 |
 |
 |
| |
HCV Drugs for COVID-19? Three Small Studies Have Positive Hints
|
| |
| |
IAS COVID-19 Virtual Conference, July 10-11, 2020
https://cattendee.abstractsonline.com/meeting/9307/presentation/3933
Mark Mascolini
COVID-19 patients treated with sofosbuvir/daclatasvir (SOF/DCV) in three small Iranian trials got out of the hospital faster and survived more often than people in comparison groups [1]. But these studies have limitations, so the value of SOF/DCV won’t be known until the 600-person double-blind, placebo-controlled, Iranian DISCOVER trial reaches a conclusion.
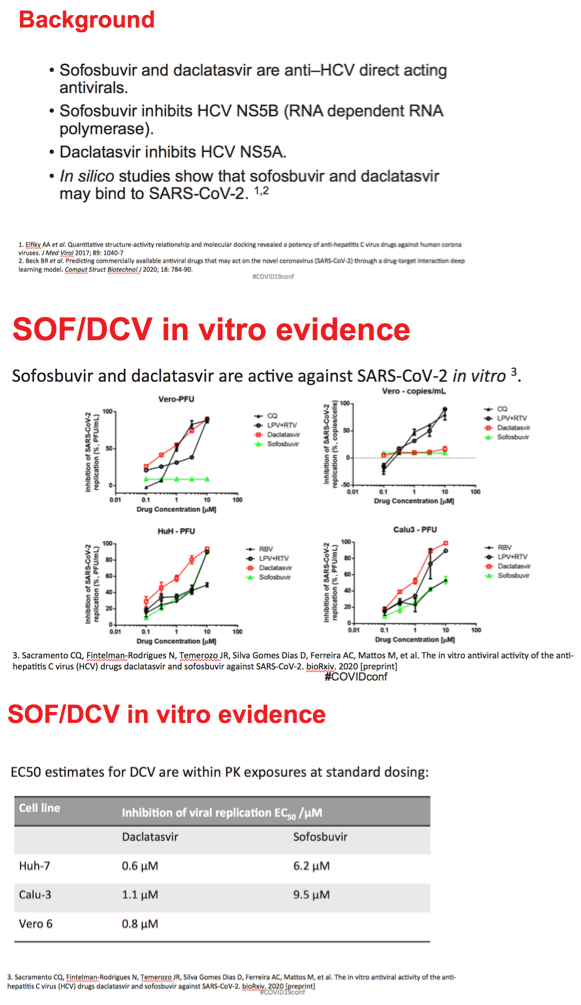
SOF and DCV inhibit HCV replication by different mechanisms. Computer-modeling (in silico) studies suggest these two direct-acting antivirals (DAAs) may bind to SARS-CoV-2, the virus that causes COVID-19 [2,3]. Unpublished in vitro evidence also suggests SOF and DCV have activity against the novel coronavirus [4]. At the IAS COVID-19 Virtual Conference, researchers from Tehran University, Imperial College London, and the University of Liverpool reviewed and pooled three small clinical trials of SOF/DCV for COVID-19 in Iran.
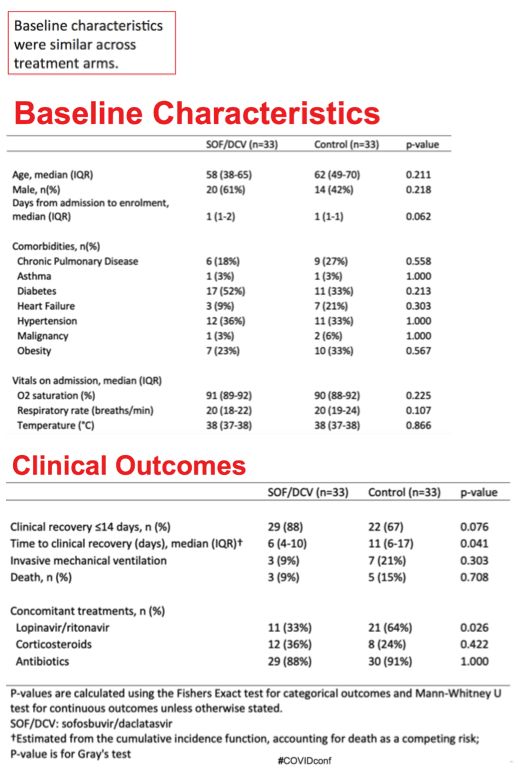
The multicenter, open-label DDRI trial (IRCT20200128046294N2, https://www.irct.ir/trial/46463) randomized 66 people with severe COVID-19 infection at four university hospitals to SOF/DCV plus lopinavir/ritonavir (LPV/r) or to LPV/r alone. Participants were at least 18 years old and admitted to the hospital with PCR-confirmed SARS-CoV-2 infection and signs of advanced COVID-19. Median age stood at 58 in the SOF/DCV arm and 62 in controls (difference not significant), 61% and 42% were men (difference not significant), and each group had a median of 1 day between admission and trial enrollment. The groups were highly similar in oxygen saturation, respiratory rate, and temperature, and similar proportions got treated with corticosteroids (about 30%) and/or antibiotics (about 90%).
A somewhat higher percentage in the SOF/DCV arm than in the control arm had clinical recovery (return to normal temperature, respiratory rate, and oxygen saturation) within 14 days (88% versus 67%, P = 0.076), and time to recovery was significantly shorter in the SOF/DCV group (median 6 versus 11 days, P = 0.041). Three people in the SOF/DCV arm and 5 in the control arm died (P = 0.708).
Meta-analysis pooled findings of this Tehran trial with a 62-person Abadan study (https://www.irct.ir/trial/46713) and a 48-person Sari study (https://www.irct.ir/trial/46885). The Abadan study randomized participants to SOF/DCV plus hydroxychloroquine (HCQ) or to HCQ plus LPV/r plus ribavirin. The Sari study randomized people to SOF/DCV plus ribavirin or to HCQ with or without LPV/r.
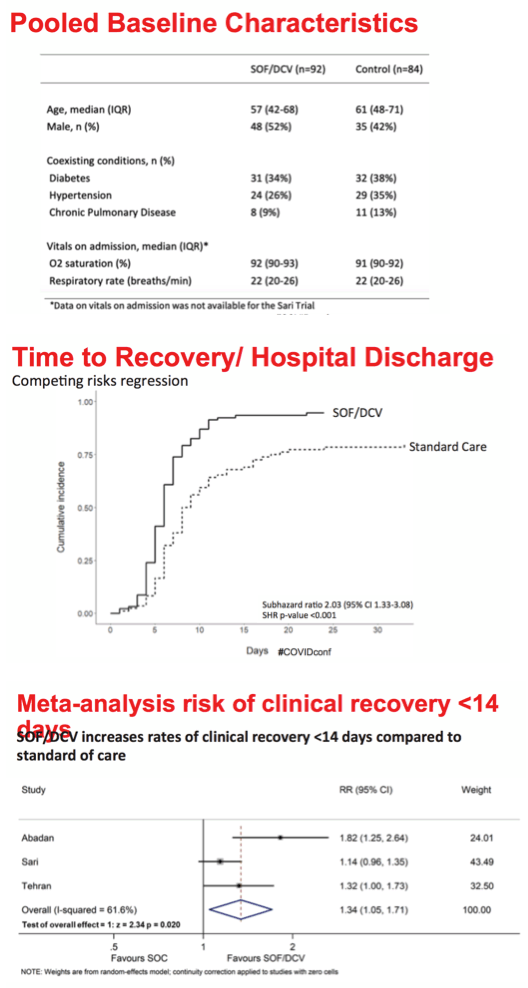
Pooled analysis involved 92 people randomized to SOF/DCV and 84 randomized to control arms. Respective median ages in the two group were 57 and 61, 52% and 42% were men, 9% and 13% had chronic pulmonary disease, and oxygen saturation and respiratory rate were similar in the two pooled groups.
Competing-risks regression analysis indicated twice faster time to recovery or hospital discharge in the combined SOF/DCV arms (subhazard ratio 2.03, 95% confidence interval [CI] 1.33 to 3.08, P < 0.001). Chance of clinical recovery within 14 days was one third better in the pooled SOF/DCV arms (RR 1.34, 95% CI 1.05 to 1.71).
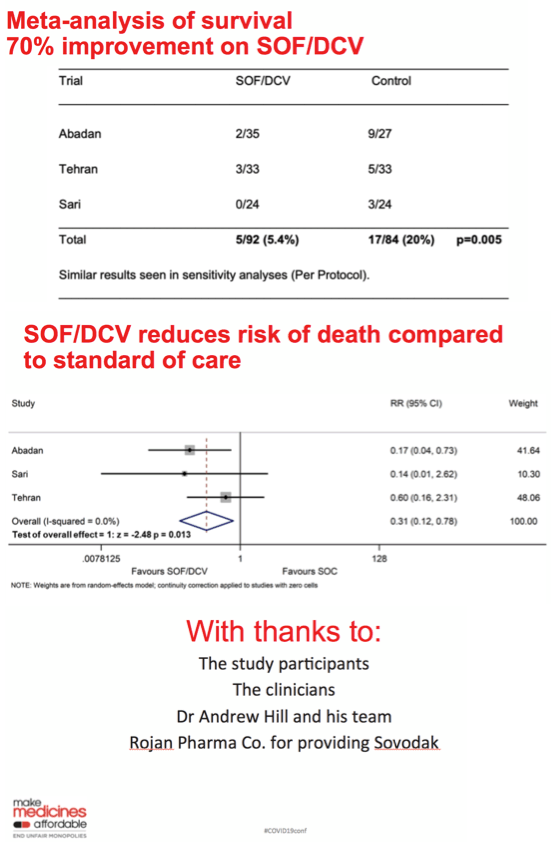
Five of 92 people randomized to SOF/DCV died, compared with 17 of 84 randomized to control arms (5.4% versus 20.2%, P = 0.005). Risk of death was 70% lower in the combined SOF/DCV arms (RR 0.31, 95% CI 0.12 to 0.78).
Researchers who conducted this analysis caution that the sample size is relatively small, one of the trials was not properly randomized, and viral load data were not examined because of limited PCR availability in Iran. They say results of the SOF/DCV DISCOVER trial are expected in September 2020. DISCOVER will randomize 600 adults admitted to the hospital with moderate or severe COVID-19 to SOF/DCV plus LPV/r or to LPV/r alone. The primary endpoint of this double-blind, placebo-controlled trial is clinical recovery within 10 days of starting treatment.
References
1. Sadeghi A, Asgari AA, Norouzi A, et al. Sofosbuvir and daclatasvir for the treatment of COVID-19: results from a randomised controlled trial. IAS COVID-19 Virtual Conference. July 10-11, 2020.
2. Elfiky AA, Mahdy SM, Elshemey WM. Quantitative structure-activity relationship and molecular docking revealed a potency of anti-hepatitis C virus drugs against human corona viruses. J Med Virol. 2017;89:1040-1047. doi: 10.1002/jmv.24736.
3. Beck BR, Shin B, Yoonjung Choi Y, Park S, Kang K. Predicting commercially available antiviral drugs that may act on the novel coronavirus (SARS-CoV-2) through a drug-target interaction deep learning model. Comput Struct Biotechnol J. 2020;18:784-790. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7118541/
4. Sacramento CQ, Fintelman-Rodrigues N, Temerozo JR, et al. The in vitro antiviral activity of the anti-hepatitis C virus (HCV) drugs daclatasvir and sofosbuvir against SARS-CoV-2. bioRxiv. 2020. Preprint.



|
| |
|
 |
 |
|
|