 |
 |
 |
| |
ViiV Healthcare to present new data on long-acting regimens for HIV prevention and treatment, alongside extensive insights into the evolving needs of people living with HIV at 23rd International AIDS Conference (AIDS 2020: Virtual) - PRESS RELEASE
|
| |
| |
Continuing to challenge the current HIV treatment paradigm, data presented will span our diverse portfolio, investigating new and innovative treatment options for people living with HIV.
London, 2 July 2020 - ViiV Healthcare, the global specialist HIV company majority owned by GSK, with Pfizer Inc. and Shionogi Limited as shareholders, and its clinical trial partners, will be presenting over 20 abstracts during next week at the 23rd International AIDS Conference (AIDS 2020: Virtual), being held 6-10 July. Because of the COVID-19 pandemic, the conference will, for the first time, be completely virtual.
Highlights from ViiV Healthcare and study partners at AIDS 2020 will include the final efficacy analysis from the global HIV Prevention Treatment Network (HPTN ) 083 study evaluating investigational long-acting cabotegravir for HIV prevention; 48-week data from CUSTOMIZE, an implementation research study on how to implement, if approved, a new, once-monthly injectable HIV treatment in US healthcare practices; and results from the Positive Perspectives Wave 2 study, providing extensive insights from more than 2,300 people living with HIV (PLHIV) in 25 countries.
ViiV Healthcare's commitment towards ending paediatric AIDS by 2025 will be showcased with a media panel to introduce the new Positive Action Breakthrough grants alongside our global partners selected to receive ViiV Healthcare funding, and a satellite symposium on delivering an Integrated Approach for Adolescent Mothers and their Children.
Kimberly Smith, Head of Research & Development, ViiV Healthcare, said: "The data we're presenting at AIDS 2020 show the continued depth and focus of ViiV Healthcare's patient-centred approach to innovation and the strength of our commitment to the community. We'll be sharing the significant advances we've made not just for the treatment of HIV, but for prevention as well, in some of the populations most disproportionately impacted by HIV. We'll also share the in-depth insights gained from more than 2,300 people living with HIV that will help address their treatment needs and challenges."
Key abstracts to be presented by ViiV Healthcare and study partners at AIDS 2020
Final efficacy analysis from the global HPTN 083 study evaluating cabotegravir for HIV prevention: The HPTN 083 study, with approximately 4,600 participants across 40 sites in North and South America, Asia and Africa, is one of the first-ever clinical trials to directly compare two active HIV prevention agents. In a planned interim review, the independent Data and Safety Monitoring Board recommended the blinded, randomised portion of the study be stopped early and results released. Detailed final efficacy results from this interim review will be presented at AIDS 2020.
48-week findings from the implementation research study, CUSTOMIZE (Cabotegravir plus Rilpivirine in the US To Optimize and Measure Implementation and Experience): Data to be presented at AIDS 2020 will outline the quantitative findings from ViiV Healthcare's implementation research study, CUSTOMIZE. ViiV Healthcare worked with 24 healthcare providers and clinic administrators from eight sites to identify and evaluate approaches to implementing the investigational once-monthly, long-acting, investigational regimen of cabotegravir and rilpivirine for the treatment of HIV, into clinical practice. Long-acting treatments have the potential to change HIV management by replacing daily pills with long-acting dosing regimens, and results from the CUSTOMIZE trial will help to find solutions to real-world challenges in implementing such treatments.
Global Positive Perspectives Wave 2 study results: ViiV Healthcare will showcase results from the second wave of Positive Perspectives research at AIDS 2020. The study captured insights from more than 2,300 people living with HIV from 25 countries and the results highlight the importance of a holistic approach involving people living with HIV in open dialogue and joint decision-making with their healthcare providers. The external Positive Perspectives advisory committee also developed a manifesto, in partnership with ViiV Healthcare, containing specific recommendations for policymakers to include quality of life in assessments and encourage open and active dialogue, which will be outlined at the conference.
Advancing HIV treatment and care in paediatrics: Leading the way with the delivery of innovative science-led, access-driven solutions to ensure children living with HIV are not left behind, ViiV Healthcare has made a commitment towards ending paediatric AIDS by 2025. Among the events at AIDS 2020, ViiV Healthcare will host two sessions focusing on paediatric treatment and care:
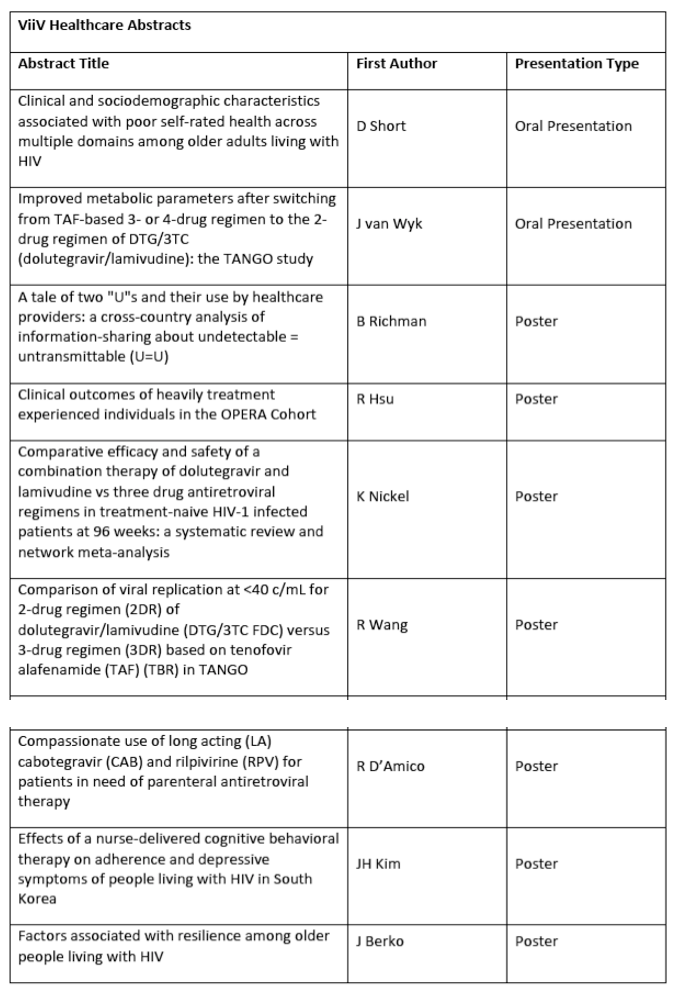
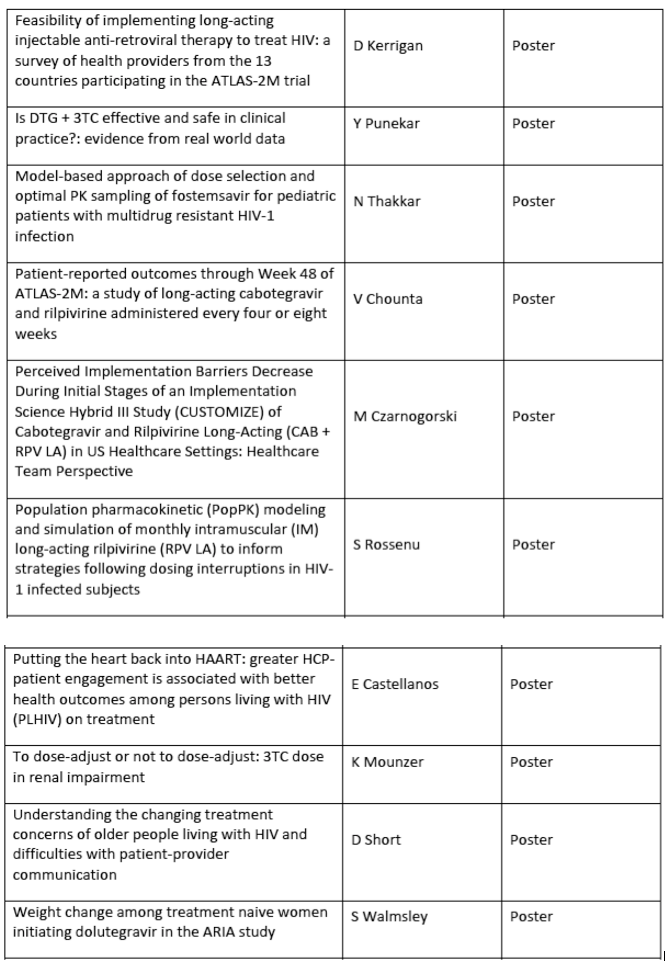
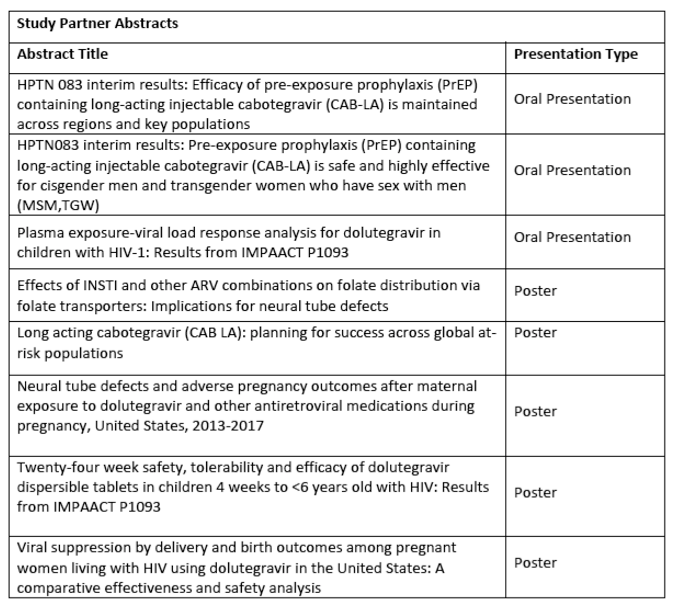
The full list of ViiV Healthcare, sponsored and relevant partners data to be presented at AIDS 2020 is outlined below:
Important Safety Information for CABENUVA
Indications and clinical use:
CABENUVA (cabotegravir and rilpivirine extended release injectable suspensions) is indicated as a complete regimen for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults to replace the current antiretroviral regimen in patients who are virologically stable and suppressed (HIV-1 RNA <50 copies/mL).
VOCABRIA (cabotegravir tablets) is indicated, in combination with EDURANT (rilpivirine tablets), as a complete regimen for short-term treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults who are virologically stable and suppressed (HIV-1 RNA <50 copies/mL) as: An oral lead-in to assess tolerability of cabotegravir prior to initiating CABENUVA
Oral bridging therapy for missed CABENUVA injections
• Geriatrics (>65 years of age): Not sufficiently studied to determine if they respond differently than patients <65 years of age
• Pediatrics (<18 years of age): Safety and efficacy not established
Contraindications:
In combination with:
• Anticonvulsants: Carbamazepine, oxcarbazepine, phenobarbital, and phenytoin
• Antimycobacterials: Rifabutin, rifampin, rifapentine
• Glucocorticoid: Systemic dexamethasone (more than a single dose)
• St John's wort (Hypericum perforatum)
Relevant warnings and precautions:
• Should not be used in patients with known or suspected resistance to cabotegravir or rilpivirine
• Patients may still develop opportunistic infections and other complications of HIV infection
• Risk of transmission: precautions should be taken
• Depressive disorders
• Hepatotoxicity (serum transaminase elevations)
• Hepatic adverse events; increased risk for worsening or development of transaminase elevations in patients with hepatitis B or C co-infection or marked elevations in transaminases prior to treatment; monitoring of liver chemistries is recommended
• Loss of virologic response due to drug interactions; review concomitant medications during therapy
• Caution when used in combination with drugs that have a risk of Torsade de Pointes
• Skin and hypersensitivity reactions; discontinue immediately if signs or symptoms develop
• Administer the oral lead-in dosing prior to administration of CABENUVA to help identify patients who may be at risk of a hypersensitivity reaction
• Residual concentrations of cabotegravir and rilpivirine injections may remain in the systemic circulation of patients for up to 12 months or longer
• Risk of resistance due to treatment discontinuation
• Post-injection reactions within minutes after the injection of rilpivirine,
including dyspnea, agitation, abdominal cramping, flushing, sweating, oral numbness, and changes in blood pressure. Reported in <0.5% of subjects and began to resolve minutes after the injection, and may have been associated with inadvertent (partial) IV administration
• Insufficient data in pregnant women; should not be used unless the potential benefits outweigh the potential risks
• HIV-1-infected mothers should not breastfeed their infants if receiving CABENUVA
For more information:
Please consult the Product Monograph at cabenuvapm.viivhealthcare.ca for additional important information relating to adverse reactions, drug interactions, and dosing. The Product Monograph is also available by calling 1-877-393-8448. To report an adverse event, please call 1-877-393-8448.
About ViiV Healthcare
ViiV Healthcare is a global specialist HIV company established in November 2009 by GlaxoSmithKline (LSE: GSK) and Pfizer (NYSE: PFE) dedicated to delivering advances in treatment and care for people living with HIV and for people who are at risk of becoming infected with HIV. Shionogi joined in October 2012. The company's aim is to take a deeper and broader interest in HIV/AIDS than any company has done before and take a new approach to deliver effective and innovative medicines for HIV treatment and prevention, as well as support communities affected by HIV.
For more information on the company, its management, portfolio, pipeline and commitment, please visit www.viivhealthcare.com.
About GSK
GSK is a science-led global healthcare company with a special purpose: to help people do more, feel better, live longer. For further information please visit www.gsk.com/about-us.
Cautionary statement regarding forward-looking statements
GSK cautions investors that any forward-looking statements or projections made by GSK, including those made in this announcement, are subject to risks and uncertainties that may cause actual results to differ materially from those projected. Such factors include, but are not limited to, those described under Item 3.D "Risk Factors" in the company's Annual Report on Form 20-F for 2019 and any impacts of the COVID-19 pandemic.
|
| |
|
 |
 |
|
|