 |
 |
 |
| |
A Case Series of Safety and Efficacy of Crushed
Sofosbuvir/Velpatasvir in Hepatitis C Infected Patients
|
| |
| |
AASLD 2021 Nov 12-16
Shobha Joshi1 Martin Cohen2, Rivka Katz2, Paul J Gaglio3, Robert S. Brown Jr.4, David Salemo5, Tatyana Kushner6 Harmit S Kalia7, Anthony Michaels8, Rishika Montiani9, Nicole Cheng10, Katherine Fuller10, Alexander Cyganowski11,
Gerard C Quigley12, Monica Graybeal13, Heather King14, Nelson Cheinqueru15, Doona Khoshabafard15 Farrah Black15
1Ochsner Health Multi-Organ Transplant Institute. 2University Hospitals, Specialty Pharmacy. 3Columbia University Medical Center-NY Presbyterian. 4Weill Cornell Medicine Center. 5-NewYork-Presbyterian Hospital 6-Icahn Schoool of Medicine at Mount Sinai.
7NYU Langone Transplanl Institute. 8-The Ohio Stale University Wexner Medical Center. 9HopeHealth, Inc. 10Emory University Hospital Midtown. 11Gallup Indian Medical Center. 12Banner Advanced Liver Disease Institute. 13Yakima Valley Farm Workers Clinic.
14Dime Community Heatth Operating as Heritage Health. 15Gilead Sciences Inc, Foster City, CA
Background: Hepatitis C virus (HCV)-related liver disease is a cause of significant morbidity and mortality if left untreated. There is limited safety and efficacy data using crushed Sofosbuvir/Velpatasvir (SOF/VEL). to treat HCV. There are multiple instances where a patient is unable to swallow a tablet and the only option is to take the medication crushed.
Methods: This case series includes patients from 13 US-based medical centers where providers reported deidentified data on patients who received crushed (SOF/VEL). Standardized case report forms were collected and the data is summarized. The data collected include specific medications, specific past medical and surgical history, genotype, viral load (HCV RNA measurement), fibrosis score, adverse events and sustained virologic response (undetectable HCV RNA 12 or more weeks after stopping treatment; SVR).
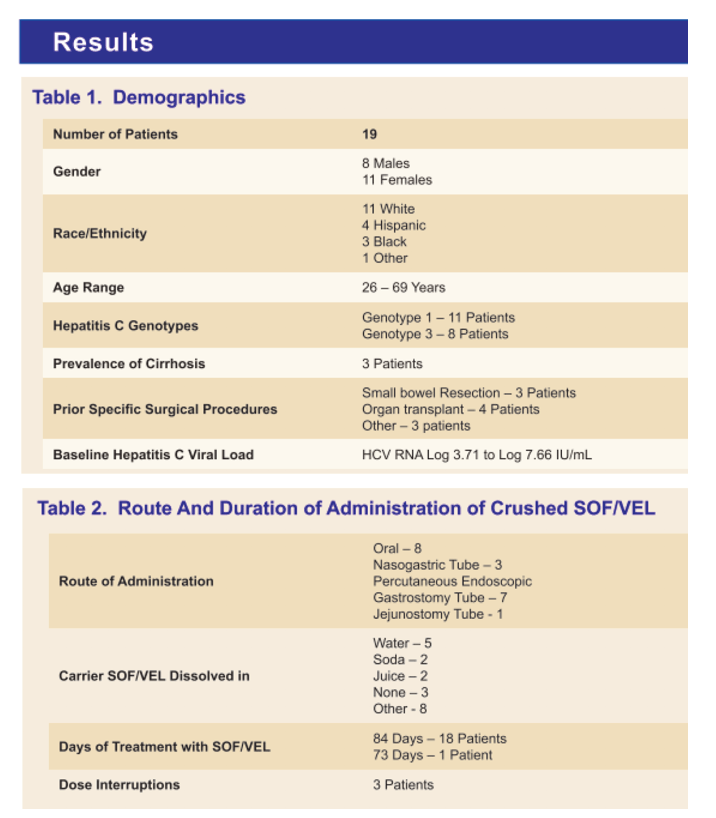
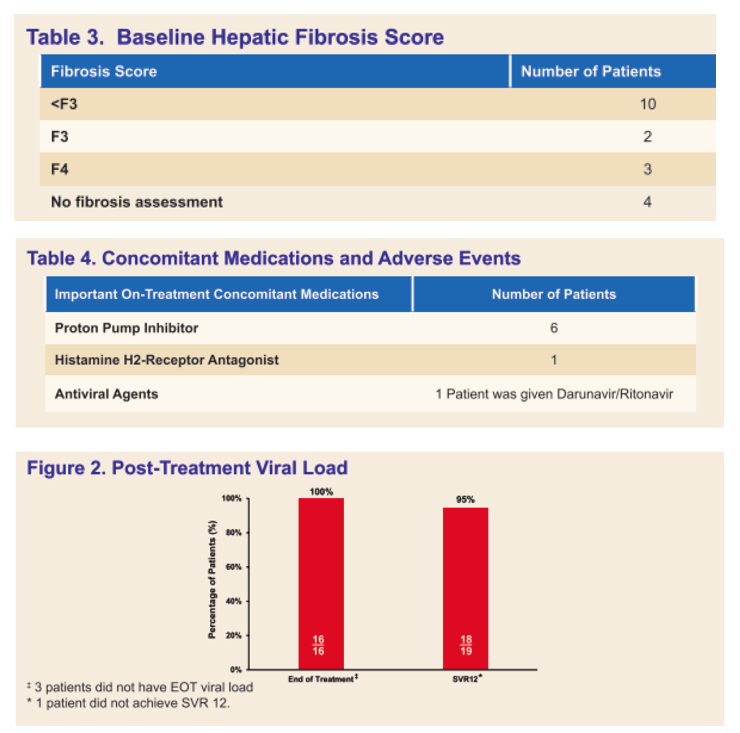
Results: A total of 19 cases were collected. 8 Male and 11 Female. Race: 11 white, 4 Hispanic, 3 black, 1 other. 16/16 patients (100%) who had end of treatment (EOT) viral load, were viral negative; 3 did not have EOT viral load. 18/19 patients (95%) had SVR 12. 11 patients had genotype 1 and 8 had genotype 3. Fibrosis score were available for 15 patients, 2 F0, 2 F0-F1, 2 F1-F2, 2 F2, 1 F2-F3, 1 F3, 1 advanced fibrosis, 1 no advanced fibrosis, and 3 F4. No patient reported on-treatment adverse events, none discontinued therapy. 3 patients had dose interruptions, of whom 2 completed 84 days of therapy and one 73 days; 16 patients received 84 consecutive days of therapy. 6 patients were on PPI during therapy, 1 on H2 blocker and 1 on antiviral Darunavir/Ritonavir. 3 patients had small bowel resection, 4 had organ transplant and 3 had other surgical procedures. 8 patients received crushed oral SOF/VEL (1 dissolved in soda, 6 dissolved in other and 1 none), 3 patients received crushed SOF/VEL via NG tube (1 in juice and 2 in water). 7 patients received dosing via PEG-tube (1 mixed in soda, 2 in water, and 4 in other). 1 received dosing via J tube mixed in water.
Conclusion: This case series demonstrates high hepatitis C cure rates and safety in patients receiving crushed SOF/VEL, despite historically considered negative predictors of failure, such as genotype 3, certain medications, prior surgical procedures and dose interruptions. There were no on-treatment adverse events or treatment discontinuation.


|
| |
|
 |
 |
|
|