 |
 |
 |
| |
Treat HCV Early; SOF/DCV Had Lower SVR Rates
|
| |
| |
REAL-WORLD EFFECTIVENESS AND FACTORS ASSOCIATED WITH SVR12 IN A DIVERSE POPULATION OF HCV INFECTED PERSONS FROM FOUR LARGE EUROPEAN COHORTS
Lars Peters1, Joanne Reekie1, Soo Aleman2,3, Maurizio Zazzi4, Akaki Abutidze5, Anders Sonnerborg2, Barbara Rossetti4 and Nikoloz Chkhartishvili5, (1)Centre of Excellence for Health, Immunity and Infections, Rigshospitalet, Copenhagen, Denmark, (2)Department of Infectious Diseases, Karolinska University Hospital, Stockholm, Sweden, (3)Department of Medicine Huddinge, Karolinska Institutet, Stockholm, Sweden, (4)Department of Medical Biotechnology, University of Siena, Siena, Italy, (5)Infectious Diseases, AIDS & Clinical Immunology Research Center, Tbilisi, Georgia
Background: Data on HCV treatment outcomes from real-life cohorts have mainly come from single national cohorts of limited size. We aimed to investigate the effectiveness of direct-acting antivirals (DAA) and factors associated with achieving a sustained virological response (SVR) in a large diverse real-life population of HCV infected persons.
Methods: We pooled data from four HCV cohorts (Georgian National HCV Cohort, n=54,557; InfCare Hepatitis, Sweden, n=15,266; Tuscany, Italy, n=5,053; EuroSIDA, n=1,413) participating in the CARE project, with ≥12 weeks of follow up after end of treatment (EOT) with IFN-free DAA. SVR12 was defined as undetectable HCV-RNA >12 weeks after EOT. Logistic regression was used to examine factors associated with SVR12.
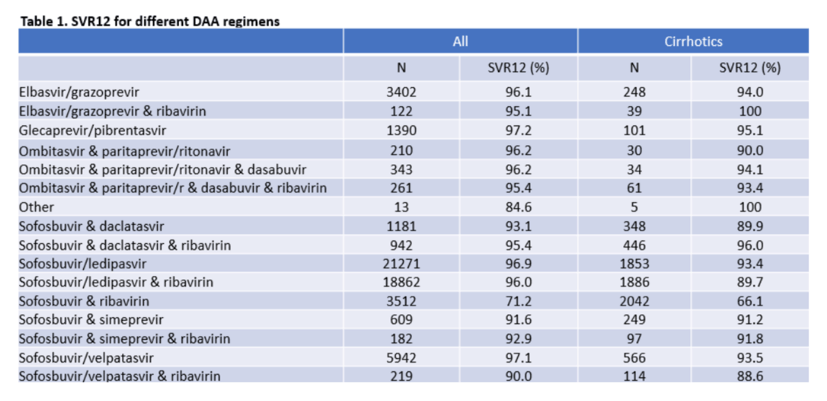
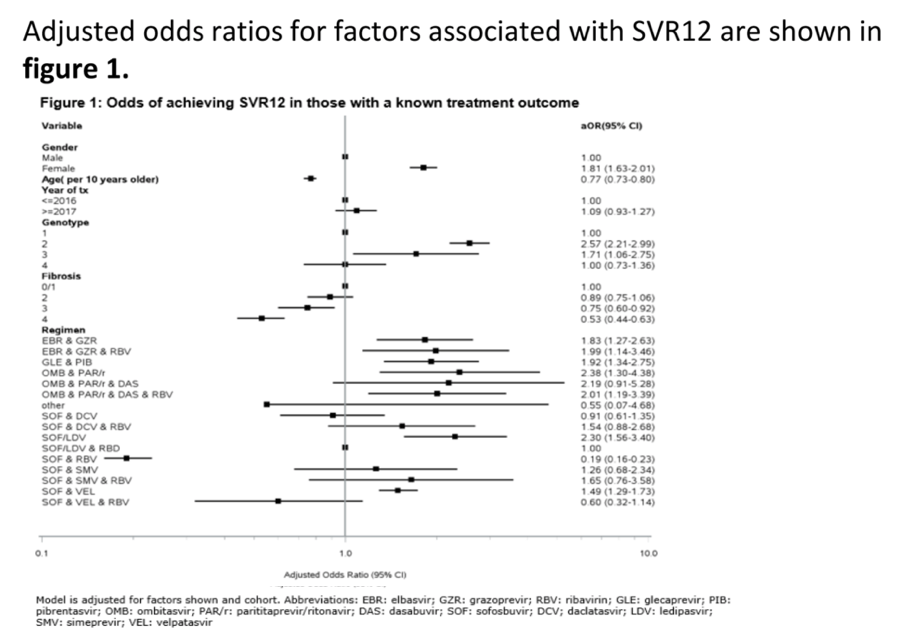
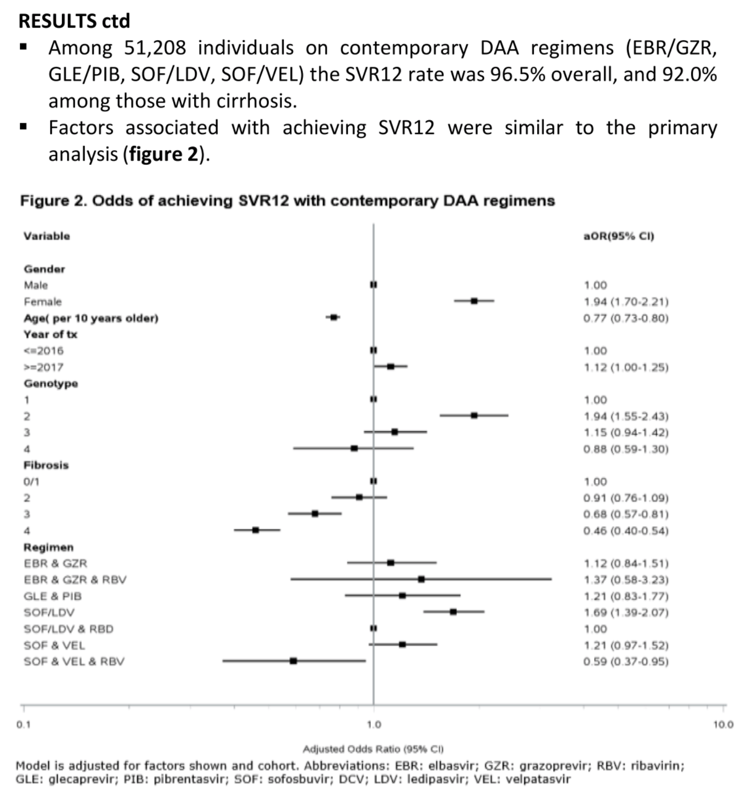
Results: Among 76,289 persons starting DAA, the median age was 48 years, 75% were males, 48% had genotype 1 and 14% had cirrhosis (F4). The most commonly used DAA regimens were SOF/LDV without RBV (36%) or with RBV (33%). A total of 58,461 (76.6%) had known SVR12 status. Participants with unknown SVR12 status were generally similar to those with known SVR12 status, but more likely to be from Georgia, male, younger, have F3/F4 fibrosis and started DAA in 2017 or later. Among those with known SVR12 status, 94.8% achieved SVR12. SVR12 rates were higher among females (96.6%) vs. males (94.1%) and decreased by age (97.2% in <40 years, 95.7% in 40-44 years, 94.3% in 45-49 years, 93.5% in 50-54 years, 93.5% in >55 years) and each stage of fibrosis (F0/F1=96.5%, F2=95.6%, F3=92.9%, F4 (cirrhosis)=85.6%), but did not differ by genotype. Adjusted odds ratios for factors associated with SVR12 are shown in the figure. In an analysis including only individuals with cirrhosis, higher odds of SVR12 was seen in females, younger persons, GT2 and GT3 (vs. GT1) and in those receiving SOF/LDV (vs. SOF/LDV + RBV) whereas those receiving SOF&DCV and SOF/VEL +/- RBV had lower odds of SVR12. Among 51,208 individuals on DAA regimens recommended by current guidelines (EBR/GZR, GLE/PIB, SOF/LDV, SOF/VEL) the SVR12 rate was 96.5% overall, and 92.0% among those with cirrhosis, but with similar trends as in the primary analysis.
Conclusion: In a large diverse population of 58,461 HCV infected persons with known SVR12 status, the overall SVR12 rate was 94.8%, and 96.5% among those on contemporary DAA regimens. We observed a gradual decrease in SVR12 with increasing age and fibrosis stage. Our findings support early DAA therapy for all HCV infected individuals using DAA regimens recommended by current guidelines to optimize treatment outcomes.



|
| |
|
 |
 |
|
|