 |
 |
 |
| |
PARTNERSHIPS FOR ELIMINATION: AMBULATORY CARE CLINICAL
PHARMACISTS DISMANTLING BARRIERS TO HEPATITIS C CURE at St Barnabas in NJ
|
| |
| |

Ammie Patel1,2,3, Su H Wang4, Alyssa Gallipani1,5, Ruth Brogden6, Binghong Xu7, Mary Adedeji7 and Jaymie Yango6, (1)Rwjbh Corporate Pharmacy, (2)Pharmacy Practice and Administration, Ernest Mario School of Pharmacy, Rutgers,(3)Pharmacy Practice and Administration, Rutgers, the State University of NJ, (4)World Hepatitis Alliance, UK, (5)School of Pharmacy & Health Science, Fairleigh Dickinson University,(6)Medicine, Saint Barnabas Medical Center, (7)Center for Asian Health, Saint Barnabas Medical Center
Background: Liver cancer rates are rising in the US, and viral hepatitis (VH) accounts for more than 65% of cases. An automated emergency department (ED) VH screening program was initiated in 2018 at a hospital in New Jersey. In 2020, it was expanded to the inpatient setting and modified to follow updated Centers for Disease Control (CDC) recommendations from birth cohort to universal hepatitis C virus (HCV) screening for all adults. Although direct acting antiviral (DAA) therapy are remarkably effective and safe at achieiving sustained virologic response (SVR) with short treatment courses, there are still barriers that limit successful uptake. 1 Drug-drug interactions (DDI) can reduce rates of cure or lead to major adverse events. High cost and requirement for prior authorization take additional time and resources, which results in delay or denial of therapy. Therefore, scale-up HCV screening and pharmacist integration are essential for HCV elimination.
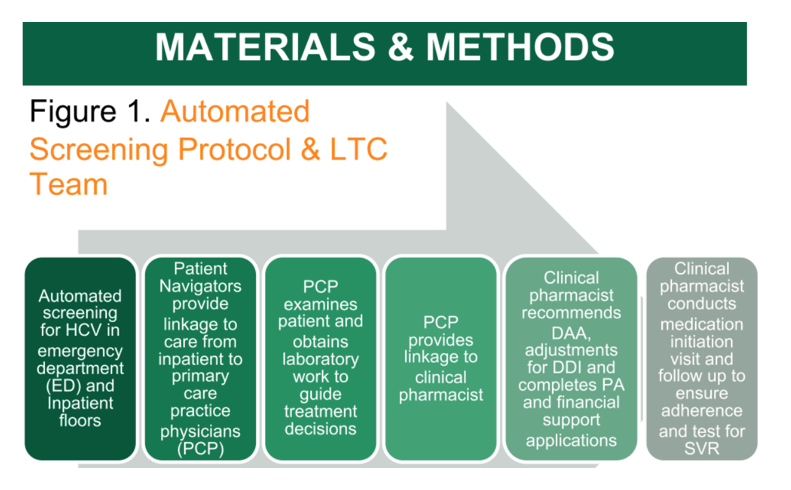
Methods: Patient navigators (PN) contacted those found to have HCV and provided linkage-to-care (LTC) for evaluation and therapy. Clinical pharmacists embedded in the primary care based practice (PCP) were consulted to recommend the HCV therapy, complete prior authorization, apply for financial assistance, monitor laboratory parameters, counsel patients on medication adherence and side effects, and follow for cure.
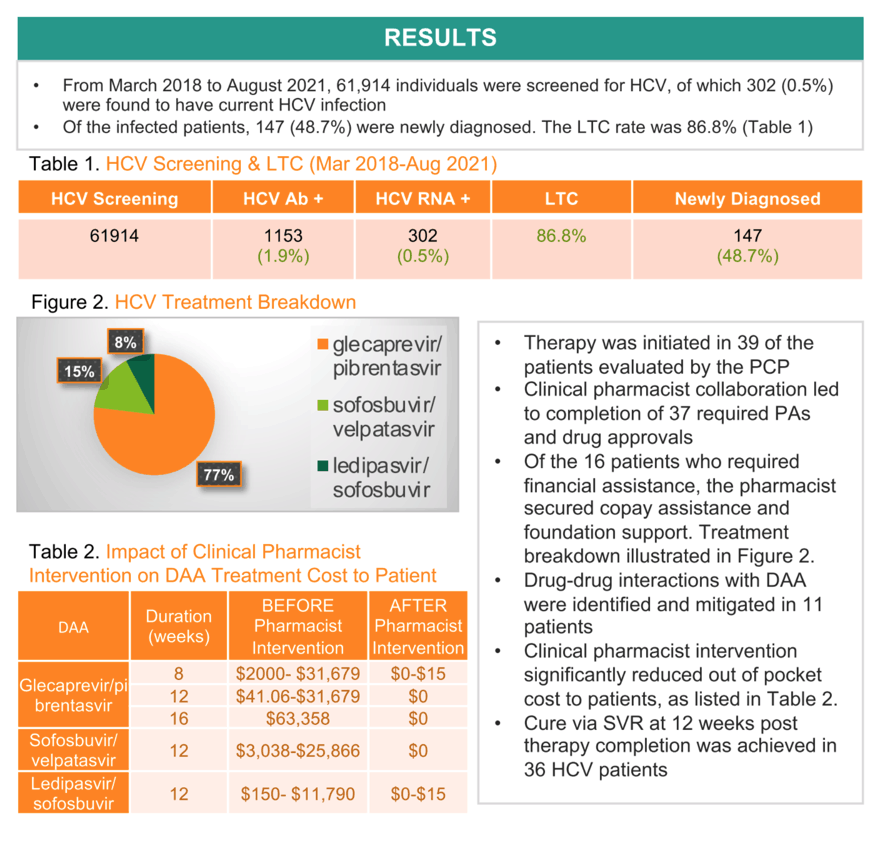
Results: From Mar 2018 to Mar 2021, we screened 50,873 individuals of HCV with 259 (0.5%) found to have current HCV infection and 128 (49.4%) were newly diagnosed. The LTC rate was 86.9%. The PCP evaluated 47 HCV patients and initiated therapy in 38. The pharmacist collaboration led to completion of 36 required prior authorization and drug approvals. Of the 15 patients who required financial assistance, the pharmacist secured copay assistance and foundation support. Drug-drug interactions with DAA were identified and mitigated in 11 patients. Treatment breakdown was: 29 (76.3%) glecaprevir/pibrentasvir, 6 (15.8%) sofosbuvir/velpatasvir & 3 (7.9%) ledipasvir/sofosbuvir. Pharmacist intervention significantly reduced out of pocket cost to patients (table 1). Thus far, 35 achieved cure with SVR at 12 weeks post therapy completion.
Conclusion: Integration of clinical pharmacist services within the VH program has contributed to the team based approach in dismantling barriers of HCV cure, including cost and access, drug interactions, adverse reactions, specialist requirements and nonadherence.


|
| |
|
 |
 |
|
|