 |
 |
 |
| |
Deep Sequencing With Unique Molecular Identifiers for Evaluation of HIV-1 Drug Resistance in the DISCOVER Pre-Exposure Prophylaxis Trial
|
| |
| |
AIDS 2020: 23rd International AIDS Conference Virtual, 6-10 July 2020
Reported by Jules Levin
Stephanie Cox,1 Urvi M. Parikh,2 Amy L. Heaps,2 Breanna J. Goetz,2 John W. Mellors,2 Moupali Das,1 Christian Callebaut1 - 1Gilead Sciences, Inc., Foster City, California, USA; 2University of Pittsburgh, Pennsylvania, USA
DISCOVER at AIDS2020
Sexually Transmitted Infections in the DISCOVER HIV PrEP Trial - (07/08/20)
DISCOVER Study for HIV Pre-exposure Prophylaxis: No Evidence of Risk Compensation in Participants Taking F/TDF or F/TAF for PrEP Through 96 Weeks - (07/09/20)

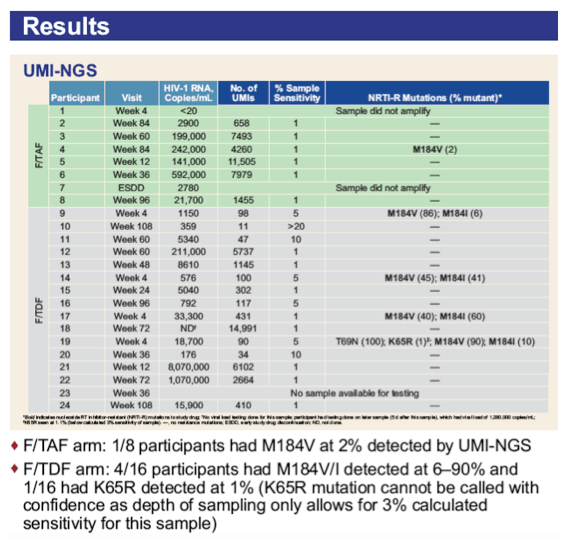
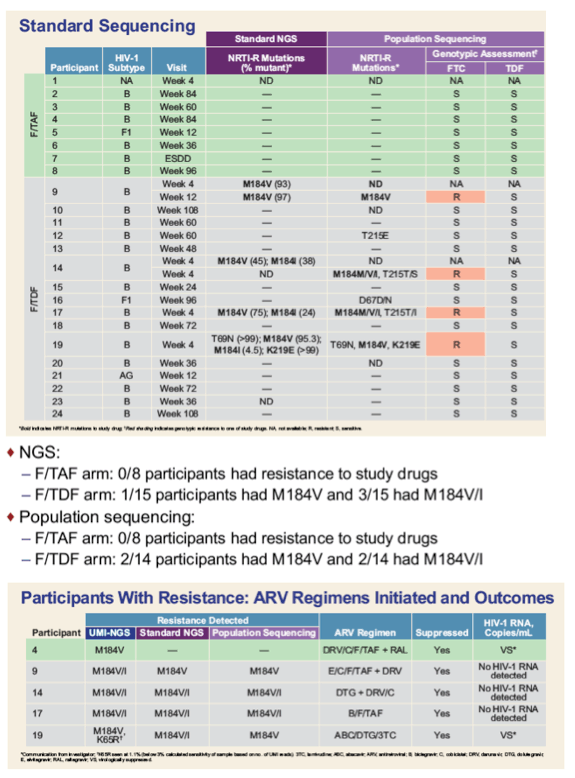
*Communication from investigator; †K65R seen at 1.1% (below 3% calculated sensitivity of sample based on no. of UMI reads). 3TC, lamivudine; ABC, abacavir; ARV, antiretroviral; B, bictegravir; C, cobicistat; DRV, darunavir; DTG, dolutegravir; E, elvitegravir; RAL, raltegravir; VS, virologically suppressed.
References: 1. Hare CB, et al. CROI 2019, oral 104; 2. Boltz VF, et al. Retrovirology 2016;13:87; 3. Clarke L, Carbon J. Cell 1976;9:91-9; 4. Hogg S. Essential Microbiology. Chichester, UK: Wiley, Blackwell; 2013; 5. Parikh U, et al. Curr Opin HIV AIDS 2016;11:49-55.
|
| |
|
 |
 |
|
|