| |
Increased Prevalence of Neurocognitive Impairment in Aging People Living With Human Immunodeficiency Virus: The ANRS EP58 HAND 55-70 Study
|
| |
| |
Download the PDF here
25 July 2019 CID
Abstract
Background
There are limited data on the comparative prevalence of neurocognitive impairment (NCI) in aging people living with human immunodeficiency virus (PLHIV) and people not living with HIV.
Methods
This was a cross-sectional study of PLHIV randomly matched by age (±4 years), gender, and education with 5 HIV-uninfected individuals from the CONSTANCES cohort. PLHIV were fluent in French and sequentially included during routine outpatient visits if aged 55-70 years, with HIV viral load <50 copies/mL, and lymphocyte T-CD4 level ≥200 cells/µL in the past 24 and 12 months, respectively. The primary outcome was NCI as defined by the Frascati criteria. Multivariate normative comparison (MNC) and -1.5 standard deviations in ≥2 neurocognitive domains were secondary outcomes of NCI.
Results
Two hundred PLHIV were matched with 1000 controls. Median age was 62 years, and 85% were men. In PLHIV, the median T-CD4 lymphocyte level was 650 cells/µL, and median nadir T-CD4 lymphocyte level was 176 cells/µL. NCI was found in 71 (35.5%) PLHIV and in 242 (24.2%) controls (odds ratio [OR], 1.74; 95% confidence interval [CI], 1.25, 2.41). After adjusting for confounders, HIV remained significantly associated with NCI (OR, 1.50; 95% CI, 1.04, 2.16). Adjusted results were similar with NCI defined by MNC (ORMNC, 2.95; 95% CI, 1.13, 3.50) or -1.5 SD (OR-1.5, 2.24; 95% CI, 1.39, 3.62).
Adjusting for the matching variables only in model 1, HIV was associated with NCI (OR, 1.74; 95% CI, 1.26-2.41).
Further adjusting for confounders, that is, physical activity, diabetes, hypertension, cardiovascular disease, smoking status, CES-D ≥ 17, alcohol consumption, and living alone, HIV remained significantly associated with NCI (OR, 1.5; 95% CI, 1.04-2.16). A sensitivity analysis that excluded individuals with 1 or more missing variables from the models did not substantially change our findings as HIV remained associated with NCI in the final model (OR, 1.84; 95% CI, 1.24-2.70; Table 3).
Conclusions
In this matched study of aging individuals, HIV was significantly associated with an increased risk of NCI after adjusting for major confounders. Results were confirmed with more stringent NCI classifications.
The increased prevalence of cardiovascular risk factors, cannabis consumption, hypertension, and chronic kidney disease is in line with previous studies [16, 33], as well as the increased rate in depressive symptomatology [34]. Multimorbidity, to which aging PLHIV are more prone than their counterparts not living with HIV, has been linked in the general population to mental health issues, including depression [35]. The differences in social behavior are important, particularly in physical activity and living conditions, as well as the rates of depression in our study. These factors may explain, at least in part, the increased prevalence of NCI in PLHIV, but HIV remained significantly associated with NCI after adjusting for them.
Aging is an important factor in the cognitive decline in PLHIV as well as in the general population [36, 37]. There was no interaction between age and HIV on associations with NCI, despite an age span of 15 years. However, the cross-sectional nature of the analysis and a lack of power to address this issue hinder any firm conclusion on a potential interaction, which would benefit from further studies. Also, survival bias limits interaction analysis of age with HIV.
In the COBRA study [24], there was no evidence of accelerated brain pathology in PLHIV who underwent magnetic resonance imaging (MRI) or of neurocognitive performance after a median follow-up of 2 years [too short], but the MACS cohort showed a greater-than-expected effect of aging on episodic memory and motor function in individuals with AIDS [38].
The association between HIV and NCI is probably related to multifactorial etiologies. HIV-specific patterns of brain abnormality on MRI have recently been identified and could be the result of chronic brain immune activation despite controlled HIV viremia [39-41]. ANI was associated with specific frontal white matter atrophy, while MND was characterized by more widespread subcortical atrophy [40]. In another study, long-standing virologically controlled PLHIV were found to have widespread abnormalities in white matter microstructure, with abnormalities correlating with cognitive function and systemic immune activation [41]. In our study, the high rates of past immunosuppression may therefore have significantly impacted neurocognitive outcomes, as previously published [21].
Figure 1. Prevalence of neurocognitive impairment using the Frascati criteria, -1.5 standard deviation cutoff, or multivariate normative comparison classifications according to age strata and human immunodeficiency virus exposure groups. Abbreviations: HIV, human immunodeficiency virus; MNC, multivariate normative comparison; NCI, neurocognitive impairment; SD, standard deviation.
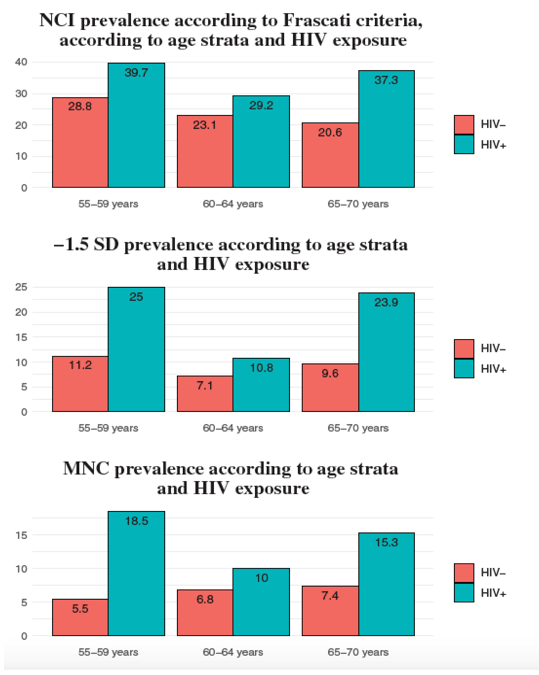
Since the advent of combined antiretroviral therapy (cART), the life expectancy for people living with human immunodeficiency virus (PLHIV) has substantially increased in resource-rich settings [1, 2]. Causes of morbidity and mortality have shifted from AIDS to non-AIDS-defining diseases [3, 4], and studies have found high proportions of neurocognitive impairment (NCI) in PLHIV, ranging from 19% to 67% [5-10]. NCI prevalence has varied widely in studies due to differing demographic characteristics and comorbidity burdens of study individuals, as well as the neurocognitive tests and classifications that were used. In PLHIV taking cART with controlled HIV viremia, the prevalence of NCI has been lower, albeit still substantial, ranging from 19% to 30% [5-7]. The incidence of NCI is expected to rise in PLHIV as the population ages [11]. There are, however, limited data on NCI prevalence or incidence in older PLHIV [5-10].
NCI prevalence may be higher in PLHIV compared to the general population. This could solely reflect the increased prevalence of NCI-associated risk factors in this population [12-17], such as cardiovascular disease or depression. Additionally, HIV infection may also be an additional risk factor and potentially the primary contributor of NCI in PLHIV [18], as studies have shown an association between NCI and HIV-related factors such as detectable viral load [6], current CD4 levels [19], nadir CD4 levels [6, 20], and history of AIDS [21]. However, clinical studies have found contradictory results. In the CIPHER study, there was no significant difference in severity or prevalence of NCI in 248 PLHIV and 45 men who have sex with men (MSM) not living with HIV [22]. In the MACS cohort, there was no association between HIV and NCI in 428 PLHIV and 207 MSM not living with HIV after adjusting for education, depression, and race [23]. The COBRA cohort showed, however, baseline poorer global cognitive performances in 134 virologically controlled PLHIV and 79 demographically similar controls not living with HIV, but the association with HIV was not addressed [24].
Further studies to evaluate the prevalence of NCI in aging PLHIV are thus needed, as well as studies to assess the association between HIV and NCI. In our study, we compared the prevalence of NCI in PLHIV aged 55 to 70 years with controlled viral load with an age, gender, and education matched control population of people not living with HIV, adjusting for a wide variety of confounding factors including behavioral and social factors.
METHODS
Study Design and Populations
The France REcherche Nord&Sud Sida-hiv Hépatites (ANRS) étude physiopathologique (EP) 58 HIV-associated neurocognitive disorder (HAND) 55-70 study was a cross-sectional study that compared the prevalence of NCI in a population of PLHIV matched by age (±4 years), gender, and education level with controls from the general population.
To limit selection bias, PLHIV were systematically and sequentially recruited by their HIV care provider during a routine clinical outpatient visit at 6 French hospitals if all of the following conditions were met: age between 55 and 70 years, living with HIV-1, HIV viral load <50 copies/mL in the past 24 months, a T-CD4 lymphocyte level ≥200 cells/µL in the past 12 months, a signed informed consent, and having health insurance. A maximum of 2 viremic blips, defined by a transient elevation of viral load ≥200 copies/mL, was allowed. Individuals were excluded if they were confused, illiterate, or nonfluent in French; had an ongoing or past history of brain disease with clinical sequelae; had major psychiatric syndromes, sensorial loss, or an extensive neurocognitive evaluation in the prior 6 months; or were vulnerable (under tutorship, guardianship, or imprisoned). The study prespecified that a third of all individuals should be included in each of the following age ranges: 55-59 years, 60-64 years, and 65-70 years.
The matched individuals not living with HIV from the CONSTANCES cohort were recruited from 17 social security health screening centers. These individuals had identical inclusion and exclusion criteria as the PLHIV participants, except for the immunovirological criteria. The CONSTANCES cohort, which included 200 000 individuals in February 2019, is a large, randomly selected cohort of the French population of salaried workers (professionally active or retired) and their families, aged 18-69 years at inception [25]. In this cohort, all individuals aged ≥45 years undergo extensive neurocognitive assessment.
Cognitive, Functional Evaluations and Endpoints
After a centralized CONSTANCES training program, neuropsychologists performed standardized evaluations of functional and neurocognitive capacities and used identical electronic case report forms in both populations [26, 27]. Cognitive functions and functional evaluations were assessed using the trail making test (TMT) A-B for executive function, the digit symbol substitution task of the Wechsler Adult Intelligence Scale-IV (WAIS-IV) for speed of information processing, the digital finger tapping test for motor skills, word fluency and formal lexical and semantic evocation for language, and the free and cued selective reminding test for episodic memory. Functional capacities were evaluated with 4 items of the instrumental activities of daily living (IADL) scale. For our study, we used the baseline neurocognitive assessments.
Norms (mean and standard deviation [SD]) for each test were determined per age strata (55-59, 60-64, and 65 to 70 years) and gender using neurocognitive scores from the CONSTANCES cohort (n =18 740) and were used to calculate age and gender z scores. Impaired functional capacities were defined as mild if 1 item or severe if 2 or more items from the 4-item IADL scale showed reduced capacity. The primary endpoint was the presence of NCI according to the Frascati criteria, used to define HAND in PLHIV [18] (see Supplementary Materials for further details). The Frascati criteria were used solely to compare NCI in both populations and not to attribute NCI to HIV infection, as the controls were not living with HIV.
Frascati criteria with a threshold of less than -1.5 SD from the norm and multivariate normative comparison (MNC) [28] in 2 or more neurocognitive domains were secondary outcomes used to potentially lower the false-positive rates of the Frascati criteria [29]. MNC is able to control the family-wise error (the probability of falsely diagnosing individuals as cognitively abnormal) by making a single multivariate comparison of the complete cognitive profile of an individual with the distribution of all the cognitive profiles of the control sample.
Covariates
Investigators recorded characteristics of both populations using similar self-administered or investigator-led questionnaires, including hypercholesterolemia, diabetes, hypertension, the Center for Epidemiologic Studies-Depression (CES-D) scale for depressive symptomatology (cutoff ≥17) [30], smoking status, alcohol consumption, physical activity, immunovirological characteristics (HIV viral load, T-CD4, and T-CD8 lymphocyte measures), and history of AIDS-defining diseases. Definitions of each covariate are listed in the Supplementary Materials.
Statistical Analyses
We used multiple imputation by chained equations [31] to impute 2.8% of the data (ie, missing values) that otherwise would have led to exclusion of 24% of the individuals. Missing values in outcome variables were not imputed.
First, we described and compared the main characteristics of PLHIV and people not living with HIV using univariable conditional logistic regression and the associated Wald test to take into account the matched design. We then assessed the association between NCI and covariates using mixed univariable logistic regression (with the Wald test), including matching classes as a random effect to account for the intraclass correlation. In this analysis, we tested the effects of sociodemographic (age, gender, education, and living alone), health and behavioral (hypercholesterolemia, diabetes, cardiovascular disease, hypertension, smoking, alcohol consumption, cannabis consumption, and physical activity), and depression variables. We assessed the adjusted association between HAND and covariates using mixed multivariable logistic regression in 2 models. The first included HIV and the variables used for matching nonexposed individuals from the CONSTANCES cohort (age, gender, and education level). The second model added potential confounding factors with a univariable P value <0.20. The first 2 univariable analyses were performed on the crude dataset. Multivariable models were then fitted on 20 imputed datasets, and results were pooled by simulations. For each fit, 500 parameter values were drawn randomly from the posterior distribution (500 × 20 = 10 000 values), and mean parameters with 95% confidence intervals (CIs) were computed. We tested association between HIV and NCI with a variant of the likelihood ratio test for multiple imputed datasets.
The same multivariable approach was used for the -1.5 SD cutoff and the MNC secondary outcomes. A sensitivity analysis used the primary outcome with the same factors in multivariable analysis but excluded individuals with 1 or more missing variables. We also tested for an interaction between age and HIV on NCI, as defined by the 3 possible outcomes.
Our sample size calculation was based on a prior NCI evaluation of 2000 volunteers not living with HIV with the same neurocognitive tests in the GAZEL cohort [32]. Using the Frascati criteria [18], the prevalence of NCI in individuals aged 55-70 years in GAZEL was 17%. We assumed a 27% prevalence of NCI in matched PLHIV under cART with controlled viral load. We initially estimated that 194 PLHIV and 387 controls should be included for an estimated odds ratio (OR) of NCI of 1.8 in PLHIV with a type 1 error of 5% and 80% power. However, to further increase statistical power, we randomly matched 1 PLHIV to 5 controls from the CONSTANCES cohort according to gender, age, and 3 education levels, as numbers of controls accrued were sufficient.
Ethics
Written informed consent was obtained from each patient before enrollment. The protocol was performed in accordance with the Declaration of Helsinki and French law for biomedical research and was approved by the CPP Sud méditerranée I Ethics Committee (Marseille, France). Authorizations for the CONSTANCES cohort were obtained from the ethical review board (IRB INSERM 01-011) and the French Data Protection Authority (CNIL 910486).
RESULTS
In total, 209 PLHIV were included in the study between 26 January 2016 and 27 October 2017. Nine individuals were excluded from analysis-5 did not meet inclusion criteria: 1 had a T-CD4 lymphocyte count of 105 cells/mL; 1 had a viral load of 1600 copies/mL; 1 had important visual sequel of cytomegalovirus retinitis; 1 declared himself autistic, impeding neurocognitive testing; and 1 was administered the wrong tests. Four other individuals did not complete neurocognitive tests or the self-administered questionnaire.
Two hundred PLHIV were randomly matched by age (±4 years), gender, and education level with 1000 individuals from the CONSTANCES cohort. Their characteristics are listed in Table 1. PLHIV significantly differed from the general population as they were less often from mainland France, were more frequently active smokers and cannabis consumers, had increased rates of cardiovascular and chronic kidney disease, had depressive symptomatology (CES-D ≥ 17), were less physically active, more often lived alone, and were more often MSM. Hypercholesterolemia and diabetes were similarly distributed in both populations.
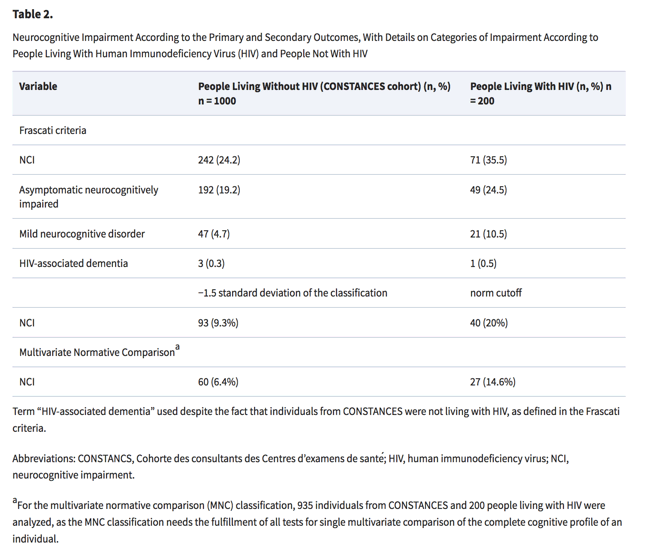
We classified NCI in 71 PLHIV (35.5%) and in 242 controls (24.2%) (OR, 1.74; 95% CI, 1.25-2.41). Distributions of NCI according to the Frascati criteria in both populations are shown in Table 2. Most impaired individuals were asymptomatic neurocognitively impaired (ANI). Only 3 individuals (0.30%) from the CONSTANCES cohort and 1 PLHIV (0.05%) were classified as having HIV-associated dementia. Neurocognitive tests significantly differed between groups in the TMT-A, TMT-B, and WAIS-IV (Supplementary Figure 1).
Adjusting for the matching variables only in model 1, HIV was associated with NCI (OR, 1.74; 95% CI, 1.26-2.41). Further adjusting for confounders, that is, physical activity, diabetes, hypertension, cardiovascular disease, smoking status, CES-D ≥ 17, alcohol consumption, and living alone, HIV remained significantly associated with NCI (OR, 1.5; 95% CI, 1.04-2.16). A sensitivity analysis that excluded individuals with 1 or more missing variables from the models did not substantially change our findings as HIV remained associated with NCI in the final model (OR, 1.84; 95% CI, 1.24-2.70; Table 3).
Using the -1.5 SD from the norm cutoff classification, these numbers were 40 (20.0%) and 93 (9.3%) (unadjusted OR [OR-1.5], 2.47; 95% CI, 1.64-3.73). Using the MNC criteria, 27 (14.6%) PLHIV and 60 (6.6%) controls were impaired (unadjusted OR for MNC [ORMNC], 2.45; 95% CI, 1.51-3.99). In both multivariable analyses, HIV remained associated with cognitive impairment whether defined by -1.5 SD (OR-1.5, 2.24; 95% CI, 1.39-3.62) or MNC (ORMNC, 2.00; 95% CI, 1.13-3.50; Table 3).
Based on the z score, prevalence of NCI using the Frascati criteria, -1.5 SD classification, and MCI classifications according to age strata and HIV exposure groups are shown in Figure 1. No interaction between age and HIV status was objectified with any neurocognitive outcome (Table 4).
|
|
| |
| |
|
|
|