 |
 |
 |
| |
2 HIV Vaccine Studies/Functional Cure & mRNA Vaccine for HIV
Functional HIV Cure in Monkey with 3 Monoconal antibodies
|
| |
| |
New HIV Treatment Strategy Can Provide Long-term Viral Suppression, according to Miller School Study-2019....https://physician-news.umiamihealth.org/new-hiv-treatment-strategy-can-provide-long-term-viral-suppression-according-to-miller-school-study/
The Campbell Foundation Awards Grant to UM Researchers Seeking to Develop Functional Cure for HIV-nov. 2020.....https://www.campbellfoundation.net/um-grant-2020
Adeno-Associated Virus Delivery of Anti-HIV Monoclonal Antibodies Can Drive Long-Term Virologic Suppression.....Chronically SHIV-infected macaques were treated with AAV-delivered bnAbs, A functional cure was achieved in one SHIV-infected macaque - Long-term delivery of anti-HIV monoclonal antibodies (mAbs) using adeno-associated virus (AAV) vectors holds promise for the prevention and treatment of HIV infection. We describe a therapy trial in which four rhesus monkeys were infected with SHIV-AD8 for 86 weeks before receiving the AAV-encoded mAbs 3BNC117, 10-1074, and 10E8. Although anti-drug antibody (ADA) responses restricted mAb delivery, one monkey successfully maintained 50-150 μg/mL of 3BNC117 and 10-1074 for over 2 years. Delivery of these two mAbs to this monkey resulted in an abrupt decline in plasma viremia, which remained undetectable for 38 successive measurements over 3 years. We generated two more examples of virologic suppression using AAV delivery of a cocktail of four mAbs in a 12-monkey study. Our results provide proof of concept for AAV-delivered mAbs to produce a "functional cure." However, they also serve as a warning that ADAs may be a problem for practical application of this approach in humans. At 86 weeks post infection, each monkey received intramuscular injections of AAV-1 vectors encoding IgG1 (immunoglobulin G1) versions of the potent broadly neutralizing mAbs 10E8 (Huang et al., 2012), 3BNC117 (Scheid et al., 2011), and 10-1074 (Mouquet et al., 2012) (Figure S1)......https://www.cell.com/immunity/fulltext/S1074-7613(19)30071-8
----------------------------------
mRNA HIV Vaccine
Induction of cross-neutralizing antibodies and protection from heterologous tier-2 SHIV challenge by an mRNA-based vaccine in macaques at IAC 2020 virtual
https://programme.aids2020.org/Search/Search?search=Paolo+Lusso
Presenter
Paolo Lusso
Authors
P. Zhang (1), M. Prabhakaran (2), S. Ding (3), Y. Tsybovsky (4), E. Narayanan (5), A. Carfi (5), S. Himansu (5), Y. Lin (1), D. Rogers (1), Q. Liu (1), H. Miao (1), X. Chen (2), E.K. Sarfo (2), D.R. Ambrozak (2), R. Gautam (6), M.A. Martin (6), D. Weiss (7), J. Misamore (7), J.R. Mascola (2), A. Finzi (3), A. McDermott (2), P. Lusso * (1)
Institutions
(1) NIH, LIR, NIAID, Bethesda, United States, (2) NIH, VRC, Bethesda, United States, (3) University of Montreal, Montreal, Canada, (4) NIH, ATRF, NCI, Frederick, United States, (5) Moderna Inc, Cambridge, United States, (6) NIH, LMM, NIAID, Bethesda, United States, (7) Bioqual Inc., Rockville, United States
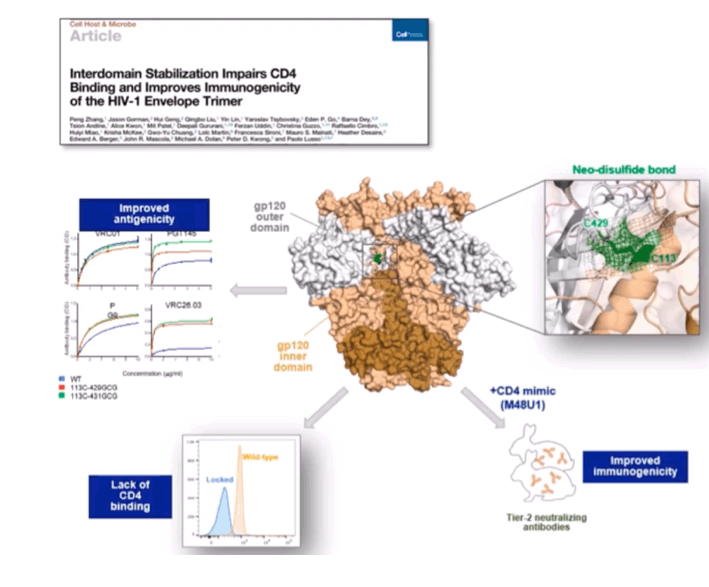
BACKGROUND: Despite intensive research over the past four decades, an HIV-1 vaccine capable of inducing broadly neutralizing antibodies (bNAbs) and protection from heterologous tier-2 strains is still wanting.
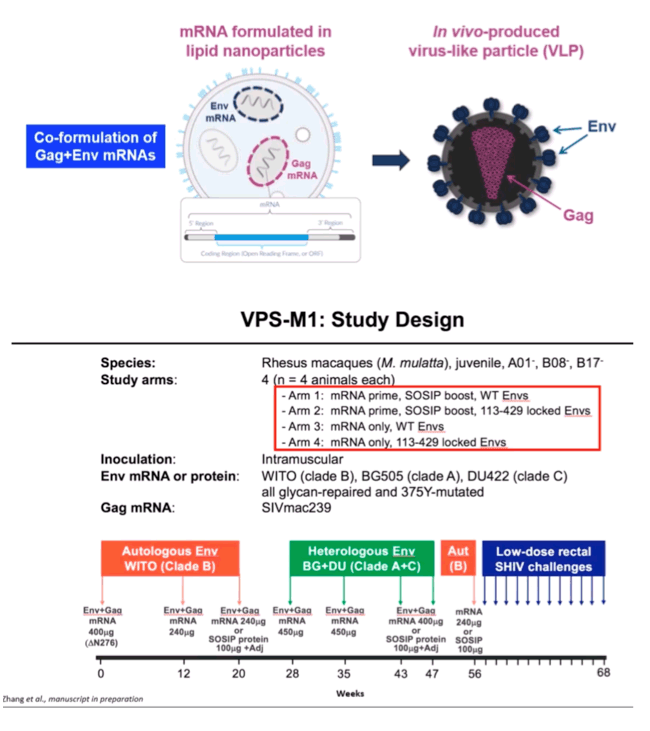
METHODS: We tested the immunogenicity and efficacy of an mRNA-based vaccine that expresses native, membrane-anchored HIV-1 envelope (Env) glycoproteins in combination with SIVmac239 Gag in order to promote the in vivo formation of virus like particles (VLP)'. Rhesus macaques were immunized with 8 sequential mRNA immunizations or mRNA followed by protein boosts with purified homologous SOSIP trimers over a period of one year. The Envs included WITO (a clade-B transmitter-founder strain) BG505 (clade A) and DU422 (clade C).
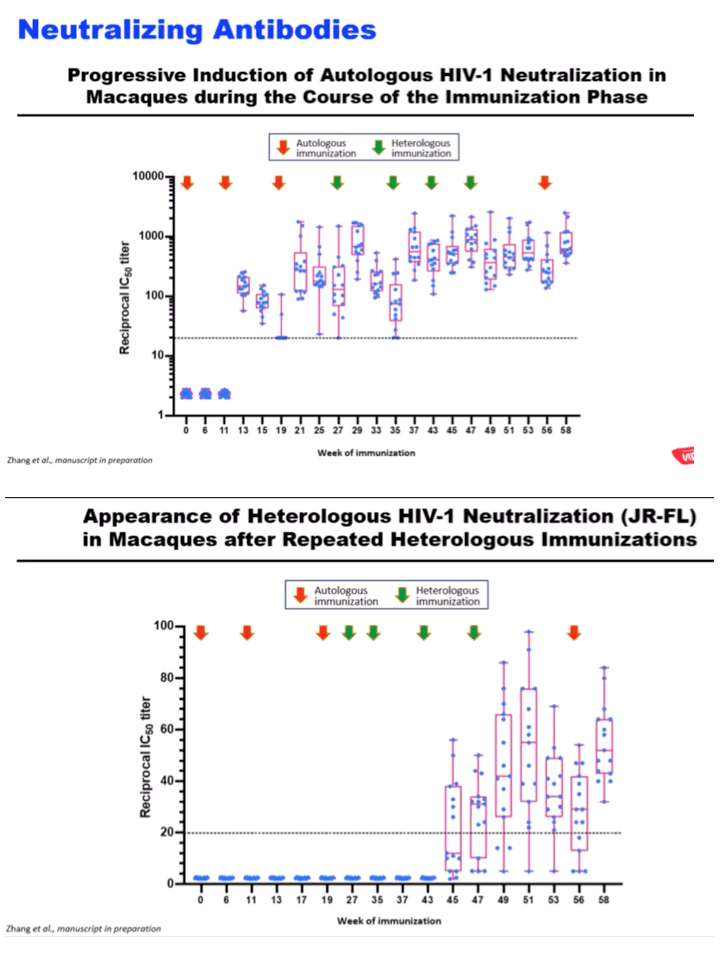
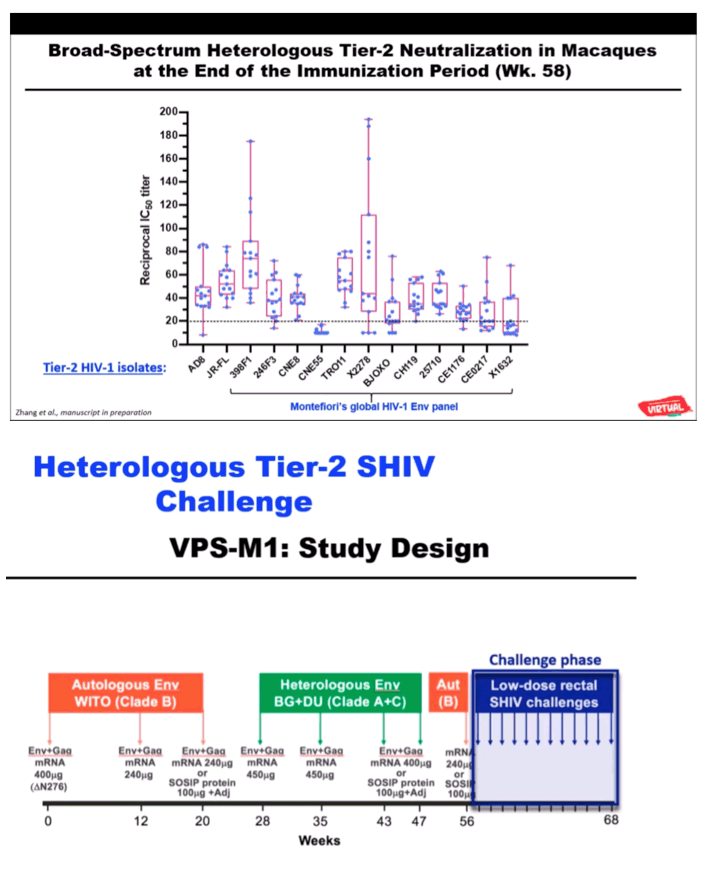
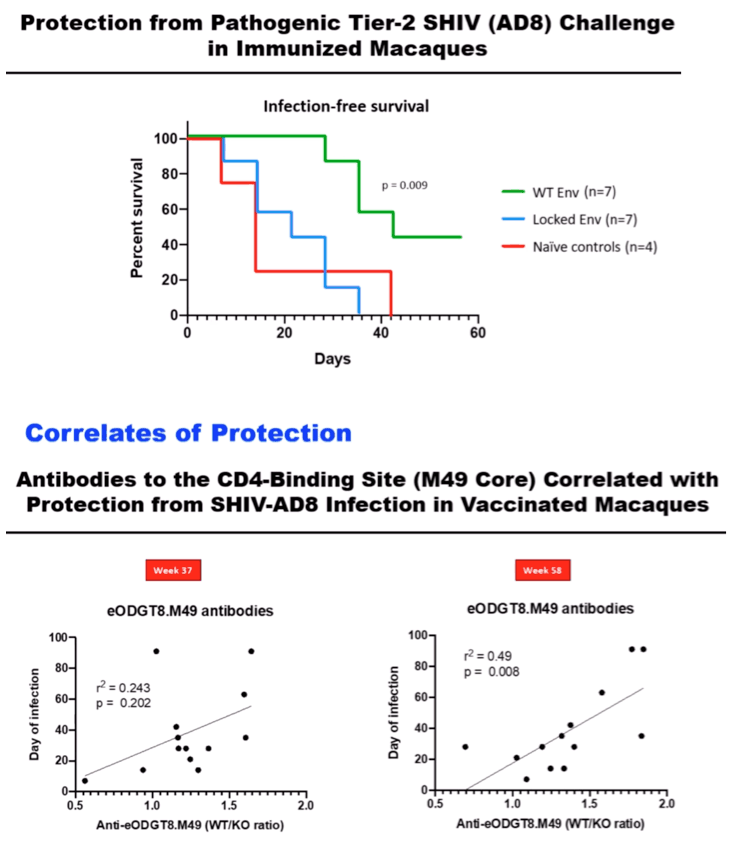
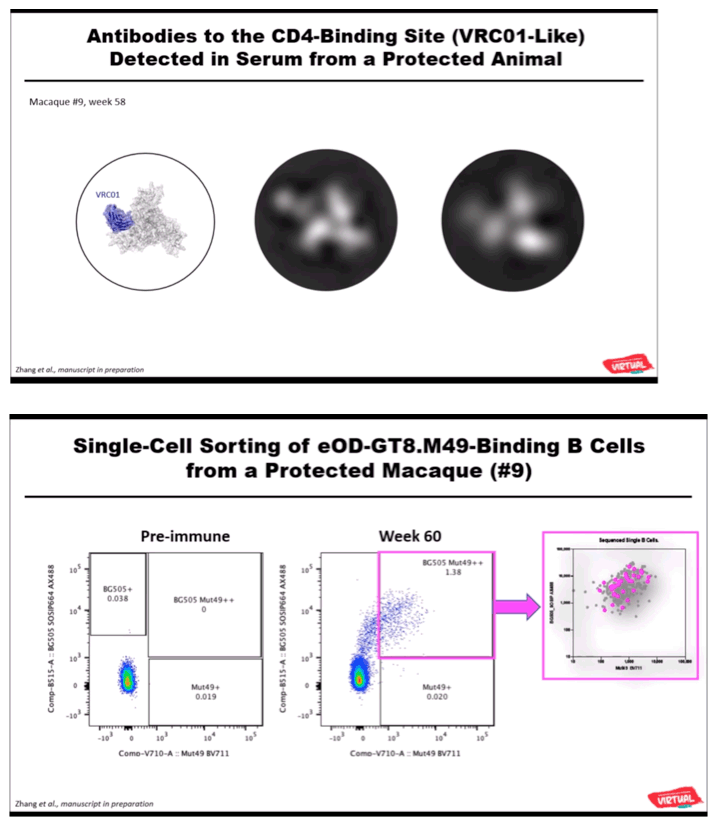
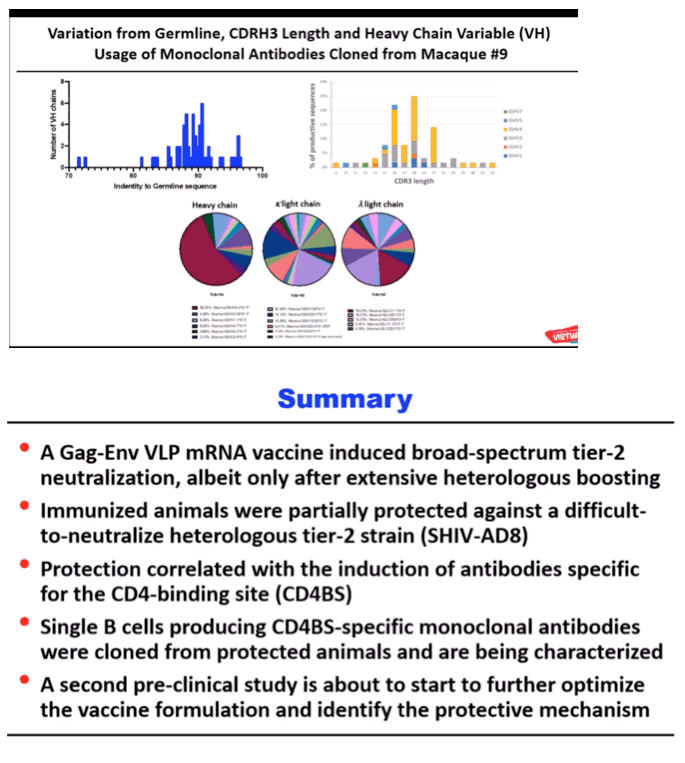
RESULTS: Env trimer-binding antibodies (Abs) were readily induced after the second immunization, showing increasing titers and durability after each booster injection. Following the third heterologous boost, cross-neutralizing Abs against tier-2 viruses of different clades started to appear in all animals, reaching higher and more stable titers after the last immunization. After challenge with heterologous tier-2 SHIV (AD8) by 12 repeated low-dose rectal inoculations, significant protection was observed in a group of animals, with no difference between those immunized with mRNA vs. mRNA+protein. Extensive immunologic analyses identified three significant correlates of protection: i) Abs to a glycanated CD4-binding site (CD4-BS) gp120 core protein; ii) Abs to the AD8 Env trimer expressed on the surface of infected cells; iii) Abs mediating ADCC against the closed AD8 Env trimer. Abs to both the CD4-BS and the trimer apex were visualized in the serum of protected animals by electron microscopy polyclonal mapping. By single B-cell cloning and antibody amplification from a protected macaque, we derived a panel of mAbs that share genetic similarities with previously identified macaque and human bNAbs against the CD4-BS. Detailed functional characterization of these mAbs will be presented at the Conference.
CONCLUSIONS: These results provide evidence that extensive immunization with mRNA encoding multiple heterologous membrane-anchored HIV-1 Envs can induce cross-clade neutralization and partial protection from heterologous tier-2 virus challenge.
Funding: Supported in part by the intramural research programs of the NIAID DIR and VRC, and by the NIH Office of AIDS Research (OAR).



|
| |
|
 |
 |
|
|