| |
FDA approves drug that lowers cholesterol in a new way
Efficacy and Safety of Bempedoic Acid in Patients With Hypercholesterolemia and Statin Intolerance
|
| |
| |
Originally published29 Mar 2019
Download the PDF here
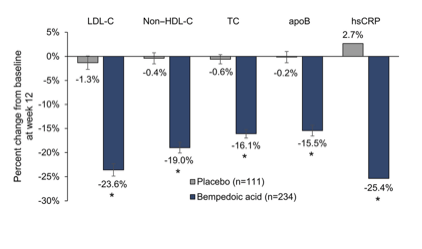
The main finding of the CLEAR Serenity trial is that bempedoic acid significantly reduces both LDL-C and hsCRP compared with placebo and is well tolerated in patients with statin intolerance.
Methods and Results
The phase 3, double-blind, placebo-controlled CLEAR (Cholesterol Lowering via Bempedoic acid, an ACL-Inhibiting Regimen) Serenity study randomized 345 patients with hypercholesterolemia and a history of intolerance to at least 2 statins (1 at the lowest available dose) 2:1 to bempedoic acid 180 mg or placebo once daily for 24 weeks. The primary end point was mean percent change from baseline to week 12 in low-density lipoprotein cholesterol. The mean age was 65.2 years, mean baseline low-density lipoprotein cholesterol was 157.6 mg/dL, and 93% of patients reported a history of statin-associated muscle symptoms. Bempedoic acid treatment significantly reduced low-density lipoprotein cholesterol from baseline to week 12 (placebo-corrected difference, -21.4% [95% CI, -25.1% to -17.7%]; P<0.001). Significant reductions with bempedoic acid versus placebo were also observed in non-high-density lipoprotein cholesterol (-17.9%), total cholesterol (-14.8%), apolipoprotein B (-15.0%), and high-sensitivity C-reactive protein (-24.3%; P<0.001 for all comparisons). Bempedoic acid was safe and well tolerated. The most common muscle-related adverse event, myalgia, occurred in 4.7% and 7.2% of patients who received bempedoic acid or placebo, respectively. Significant reductions in LDL-C at week 12 with bempedoic acid versus placebo were observed in all subgroups, including baseline LDL-C, history of diabetes mellitus, age, race, sex, body mass index, background lipid-modifying therapy, and cardiovascular disease risk category (P≤0.01; Figure 5).
FDA approves drug that lowers cholesterol in a new way
Nexletol was approved for people genetically predisposed to have sky-high cholesterol and people who have heart disease and need to further lower their bad cholesterol.
https://www.nbcnews.com/health/health-news/fda-approves-drug-lowers-cholesterol-new-way-n1140646
Feb. 21, 2020
By Associated Press
TRENTON, N.J. - U.S. regulators on Friday approved a new type of cholesterol-lowering drug aimed at millions of people who can't tolerate - or don't get enough help from - widely used statin pills like Lipitor and Crestor.
The Food and Drug Administration approved Esperion Therapeutics Inc.'s Nexletolfor people genetically predisposed to have sky-high cholesterol and people who have heart disease and need to further lower their bad cholesterol. The daily pill is to be taken in conjunction with a healthy diet and the highest statin dose patients can handle, the FDA said.
High LDL, or bad cholesterol, is one of the top risks for heart attacks and other problems. Studies showed that Nexletol could lower LDL by about 25% when taken alone and by an additional 18% when combined with a statin.
Esperion, which is based in Ann Arbor, Mich., said the drug's brand name will be Nexletol. Esperion also has applied to the FDA for approval of a combination pill of bempedoic acid and ezetimibe, a non-statin pill approved in 2002. In studies, the combination pill reduced cholesterol by about 38 to 44 percent, depending on whether a person was taking a statin and how much, the company said. The FDA decision is expected shortly.
Statins, which were introduced in the 1980s, are the premier medicines for high cholesterol and typically are the first drugs prescribed along with changes in diet and exercise. But they can cause muscle aches and cramps, making it hard for some people to take the required dose, said Robert Rosenson, director of cardiometabolic disorders for Mount Sinai Hospital in New York. More than 10 percent of people taking high-dose statins had muscle pain, according to a 2005 study.
Rosenson, who was not involved in the development of bempedoic acid, said the drug reduces cholesterol by a relatively modest amount but could nevertheless be helpful in achieving patients' goals.
empedoic acid also can be used with another class of heart drugs, called PCSK9 inhibitors, which are injectable medications launched in 2015. They can reduce LDL cholesterol sharply when combined with a statin. But they initially cost about $14,000 a year, causing insurers to balk.
Since then, prices have fallen sharply, but uptake remains slow. While the drugs "are highly effective, there are a lot of hassles with prior authorization and they often get denied," said Christie Ballantyne, a cardiologist at Baylor College of Medicine in Houston who ran the late-stage clinical trial for bempedoic acid.
Ballantyne said bempedoic acid, along with the possible new combination with ezetimibe, will give doctors and patients more ways to attack high cholesterol - an approach that has worked to treat high blood pressure. "For blood pressure, we tend to use lots and lots of combinations, often in the same tablet, and have been pretty successful," he said.
The FDA approved bempedoic acid based on its cholesterol-lowering effect. Whether the drug reduces the risk of heart attacks and strokes is being studied as part of a large outcomes trial involving people who cannot tolerate statins. The trial is scheduled to be completed in two years.
Recommended cholesterol levels vary by age and gender, but the "optimal" level for LDL cholesterol is 100 milligrams per deciliter or below, according to the
National Heart, Lung and Blood Institute. Experts say high-risk people, including those who have had a heart attack, should aim for below 70 mg/dl.
Esperion officials did not announce a price Friday but previously indicated it would be about $10 a day, which would make the drug less expensive than the PCSK9 inhibitors but more costly than statins, which are available in cheap generic versions.
"This is a nice alternative" to statins, but those medicines will still be the first choice, said Dr. Christie Ballantyne, Baylor College of Medicine's cardiology chief. He consults for Esperion and helped test the drug.
https://www.washingtonpost.com/health/2020/02/21/fda-approves-first-non-statin-pill-treat-high-cholesterol-almost-two-decades/
Millions of people take cheap, generic statins, but the medicines don't lower LDL cholesterol enough for many patients and others experience side effects such as muscle pain. Other options include Zetia pills, also sold in generic form as ezetimibe for about $13 to $50 a month.
Far fewer patients use Repatha and Praluent, newer drugs that cost $6,000 or more a year. Insurers often restrict coverage of those medicines, which are shots patients give themselves once or twice a month.
Esperion, based in Ann Arbor, Michigan, did not immediately disclose the drug's list price but previously said it planned a price of about $300 per month. Nexletol, also known as bempedoic acid, should be available in late March, the company said.
It works in the liver by blocking an enzyme needed to make LDL. Statins also block cholesterol production in the liver, but in a different way, so the drugs together can reduce LDL more.
The new drug "would be valuable to add to treatment" for patients who can't get their LDL down enough with existing drugs or can't afford the injected ones, Duke Heart Center co-director Dr. Manesh Patel said. Patel, who was not involved in the research, estimated 10% to 15% of his patients might be eligible for Nexletol.
In company-funded studies, common Nexletol side effects included high blood pressure and muscle and joint pain. Serious side effects include tendon rupture. Women who are pregnant or breast-feeding should not take Nexletol because of possible harm to the baby, the FDA said.
In one study of more than 2,200 patients, there were several more deaths from heart problems in the Nexletol group than among those given placebo pills. Patel said that's likely just a random imbalance, not a big concern.
The company has a large study underway to see whether Nexletol can lower the risk of heart attacks and other problems, not just cholesterol. Results are expected late in 2022. The company estimates Nexletol could be appropriate for up to 18 million Americans and more than 100 million people in other countries.
Michael Valant, 71, of League City, Texas, took Nexletol for about 19 months in one study. Valant, a retired global supply chain manager, had a heart bypass operation more than a decade ago and then took a statin for years, but it wasn't helping enough.
Adding Nexletol to that statin during the study cut his bad cholesterol even more.
"It definitely made a difference," Valant said
|
|
| |
| |
|
|
|