhosis and COVID-19 and suggest the need to maintain a low threshold for SARS-CoV-2 testing in the presence of new hepatic decompensation.
| |
| |
| |
|
|
|