 |
 |
 |
| |
Early Weight Gain Up to 4.5 Kg/Year With Switch From TDF to TAF:
Data Suggest Independent Effect of TAF on Weight
|
| |
| |
AIDS 2020: 23rd International AIDS Conference Virtual, July 6-10, 2020
Mark Mascolini
Virologically suppressed US adults with HIV gained 1.8 to 4.47 kg in weight in the first 9 months after switching from tenofovir disoproxil fumarate (TDF) to tenofovir alafenamide (TAF), according to analysis of a large US cohort [1]. At that point weight gain slowed, plateaued, or even reversed. The consistency of the immediate postswitch finding from regimen to regimen suggested to the researchers that TAF has an independent effect favoring weight gain.
Starting or switching to an integrase inhibitor, particularly dolutegravir, has led to weight gain in several trials and cohort studies. Research links TAF to more weight gain than TDF. In the ADVANCE trial involving 1053 antiretroviral-naive people, starting treatment with dolutegravir plus TAF/FTC led to more weight gain through 96 weeks than starting dolutegravir plus TDF/FTC or efavirenz plus TDF/FTC, especially in women [2].
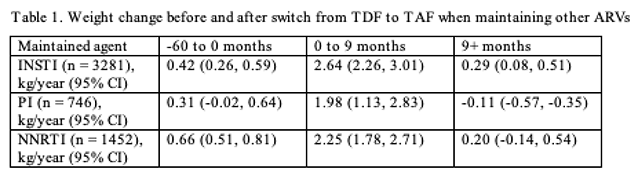
Researchers working with the longitudinal OPERA cohort analyzed weight change in people switching from TDF to TAF and maintaining other elements of their regimen or switching from a nonnucleoside (NNRTI) or a protease inhibitor (PI) to an integrase inhibitor. OPERA includes more than 115,000 US residents receiving care for HIV infection in 65 cities across the country. For this analysis researchers focused on antiretroviral-experienced adults on a three-drug regimen including TDF who had two or more consecutive viral loads below 200 copies when they substituted TAF for TDF. They used linear mixed models to explore (1) weight changes with the switch from TDF to TAF and (2) rates of weight change over time. Those models adjusted for age, sex, race, body mass index, CD4 count, endocrine disorders, and concurrent therapy that could affect weight.
The analysis included 1454 people who maintained an NNRTI, 747 who maintained a boosted PI, 3288 who maintained an integrase inhibitor, and 1430 who switched from an NNRTI or a PI to an integrase inhibitor. Median ages across those four groups were 45, 51, 44, and 49. Proportions of women were 19%, 21%, 15%, and 18%, blacks 41%, 39%, 37%, and 38%, and Hispanics 24%, 25%, 26%, and 26%. Median body mass index was 26 or 27 kg/m2 in all four groups, that is, low in the overweight range. About one quarter of patients had an endocrine disorder or took non-HIV medications that could alter weight.
The people who maintained their other antiretrovirals had a slight weight gain in the 60 months before the TDF-to-TAF switch (Table 1). That gain rose steeply in the first 9 months after the switch (1.98 to 2.64 kg/year), then appeared to level off after 9 months in all three treatment groups (Table 1).
Among people who maintained an integrase inhibitor when switching from TDF to TAF, those maintaining elvitegravir/cobicistat or dolutegravir gained a little more weight in the 9 months after the switch than those maintaining raltegravir (Table 2). Weight change after those initial postswitch 9 months differed depending on which integrase inhibitor a person maintained (Table 2).

Among cohort members who switched from an NNRTI or a PI to an integrase inhibitor when trading TDF for TAF, the weight gain in the 9 months after the switch was greatest in those who also switched to bictegravir, followed by dolutegravir, then elvitegravir/cobicistat (Table 3). After the 9-month postswitch period, people also switching to bictegravir lost almost twice the weight they gained in those 9 months (Table 3), but the confidence interval with this estimate is wide because follow-up on bictegravir was shorter than in the other two integrase inhibitor groups. Among people switching to elvitegravir/cobicistat or dolutegravir, weight stabilized after the 9-month postswitch period (Table 3).
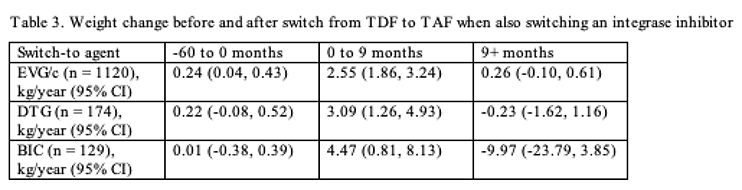
The researchers concluded that in this large and diverse cohort, people who switched from TDF to TAF had "pronounced weight increases in all groups over the first approximate 9 months." After that 9-month period, weight gains slowed, plateaued, and sometimes even reversed, depending on the maintained antiretroviral or new integrase inhibitor.
The bottom line, the investigators proposed, is that, because weight gains in the months after the switch to TAF were consistent across regimens, TAF has an independent effect favoring weight gain regardless of whether other antiretrovirals in the regimen change or stay the same.
References
1. Mallon P, Brunet L, Hsu R, et al. Weight gain before and after switch from TDF to TAF. AIDS 2020: 23rd International AIDS Conference Virtual. July 6-10, 2020. Abstract OAB0604.
2. Venter WDF, Moorhouse M, Sokhela S, et al. Dolutegravir plus two different prodrugs of tenofovir to treat HIV. N Engl J Med. 2019;381:803-815. doi: 10.1056/NEJMoa1902824.
|
| |
|
 |
 |
|
|