 |
 |
 |
| |
PATIENT PERSPECTIVES ON IMPLEMENTATION OF A LONG-ACTING INJECTABLE ANTIRETROVIRAL THERAPY REGIMEN IN HIV US HEALTHCARE SETTINGS: INTERIM RESULTS FROM THE CUSTOMIZE STUDY
|
| |
| |
IDWeek 2020 Oct 21-25 virtual
Reported by Jules Levin
Cindy Garris,1 Ronald D'Amico,1 Paul Wannamaker,1 Nobuhle Mpofu,2 Colleen A. McHorney,3 Sonal G. Mansukhani,3 Jason Flamm,4 Blair Thedinger,5 Paul Benson,6 Daniela Salazar,7 Maggie Czarnogorski1
1ViiV Healthcare, Research Triangle Park, NC, USA; 2GlaxoSmithKline, Philadelphia, PA, USA; 3Evidera, Bethesda, MD, USA; 4Kaiser Permanente Sacramento, Sacramento, CA, USA; 5Kansas City Free Health Clinic, Kansas City, MO, USA;
6Be Well Medical Center, Berkley, MI, USA; 7University of Florida, Gainesville, FL, USA
Abstract
Background: Cabotegravir and rilpivirine long-acting (CAB+RPV LA) administered by monthly injection demonstrated non-inferiority compared to standard daily oral antiretroviral therapy (ART) at 48 weeks. This novel treatment offers a less frequent dosing alternative to daily oral pills but requires more frequent clinic visits. Patient perspectives on implementation of CAB + RPV LA in US healthcare settings were evaluated in an innovative Hybrid III implementation-effectiveness study (CUSTOMIZE).
Methods: This single-arm study enrolled virologically suppressed patients to receive monthly CAB+RPV LA across eight diverse US clinics. Patients were surveyed at Baseline (BL) prior to the first injection and at Month 4 (M4) prior to the fourth injection to evaluate clinic implementation of CAB+RPV LA, including Acceptability of Intervention (AIM) and Intervention Appropriateness (IAM) Measures. Subgroups were compared with Fisher's exact test.
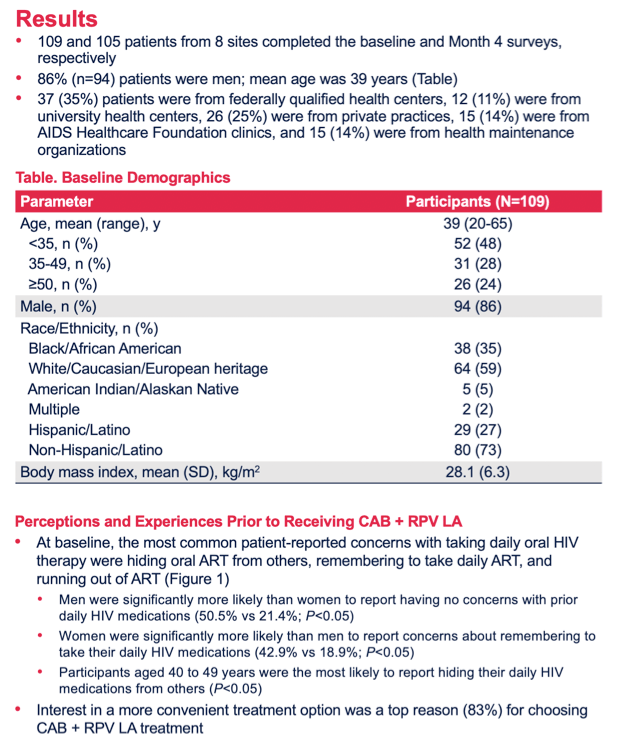
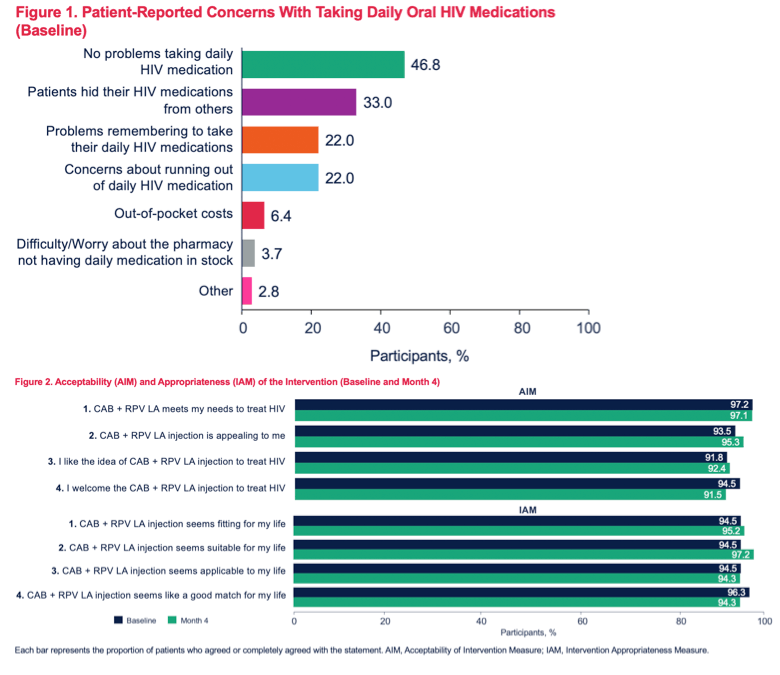
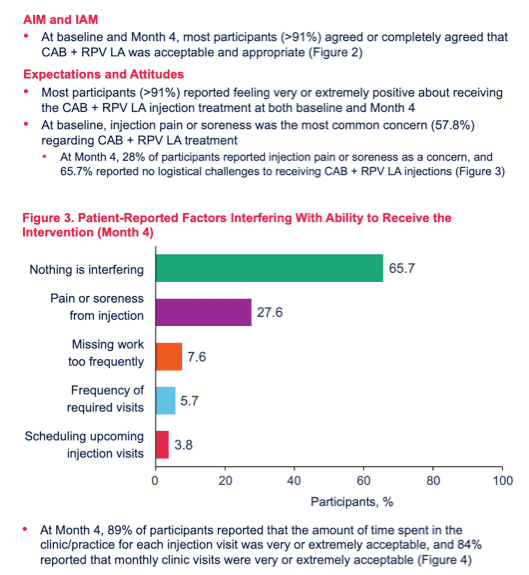
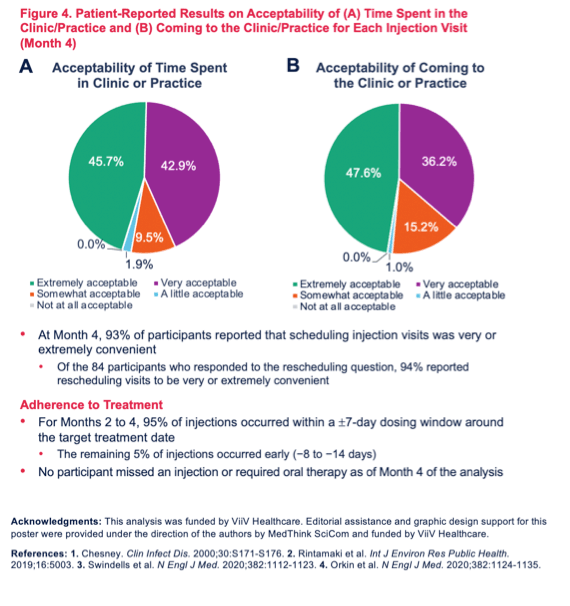
Results: A total of 109 and 105 patients completed BL and M4 surveys, respectively, and were 87% male; 59% Caucasian and 35% African American; 27% Hispanic/Latino; mean age 39 years (range 20-65). At BL, 33% reported hiding their oral ART from others, 22% reported problems remembering to take daily ART (female 43% > male 19%, p< 0.05), while 47% reported no problems with daily ART (male 51% > female 21%, p< 0.05). Patient "interest in a more convenient treatment option" (83%) was a top reason for choosing CAB+RPV LA treatment. Acceptability and appropriateness of CAB+RPV LA were high at BL and M4 (Figures). At M4, 84% of patients reported that monthly clinic visits were very/extremely acceptable and 66% reported no logistical challenges to clinic administered CAB+RPV LA. Injection pain/soreness was the most common worry at BL (58%); at M4, 28% reported injection pain/soreness as a concern. For Months 2-4, 95% of injections were within a +/-7-day dosing window (5% were early, -7 to -14 days). No patients missed an injection or required oral bridging.Figure 1. Acceptability of CAB+RPV LA: AIM
Conclusion: Most patients found CAB+RPV LA to be acceptable and appropriate, and a majority reported monthly appointments were highly acceptable. Initial implementation data suggest CAB+RPV LA is a convenient, appealing alternative treatment option for patients, with few reported logistical challenges.

|
| |
|
 |
 |
|
|