 |
 |
 |
| |
The ADVANCE trial: Phase 3, randomised comparison of TAF/FTC+DTG, TDF/FTC+DTG or TDF/FTC/EFV for first-line treatment of HIV-1 infection.
|
| |
| |
More Weight, Trunk Fat, Metabolic Syndrome With DTG+TAF/FTC in South African Trial
AIDS 2020: 23rd International AIDS Conference Virtual, July 6-10, 2020
Mark Mascolini
After 96 to 144 weeks in the South African ADVANCE trial, people randomized to dolutegravir (DTG) plus tenofovir alafenamide (TAF) and emtricitabine (FTC) gained more weight and trunk fat and ran a higher risk of metabolic syndrome than those randomized to DTG plus tenofovir disoproxil fumarate (TDF) and FTC or to efavirenz (EFV) plus TDF/FTC [1]. Rates of viral suppression below 50 copies did not differ significantly between the three arms at 96 weeks
Integrase inhibitors have emerged as preferred first-line therapy in high-income countries, and TAF has become a popular alternative to TDF. To help determine the place of TAF and the integrase inhibitor dolutegravir in South Africa and countries with similar epidemics, researchers are conducting the ADVANCE trial, an investigator-led, three-arm, open-label, randomized trial.
The ADVANCE team has published 48-week results [2]. This presentation at AIDS 2020 focused on findings from weeks 96 and 144.
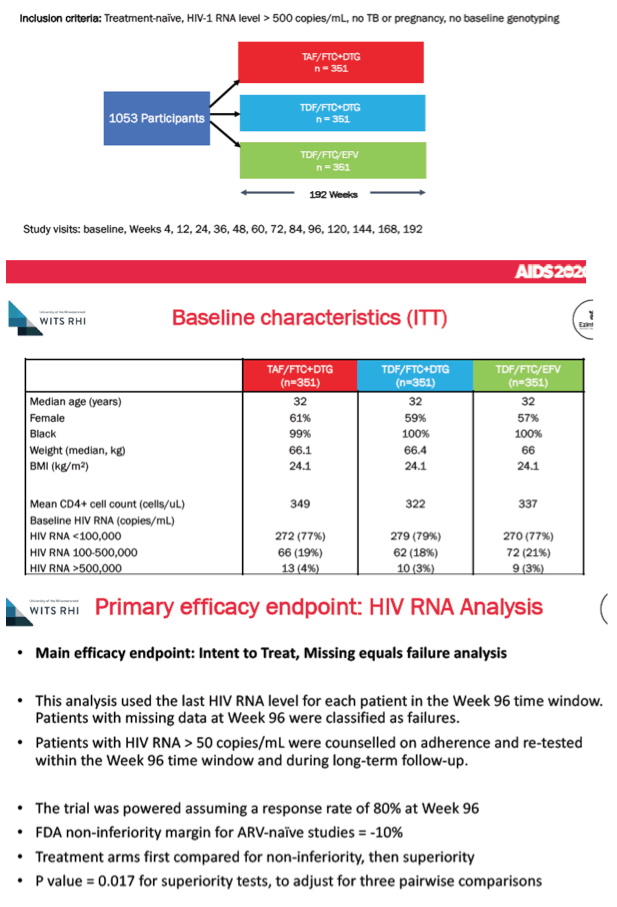
ADVANCE randomized 1053 antiretroviral-naive nonpregnant South Africans to DTG+TAF/FTC, DTG+TDF/FTC, or EFV/TDF/FTC. All three study arms had a median age of 32 years, about 59% were women, and 99% were black. Pretreatment CD4 count averaged about 335 across the three study arms, and each arm had a pretreatment body mass index of 24.1 kg/m2, putting the group just below the overweight threshold.
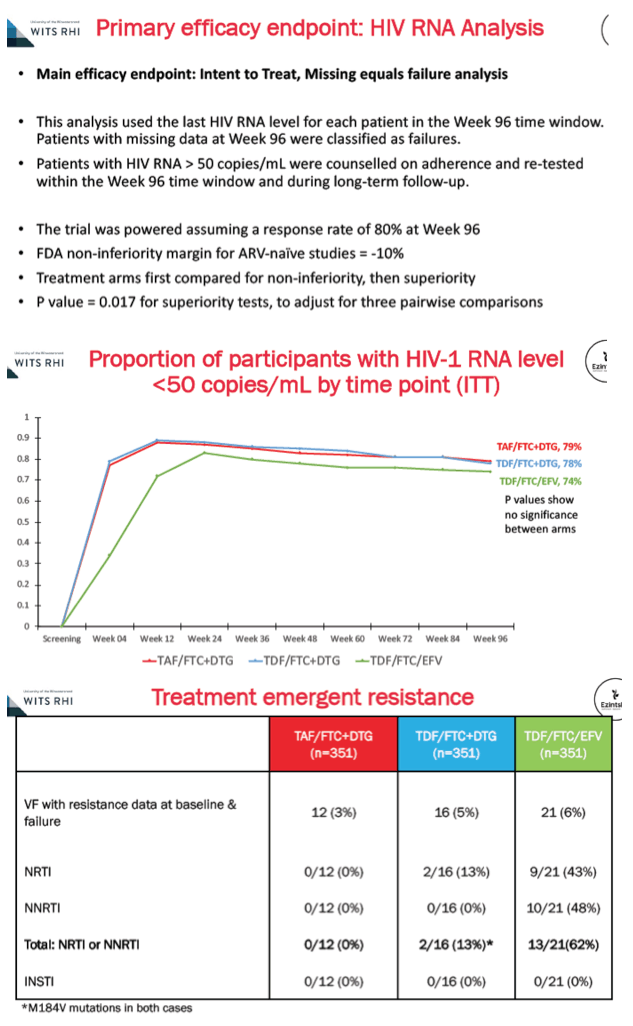
A 96-week intention-to-treat missing-data-equals failure analysis determined that similar proportions in the three arms had a viral load below 50 copies: 79% on DTG+TAF/FTC, 78% on DTG+TDF/FTC, and 74% on EFV/TDF/FTC. In people with resistance data, treatment-emergent resistance to nucleos(t)ides or nonnucleosides could be detected in 0 of 12 people taking DTG+TAF/FTC, 2 of 16 (13%) taking DTG+TDT/FTC (both with the M184V FTC mutation), and 13 of 21 (62%) taking EFV/TDF/FTC. Resistance to integrase inhibitors did not emerge in any study arm.
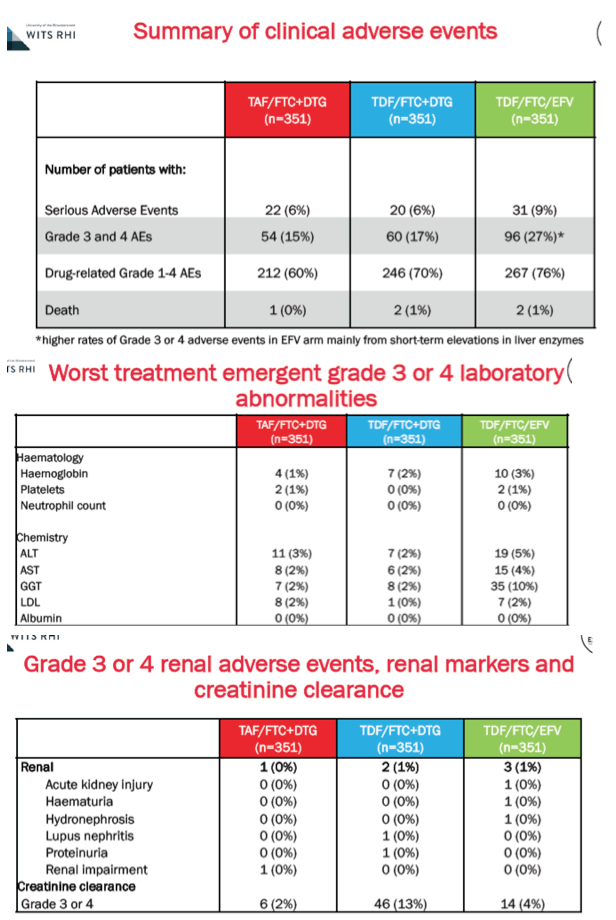
Somewhat lower proportions taking DTG+TAF/FTC or DTG+TDF/FTC than taking EFV/TDF/FTC had serious adverse events (6%, 6%, 9%), grade 3 or 4 adverse events (15%, 17%, 27%), or any drug-related adverse event (60%, 70%, 76%). Most grade 3 or 4 adverse events with EFV/TDF/FTC were short-term jumps in liver enzymes. Grade 3 or 4 kidney problems and kidney marker changes arose in 1% or fewer in each study arm. Proportions with grade 3 or 4 creatinine clearance were 2% with DTG+TAF/FTC, 13% with DTG+TDF/FTC, and 4% with EFV/TDF/FTC.
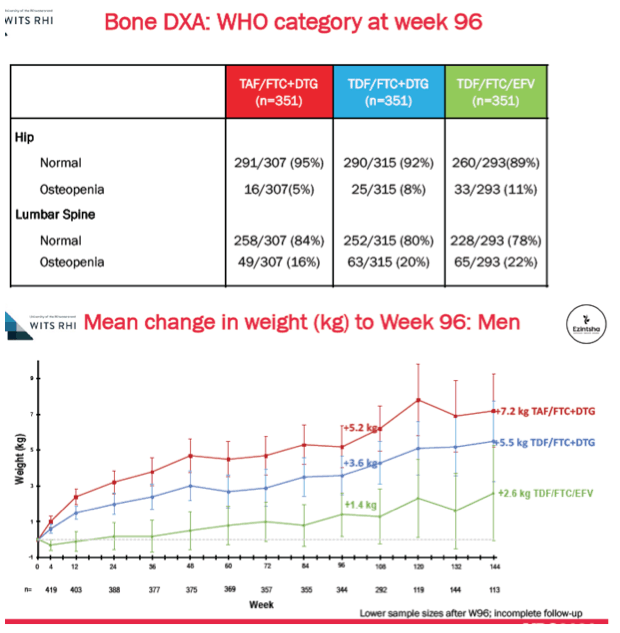
Through 96 weeks hip osteopenia developed in 5% on DTG+TAF/FTC, 8% on DTG+TDF/FTC, and 11% on EFV/TDF/FTC. Respective proportions with week-96 lumbar spine osteopenia were 16%, 20%, and 22%.
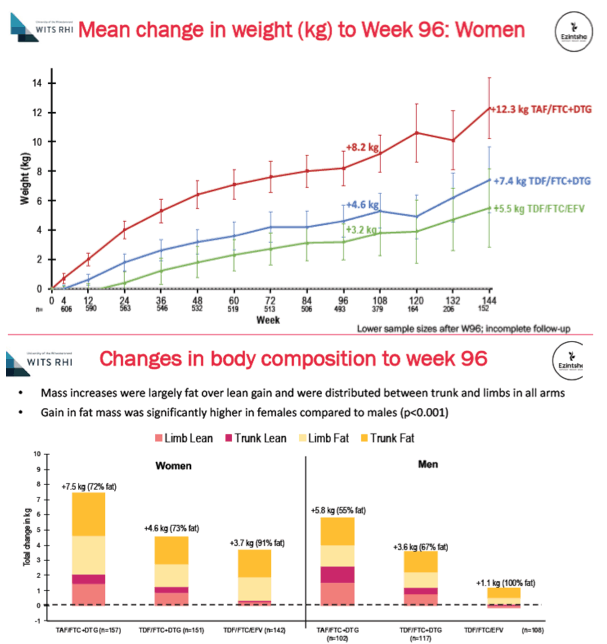
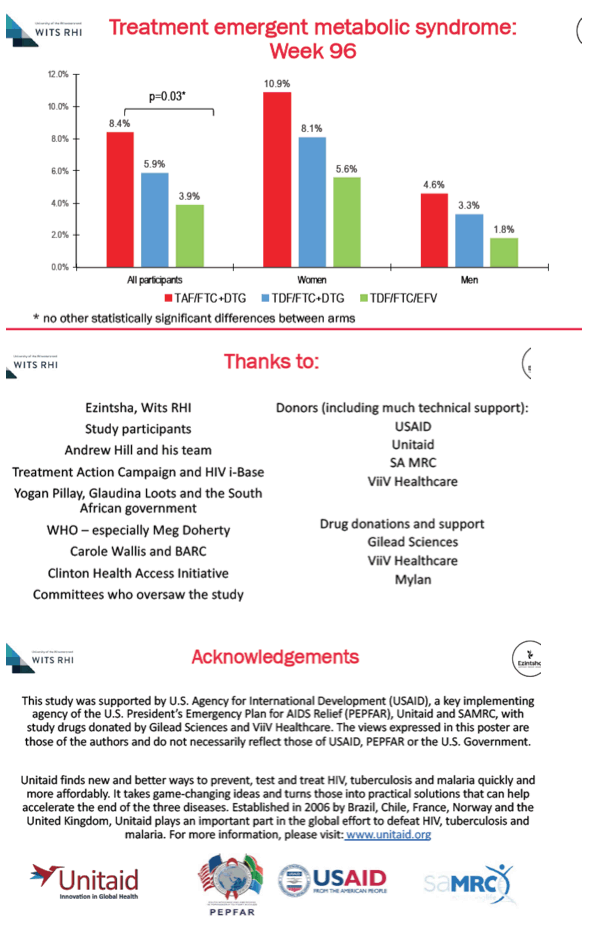
Through 144 weeks women gained an average 12.3 kg on DTG+TAF/FTC, 7.4 kg on DTG+TDF/FTC, and 5.5% on EFV/TDF/FTC. For men on those three regimens, week-144 gains averaged 7.2 kg, 5.5 kg, and 2.6 kg. Women gained significantly more fat mass than men through 96 weeks (P < 0.001), and women taking DTG+TAF/FTC gained more trunk fat than women taking the other two regimens. By week 96 treatment-emergent metabolic syndrome developed in significantly more people taking DTG+TAF/FTC (8.4%) than DTG+TDF/FTC (5.9%), or EFV/TDF/FTC (3.9%) (P = 0.03). Among women metabolic syndrome emerged in 10.9% assigned to DTG+TAF/FTC, 8.1% assigned to DTG+TDF/FTC, and 5.6% assigned to EFV/TDF/FTC.
The ADVANCE investigators noted that viral suppression through 96 weeks did not differ between the three study arms in intention-to-treat analysis, while people randomized to EFV/TDF/FTC had slightly more treatment-emergent resistance to nucleos(t)ides and nonnucleosides. People randomized to DTG+TAF/FTC did worse in weight gains, trunk fat gains, and new metabolic syndrome than those randomized to other arms. The researchers believe their findings support current World Health Organization guidelines reserving DTG+TAF/FTC for people with osteoporosis or impaired kidney function.
References
1. Venter F, Moorhouse M, Sokhela S, et al. The ADVANCE trial: Phase 3, randomised comparison of TAF/FTC+DTG, TDF/FTC+DTG or TDF/FTC/EFV for first-line treatment of HIV-1 infection. AIDS 2020: 23rd International AIDS Conference Virtual. July 6-10, 2020. Abstract OAXLB0104.
2. Venter WDF, Moorhouse M, Sokhela S, et al. Dolutegravir plus two different prodrugs of tenofovir to treat HIV. N Engl J Med. 2019;381:803-815. doi: 10.1056/NEJMoa1902824.

|
| |
|
 |
 |
|
|