| |
Antiretroviral drug concentrations in brain tissue of adult decedents
|
| |
| |
Download the PDF here
"higher drug concentrations in brain were
associated with worse neurocognitive performance
We also identified associations between drug concentrations in brain tissue and plasma HIV RNA
and neurocognitive performance. The direction of the relationship with plasma HIV RNA suggests that
drug concentrations in brain may reflect those in blood since lower plasma HIV RNA was associated
with higher drug concentrations in brain. The direction of the relationship with neurocognitive
performance, however, indicates that this may come at a cost: higher drug concentrations in brain were
associated with worse neurocognitive performance. This is consistent with recent concerns about ART
neurotoxicity.[26, 27] The quadratic relationship between drug concentrations in brain and CSF HIV RNA
could be artifactual but might also be due to the influence of two factors that were not measured in this
analysis, ART drug resistance and neuroinflammation. Both of these conditions would be more common
in persons with advanced HIV disease or AIDS, such as our participants. When HIV is susceptible to the
administered ART, HIV RNA would be suppressed by high ART drug levels and neuroinflammation
would be relatively low. When ART drug resistant HIV is present in the CNS, high ART drug
concentrations may not suppress HIV RNA in CSF (and the brain), leaving only the toxicity of the drugs
without the benefit of viral suppression. In this scenario, neuroinflammation would be relatively worse,
which could also increase BBB permeability and result in high drug concentrations in the brain. Either
scenario would result in high drug concentrations in the brain but only one would result in HIV
suppression, consistent with our observations.
These analyses identified that higher ART drug concentrations in brain tissue were
associated with longer estimated duration of HIV infection (rho=0.38, p=0.0007), longer duration of
ART (rho=0.23, p=0.042), lower HIV RNA in plasma (rho=-0.40, p=0.0006, Figure 3), worse GDS
(rho=0.40, p=0.0015), and possibly lower nadir CD4+ T-cell count (rho=-0.21, p=0.085).
Analyses that adjusted for ART drug and brain region confirmed the associations with estimated duration of HIV
infection (β=0.797, p=0.015), HIV RNA in plasma (β=-49.3, p=0.0001), and GDS (β=42.3, p=0.017)
with weaker associations with nadir CD4+ T-cell count (β=-0.35, p=0.053) and duration of ART
(β=1.03, p=0.101). Including all of these covariates in a single model identified that higher ART drug
concentrations were most strongly associated with lower HIV RNA in plasma (β=-75.6, p=0.031) and
worse GDS (β=51.6, p=0.016). ART drug concentrations in brain tissue were not associated with age,
sex, race/ethnicity, or current CD4+ T-cell count. The relationship between ART drug concentrations in
brain tissue and HIV RNA in CSF followed a quadratic pattern (R2=0.137, p=0.0036, Figure 3).
AIDS July 17 2020 - Micol Ferrara1, Namandjé N. Bumpus2, Qing Ma3, Ronald J. Ellis4, Virawudh Soontornniyomkij4,
Jerel A. Fields4, Ajay Bharti4, Cristian L. Achim4, David J. Moore4, and Scott L. Letendre4.
1University of Torino, Torino, Italy; 2Johns Hopkins University, Baltimore, Maryland; 3University at Buffalo, Buffalo, New York; 4University of California, San Diego, San Diego, California.
Abstract
Objective:
Determine concentrations of antiretroviral therapy (ART) drugs in the human brain.
Design:
Cohort study of persons with HIV (PWH) who consented to antemortem assessment and postmortem autopsy.
Methods:
Eleven PWH who were taking ART at the time of death and had detectable concentrations of at least one ART drug in intracardiac aspirate at autopsy were evaluated. Autopsies were performed within 24 hours of death and brain tissue was stored at -80oC. Concentrations of 11 ART drugs were measured in three brain regions [globus pallidus (GP), cortical gray matter (CGM), white matter (WM)] by high performance liquid chromatography tandem mass spectrometry with a lower limit of quantification of 25 ng/mL.
Results:
Participants were mostly men (82%) with a mean age of 40.4 years. Drug concentrations in brain tissue were highly variable and exceeded published concentrations in CSF for several drugs, including for tenofovir, efavirenz, and lopinavir. Drug concentrations correlated most strongly between CGM and GP (rho = 0.70) but less well between GP and WM (rho = 0.43). Combining all drugs and brain regions (n = 89), higher drug concentrations in brain were associated with longer estimated duration of HIV infection (p = 0.015), lower HIV RNA in plasma (p = 0.0001), lower nadir CD4+ T-cell count (p = 0.053), and worse neurocognitive performance (p = 0.017).
Conclusions:
This is the first analysis of ART drug concentrations in human brain tissue. Concentrations of several drugs in this analysis were similar to published concentrations in CSF but others exceeded published concentrations. The association between higher drug concentrations in the brain and worse neurocognitive performance may indicate ART neurotoxicity.
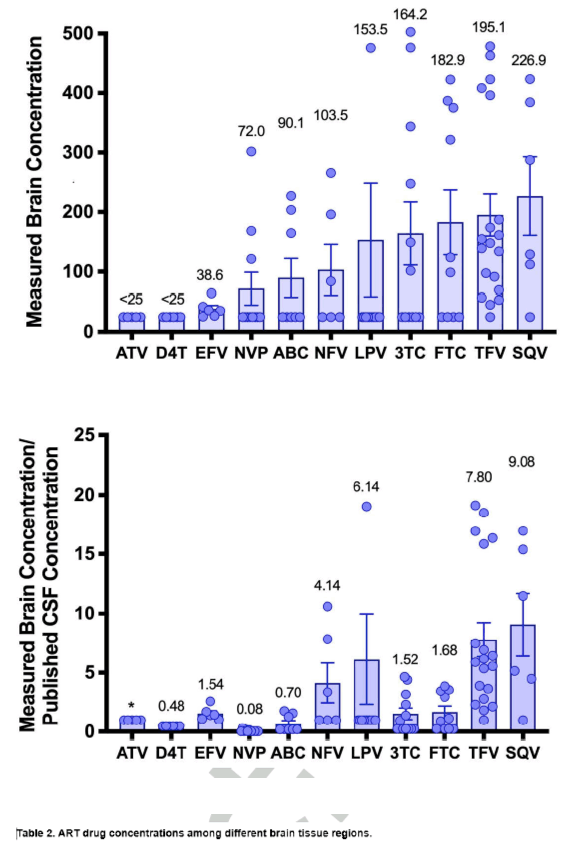
Figure 1. Drug Concentrations in Brain Tissue (left) and the Same Concentrations Divided by
Published Drug Concentrations in CSF (right, see Table 2). The left panel graphically depicts the
substantial variability in concentrations and the differences in concentrations between drugs and the
right panel more clearly depicts that certain drugs appear to concentrate in brain tissue relative to
published CSF concentrations (NFV, LPV, TFV, SQV). Bars are means and error bars are the standard
error of the mean. Values over bars are means. One concentration data point is not shown to better
display the patterns in the data (LPV, 1116 ng/mL). *No value is provided for ATV since both the
measured values and the published CSF concentrations in CSF are below the assay sensitivity (25
ng/mL).
Discussion
In this project, ART drug concentrations were measured in brain tissue that was collected at autopsy
from adults who died with HIV disease. Brain tissue drug concentrations were higher than published
drug concentrations in CSF for at least one drug in every ART class tested, which included NRTIs
(TFV), NNRTIs (EFV), and PIs (LPV and possibly SQV). Brain tissue concentrations exceeded
published concentrations in CSF to the greatest extent for TFV and SQV (and in WM for LPV). ART
drug concentrations in gray matter (GM) (either GP or CGM) appeared to be more similar to each other
than to ART drug concentrations in WM, although these differences did not reach statistical significance
in this small study. If ART drug concentrations in WM do differ from those in GM, this may be due to a
different vascular pattern and lipid content in WM that could influence drug delivery and
accumulation.[16]
In the current ART prescribing environment, the discordance between published TFV concentrations
in CSF and those measured here in brain tissue may be the most impactful finding. TFV has low plasma
protein binding (<1%), which favors distribution into the CNS, but is not highly lipophilic (LogP: -1.6)
and has highly positive polarity that may require active transport across the BBB, characteristics which
do not favor distribution into the CNS. Transporter enzymes that contribute to TFV distribution through
the BBB and BCB include organic anion transporters 1 and 3, and multidrug resistance associated
proteins (MRP) 2 and 4 but not the permeability glycoprotein (P-glycoprotein).[17-19] The different
distribution of the active transporters in these interfaces could influence TFV distribution into the CNS.
One animal study reported good distribution of TFV into CSF without reaching high concentrations
in deep brain tissue[20] and a non-human primate study found comparable zidovudine concentrations in
CSF and brain tissue.[21] More recent analyses in non-human primates found that TFV concentrations in
brain tissue are higher than concentrations in CSF.[11] A potential explanation for high TFV
concentrations in brain tissue despite poor CSF distribution[7] could be related to different expression of
efflux transporters at the blood-brain and blood-CSF barriers.[22] Furthermore, TFV is administered as a
prodrug, which may reach higher intracellular concentrations, which could theoretically result in lower
extracellular drug concentrations, e.g., in CSF.
In addition to TFV, the other drugs that diverged most from reported concentrations in CSF were
PIs, namely LPV, SQV, and NFV. While SQV and NFV are no longer used in clinical settings, LPV is
still sometimes prescribed in low- and middle-income countries. LPV is highly lipophilic (LogP: 5.9), is
a smaller molecule than ATV, and is substantially bound to drug-binding proteins (98-99%). LPV and
other PIs are substrates for molecular transporters that can influence CNS exposure, such as the active
efflux transporter P-glycoprotein.[23] Although LPV concentrations in CSF are much lower than those in
plasma, they appear to be in the therapeutic range[24] even as a monotherapy regimen.[25] Our findings
indicate a potentially more complex situation with LPV concentrations present principally in WM – and
at much higher concentrations than published concentrations in CSF – and below 25 ng/mL in cortical
and deep GM. Since the myelin in WM is lipid-rich, an explanation for the observed substantial LPV
regional variation may its high lipophilicity.
We also identified associations between drug concentrations in brain tissue and plasma HIV RNA
and neurocognitive performance. The direction of the relationship with plasma HIV RNA suggests that
drug concentrations in brain may reflect those in blood since lower plasma HIV RNA was associated
with higher drug concentrations in brain. The direction of the relationship with neurocognitive
performance, however, indicates that this may come at a cost: higher drug concentrations in brain were
associated with worse neurocognitive performance. This is consistent with recent concerns about ART
neurotoxicity.[26, 27] The quadratic relationship between drug concentrations in brain and CSF HIV RNA
could be artifactual but might also be due to the influence of two factors that were not measured in this
analysis, ART drug resistance and neuroinflammation. Both of these conditions would be more common
in persons with advanced HIV disease or AIDS, such as our participants. When HIV is susceptible to the
administered ART, HIV RNA would be suppressed by high ART drug levels and neuroinflammation
would be relatively low. When ART drug resistant HIV is present in the CNS, high ART drug
concentrations may not suppress HIV RNA in CSF (and the brain), leaving only the toxicity of the drugs
without the benefit of viral suppression. In this scenario, neuroinflammation would be relatively worse,
which could also increase BBB permeability and result in high drug concentrations in the brain. Either
scenario would result in high drug concentrations in the brain but only one would result in HIV
suppression, consistent with our observations.
Our study has multiple limitations. The sample size is very small. This project was intended,
however, to be a pilot project and it fulfilled its primary aim, which was to describe ART drug
concentrations in brain tissue from adults who died with HIV. Prior published studies have focused on
animals and have provided valuable insights into ART drug concentrations in the brain, but ultimately
human data are needed to determine how well the animal studies generalize to humans. Another
limitation is the lack of dosing data immediately prior to death. While we attempted to limit the
influence of this by qualitatively measuring drug concentrations in intracardiac aspirate from autopsy as
a coarse indicator of recent dosing, the long half-life of several of the ART drugs means that they could
have been dosed days prior to death. The measured drug concentrations may provide useful information
about the accumulation of ART drugs in the brain but their generalizability to regularly dosed, living
persons is uncertain. Our study also lacked drug concentrations quantitatively measured in plasma and
CSF since autopsies were not performed sufficiently quickly to collect them. While using published
drug concentrations in CSF provided some insights, historical data remains an inferior standard
compared with actual data from the same participants. Finally, the brain tissue used in these analyses
were collected between 2000 and 2006. While the specimens were stored in monitored, -80oC freezers,
the length of storage could affect our results. Also, our study did not include drugs like tenofovir
alafenamide or integrase inhibitors, which are commonly prescribed today. Research characterizing
concentrations of these drugs in the brain are needed.
Conclusions
The manuscript reports ART drug concentrations from human brain tissue collected at autopsy,
finding that they exceed published ART drug concentrations in CSF and that higher concentrations are
associated with viral suppression in blood, longer duration of HIV infection and its treatment, and worse
global neurocognitive performance. Larger studies are needed in which modern ART drugs are
quantified in CSF, brain tissue, and blood to validate these pilot findings and to better understand the
mechanisms of different ART distribution patterns into the CNS.
Results
As summarized in Table 1, participants were mostly middle-aged (mean 40.4 years) men (81.8%) of
European ancestry (54.5%). At the time of last antemortem assessment, the mean duration of the current
ART regimen was 2.0 years (SD 2.9). The median plasma HIV RNA 2.82 log10 c/mL (IQR 2.03-4.32)
and median CD4+ T-cell count was 56 cells/μL (IQR 32.5-120.0). All participants had AIDS at the time
of death with a mean estimated duration of HIV disease of 14 years (SD 5.4) and a median nadir CD4+
T-cell count of 13 cells/μL (IQR 7-53). The most common causes of death were respiratory [pneumonia
(n=3), respiratory failure (n=1), pulmonary embolism (n=1)]. Other causes were disseminated Kaposi’s
Sarcoma (n=1) and hepatocellular carcinoma (n=1). Cause of death was not recorded for two
participants.
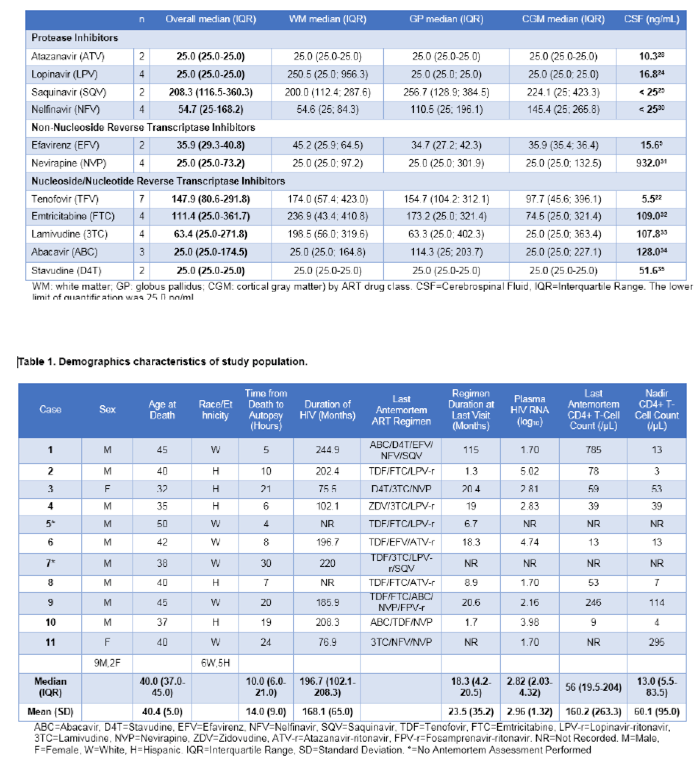
Table 2 summarizes the ART drug concentrations measured in the three brain tissue regions. The
table also includes a summary measure of published concentrations of each ART drug in CSF. Among
nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), median (IQR) concentrations in ng/mL
across all brain regions were 147.9 (80.6-291.8) for TFV, 111.4 (25.0-361.7) for FTC, 63.4 (25.0-271.8)
for 3TC, and 25.0 (25.0-174.5) for abacavir (ABC). All stavudine (D4T) concentrations were below the
lower limit of quantification of the assay. Among protease inhibitors (PIs), median (IQR) concentrations
in ng/mL across all brain regions were 208.3 (116.5-360.3) for saquinavir (SQV), 54.7 (25.0-168.2) for
nelfinavir (NFV), and 25.0 (25.0-25.0) for LPV. All ATV concentrations were below the lower limit of
quantification of the assay. While LPV concentrations were below the lower limit of quantification of
the assay in GP and CGM, high concentrations were present in WM [250.5 (25.0; 956.3)]. Among nonnucleoside
reverse transcriptase inhibitors (NNRTIs), median (IQR) concentrations in ng/mL were 35.9
(29.3-40.8) for EFV and 25.0 (25.0-73.2) for nevirapine (NVP). Figure 1 graphically depicts the range
of concentrations of each drug in brain tissue as well as the ratio of the measured concentration of each
drug in all brain regions to its published concentration in CSF.
Compared with published drug concentrations in CSF, the measured concentrations in brain tissue
were statistically significantly higher for TFV (p<0.001), LPV (p<0.001), and EFV (p=0.031) with a
trend toward higher concentrations of SQV (p=0.06). Brain tissue concentrations were lower than
published drug concentrations in CSF for NVP (p=0.001) and possibly ABC (p=0.10). Brain tissue drug
concentrations did not statistically differ from published drug concentrations in CSF for NFV (p=0.25),
FTC (p=0.43), and 3TC (p=0.74). Comparisons were not performed for drugs that were uniformly below
the lower limit of quantification of the assay (ATV, D4T).
Drug concentrations did not differ between brain regions for individual drugs (all p values > 0.20).
The largest difference was for LPV (p=0.26), particularly comparing WM to other brain regions (mean
175.4 vs. 25.0, p=0.10). Since the power of these comparisons was limited by the small sample sizes, we
combined all regional brain concentrations of all drugs (n=89). Concentrations most strongly correlated
between CGM and either GP (rho=0.70, p<0.0001, Figure 2) or WM (rho=0.66, p<0.0001). The
correlation between drug concentrations in GP and WM was weaker but still statistically significant
(rho=0.43, p=0.018). As shown in Figure 2, most drug concentrations in WM fell above the line of
identity compared with drug concentrations in CGM but this difference was not statistically significant
(p=0.16).
We next combined drug concentration data from all participants and brain regions and performed
exploratory modeling by multivariate regression (N=89 drug concentrations for all participants in all
brain regions). These analyses identified that higher ART drug concentrations in brain tissue were
associated with longer estimated duration of HIV infection (rho=0.38, p=0.0007), longer duration of
ART (rho=0.23, p=0.042), lower HIV RNA in plasma (rho=-0.40, p=0.0006, Figure 3), worse GDS
(rho=0.40, p=0.0015), and possibly lower nadir CD4+ T-cell count (rho=-0.21, p=0.085). Analyses that
adjusted for ART drug and brain region confirmed the associations with estimated duration of HIV
infection (β=0.797, p=0.015), HIV RNA in plasma (β=-49.3, p=0.0001), and GDS (β=42.3, p=0.017)
with weaker associations with nadir CD4+ T-cell count (β=-0.35, p=0.053) and duration of ART
(β=1.03, p=0.101). Including all of these covariates in a single model identified that higher ART drug
concentrations were most strongly associated with lower HIV RNA in plasma (β=-75.6, p=0.031) and
worse GDS (β=51.6, p=0.016). ART drug concentrations in brain tissue were not associated with age,
sex, race/ethnicity, or current CD4+ T-cell count. The relationship between ART drug concentrations in
brain tissue and HIV RNA in CSF followed a quadratic pattern (R2=0.137, p=0.0036, Figure 3).
Introduction
Antiretroviral therapy (ART) can suppress HIV replication below the limit of detection for most
persons with HIV. Nevertheless, ART does not eradicate the viral reservoir that persists in lymphocytes
and other cells, including those in protected anatomic compartments such as the central nervous system
(CNS). ART drug concentrations in cerebrospinal fluid (CSF) provide an estimate of those in brain
tissue and are often substantially lower than ART drug concentrations in blood. Multiple factors can
influence distribution of ART drugs into the CNS, including the multicellular structure of the bloodbrain
barrier (BBB) and blood-CSF barrier (BCB), physical and chemical characteristics of the drugs
themselves (e.g., fat solubility and protein-binding), and concomitant clinical characteristics (e.g.,
age).[1] The distribution of many ART drugs into the CNS is not mediated by passive processes that
depend on drug physicochemical characteristics alone but can involve drug transporters (e.g., organic
cation or anion transporters).[2, 3] Transport occurs in both directions (influx and efflux) and can be
affected by concomitant drugs, for example by inhibition or induction of the transporters.[4]
Age-related comorbidities may also affect BBB permeability and drug distribution. These conditions
include cerebrovascular disease,[5], diabetes, and neurodegenerative disorders.[6] Conditions that increase
the concentration of drug-binding proteins, such as albumin, in the CNS could also reduce the
concentration of unbound, active drug. Since the concentrations of drug-binding proteins in the CNS are
typically very low, even a small change could substantially alter the already low concentrations of active
ART drugs in this protected compartment. This could be important since lower concentrations of ART
drugs in CSF appear to correlate with higher HIV RNA levels in CSF.[7, 8]
The clinical value of estimating ART drug concentrations in the CNS continues to be debated, in
part because estimation methods have historically been based on drug concentrations in CSF, which may
not accurately reflect drug concentrations in the brain. For example, efavirenz (EFV) concentrations are
much lower in CSF than in blood[9] but murine data and physiology-based pharmacokinetic modeling
supports that EFV concentrations are much higher in brain tissue than in blood.[10] This report was
recently confirmed in non-human primates and extended to include other ART drugs such as tenofovir
(TFV), atazanavir (ATV), and maraviroc.[11]
This manuscript reports ART drug concentrations measured in postmortem human brain tissue.
While some of the ART drugs used by participants in this project are no longer used in the clinic, the
overall finding that ART drug concentrations in brain tissue exceed those in CSF is important. In
addition, some of the measured drugs still have clinical relevance. For example, TFV, emtricitabine
(FTC), and lamivudine (3TC) continue to be recommended for use in all clinical environments (i.e., in
low-, middle-, and high-income countries). TFV and FTC are also used as pre-exposure prophylaxis to
prevent HIV infection. As low- and middle-income countries continue to transition to the use of
integrase strand transfer inhibitors like dolutegravir, use of older drugs like EFV and ATV continue to
be used to treat persons with HIV. Thus, the findings presented here have clinical relevance even though
some of the drugs measured in this pilot project are no longer routinely used.
ART drug concentrations in brain tissue are difficult to assess in living humans since brain biopsy is
only rarely performed for conditions such as primary CNS lymphoma, which likely alter the BBB. At
present, collection of postmortem tissue at autopsy is the only method of obtaining the tissue needed to
measure ART drug concentrations in the brain. This method presents challenges such as accurately
predicting time-of-death and ensuring that ART is dosed until death. Even with these challenges,
measuring ART drug concentrations in human postmortem brain tissue would provide valuable data. To
address this, we meaured tissue concentrations of multiple ART drugs in three brain regions from adults
who participated in the California NeuroAIDS Tissue Network (https://cntn.hivresearch.ucsd.edu) and
died with HIV infection.
|
|
| |
| |
|
|
|