 |
 |
 |
| |
HCV TREATMENT AMONG ACTIVE INNER CITY DRUG USERS
WITH GLECAPREVIR/PIBRENTASVIR: THE GRAND PLAN STUDY
|
| |
| |
AASLD 2021 Nov 12-15
Brian Conway, David Truong, Leo Yamamoto, Rossitta Yung and Shawn Sharma, Vancouver Infectious Diseases Centre
Background: In order to achieve HCV elimination by 2030, specific strategies will need to be developed to diagnose and treat inner city residents and active drug users in a comprehensive fashion. Clinical trials suggest preserved efficacy of all oral treatment regimens in this population. There remains a need to confirm these data in challenging patient groups, such as those with unstable housing and drug use behaviors.
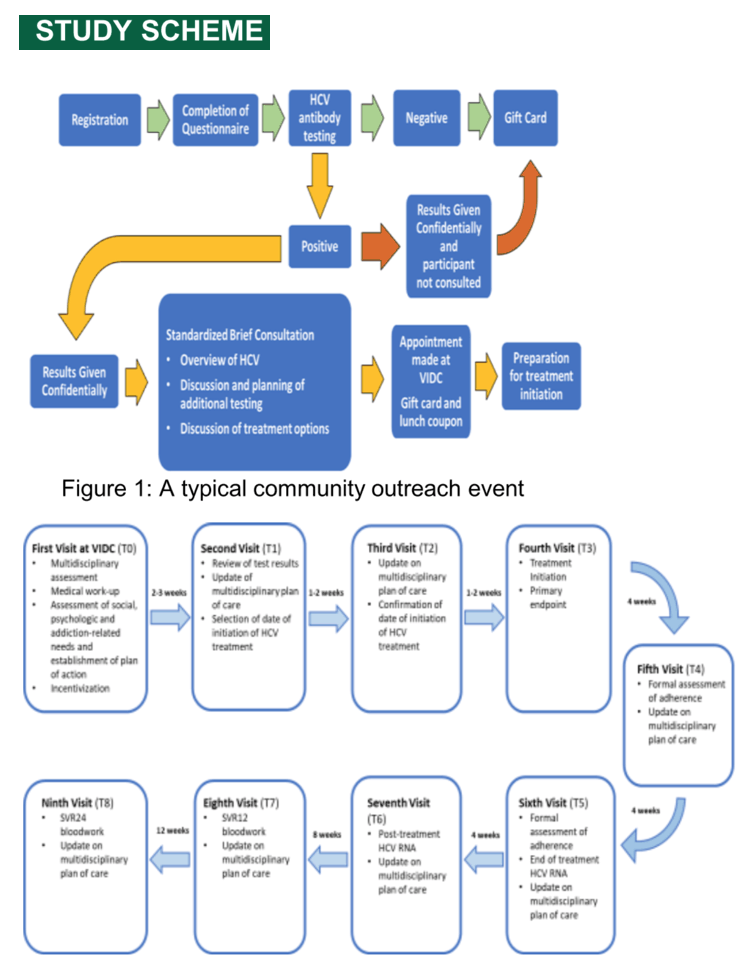
Methods: The current prospective observational study was undertaken to evaluate the safety and efficacy of HCV therapy among unselected inner city residents presenting for care. All participants were actively recruited through community outreach events and received HCV therapy with glecaprevir/pibrentasvir (G/P) for 8 weeks within the context of a dedicated multidisciplinary program of care. The primary outcome of this analysis was the cure of HCV infection and its correlates.
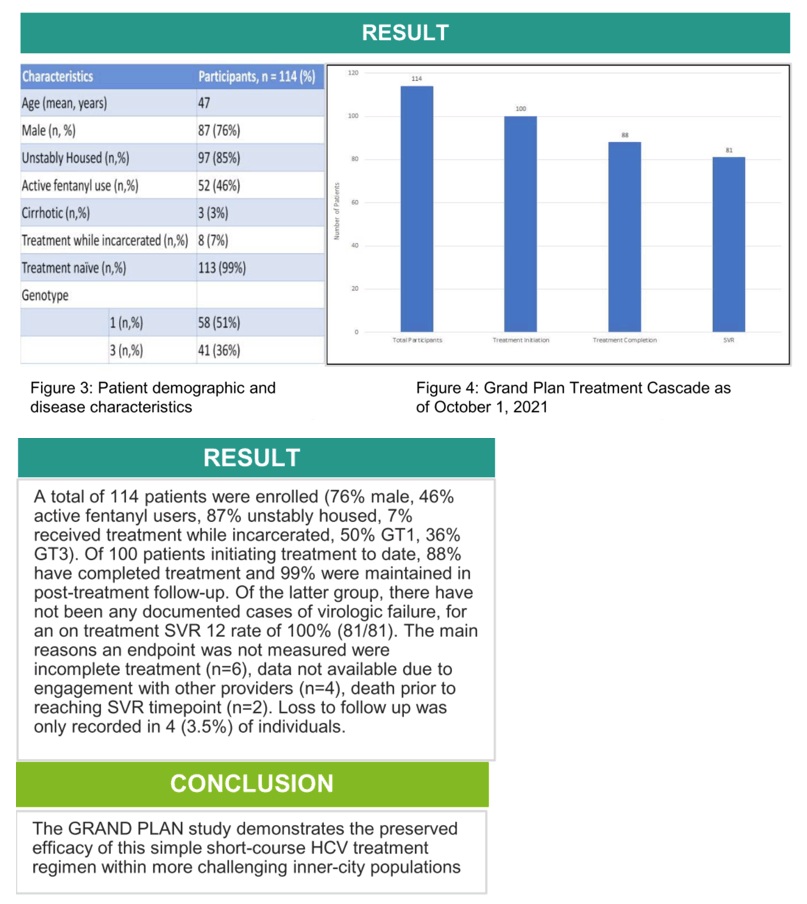
Results: A total of 98 patients were enrolled (82% male, 47% active fentanyl users, 87% unstably housed, 9% received treatment while incarcerated, 55% GT1, 35% GT3, 2% cirrhotic, 99% treatment naive). Of 89 patients initiating treatment, 97% completed treatment and 97% were maintained in post-treatment follow-up. Of the latter group, there have not been any documented cases of virologic failure. The main reasons an endpoint was not measured were incomplete treatment (n=4), loss to follow up (n=5), data not availabe due to engagement with other providers (n=2) and unstable to initiate treatment (n=1).
Conclusion: The GRAND PLAN study demonstrates the preserved efficacy of this simple short-course HCV treatment regimen within more challenging inner city populations compared to those previously evaluated in clinical trials.


|
| |
|
 |
 |
|
|