 |
 |
 |
| |
LONG-TERM PERSISTENCE OF RESISTANCE-ASSOCIATED SUBSTITUTIONS
(RASS) AFTER DAA TREATMENT FAILURE IN PATIENTS WITH CHRONIC HCV INFECTION
|
| |
| |
AASLD 2021 Nov 12-15
AASLD: Excellent Efficacy and Safety of Sofosbuvir, Glecaprevir, Pibrentasvir and Ribavirin for Retreatment of Chronic Hepatitis C after Sofosbuvir, Velpatasvir and Voxilaprevir Failure - (11/16/21)

Julia Dietz1, Christiana Graf1, Beat Muellhaupt2, Peter Buggisch3, Jörn M Schattenberg4, Kai-Henrik Peiffer5, Georg Dultz1, Thomas Discher6, Julian Schulze Zur Wiesch7, Tobias Mueller8, Thomas Berg9, Christoph Neumann-Haefelin10, Christoph Berg11, Stefan Zeuzem12 and Christoph Sarrazin1, (1)Department of Internal Medicine I, Goethe University Hospital Frankfurt, Germany, (2)Department of Gastroenterology and Hepatology, University Hospital Zurich, Switzerland, (3)Ifi-Institute for Interdisciplinary Medicine, (4)Metabolic Liver Disease Research Program, I. Department of Medicine, University Medical Center of the Johannes Gutenberg-University, Mainz, Germany, (5)Internal Medicine I, Hospital of the Goethe University Frankfurt, (6) Department of Internal Medicine II, Justus-Liebig-University Giessen, (7)University Medical Center Hamburg-Eppendorf,(8)Department of Hepatology and Gastroenterology, Charité Universitätsmedizin Berlin, Germany, (9)Department of Medicine II - Division of Hepatology, Leipzig University Medical Centre, (10)University Hospital Freiburg, (11)University Hospital Tübingen, (12)Department of Internal Medicine I, Goethe University Hospital
Background: Treatment of chronic hepatitis C virus (HCV) infection with direct acting antivirals (DAAs) is highly effective. Virologic treatment failure is associated with the selection of resistance-associated substitutions (RASs) in DAA target regions and data on the long term persistence of RASs after treatment failure are limited. This study evaluated the persistence of NS3, NS5A and NS5B RASs for a time period of more than 24 months after end of treatment (EOT).
Methods: We included samples from 749 patients with a HCV genotype (GT) 1, 3 or 4 infection and virologic treatment failure to a DAA treatment which were collected in the European resistance Database. NS3, NS5A and NS5B were population sequenced and RASs conferring a >2-fold increased DAA susceptibility were analyzed.
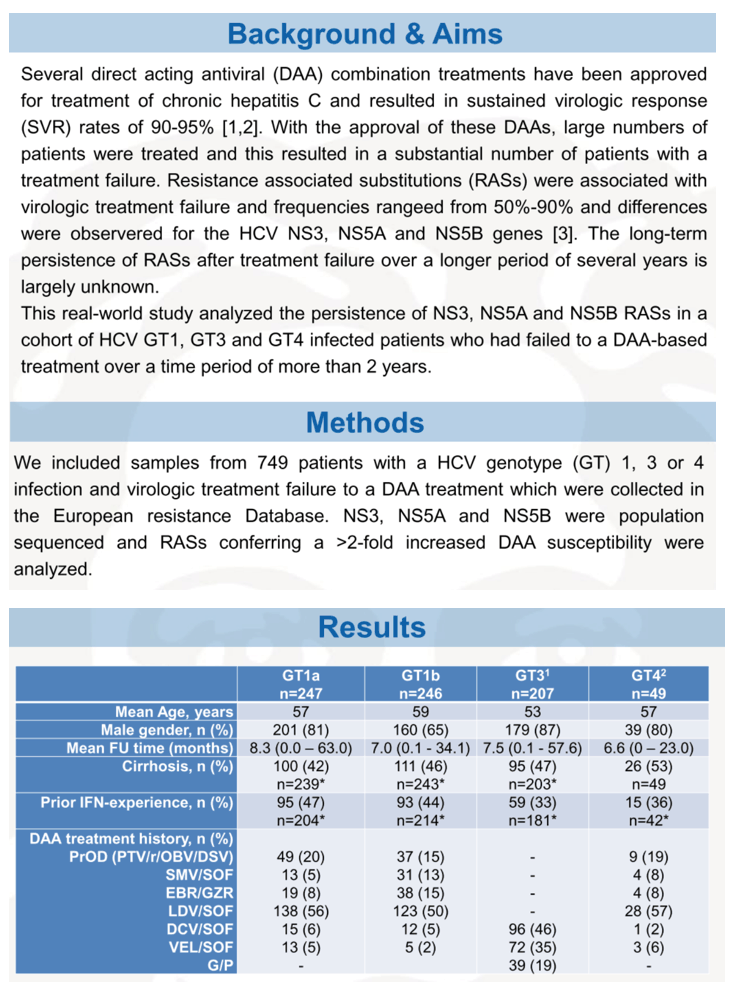
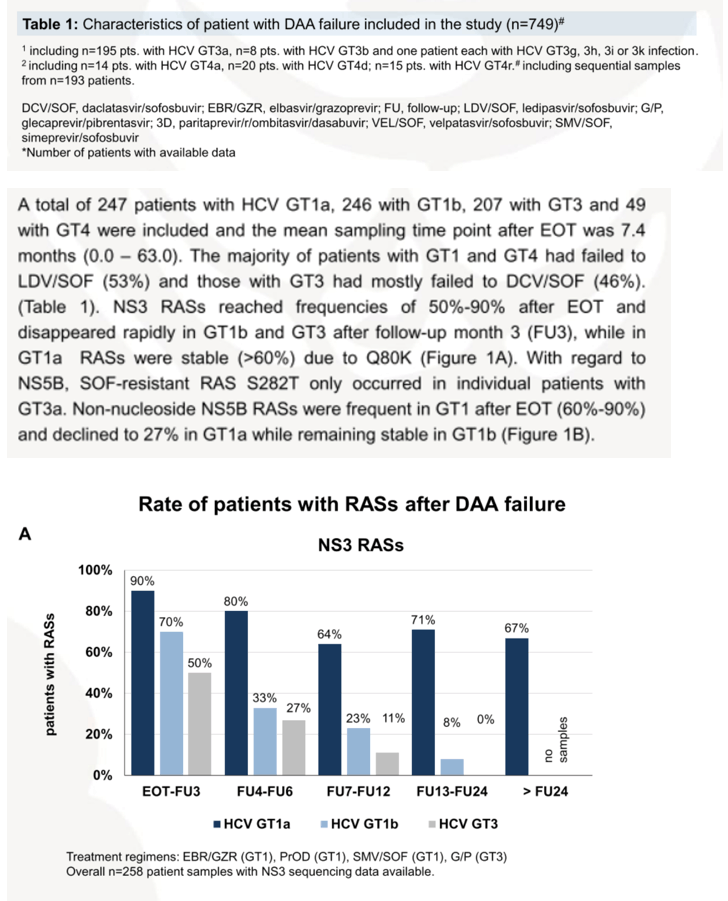
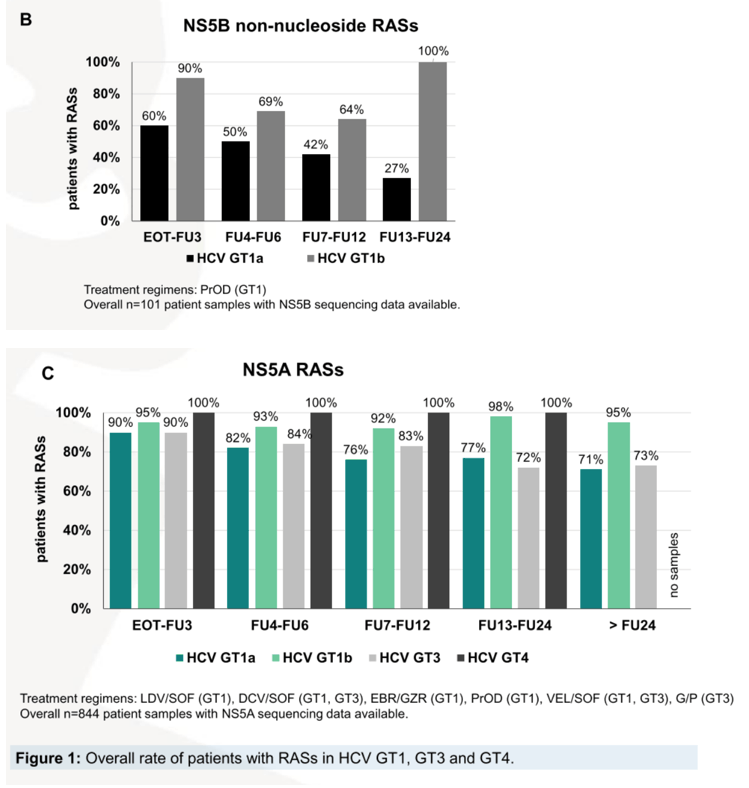
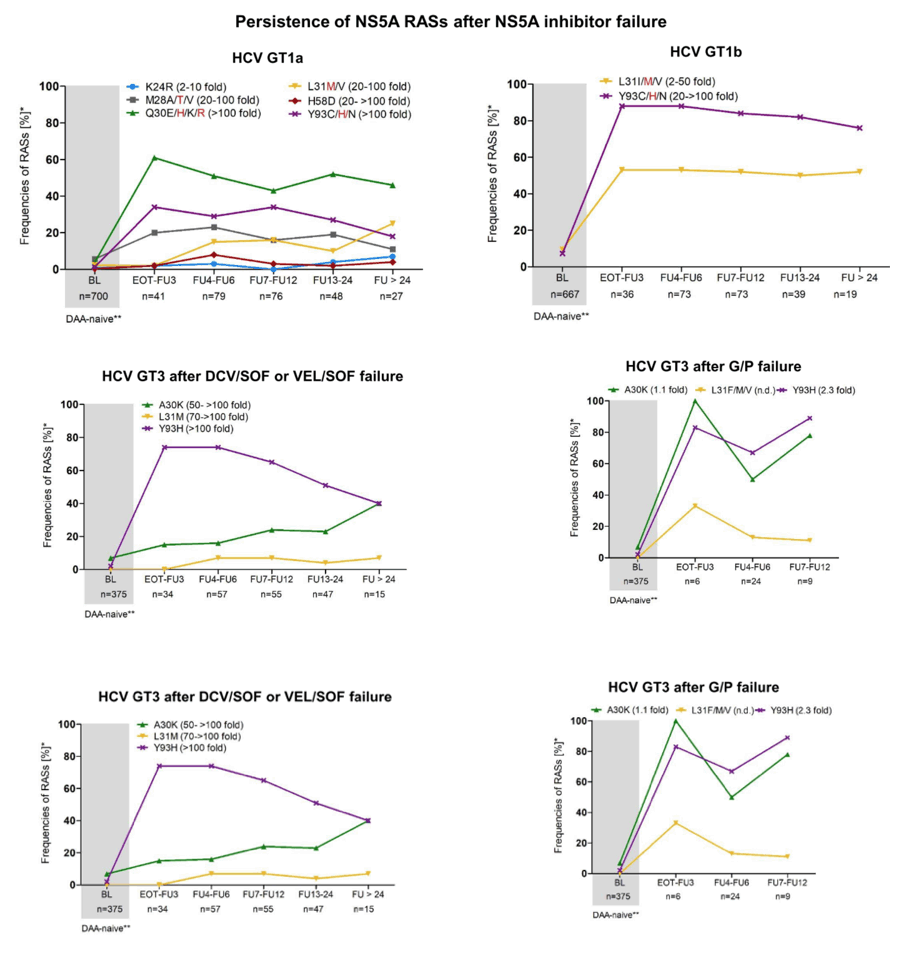
Results: A total of 247 (33%) patients with GT1a, 246 (33%) with GT1b, 207 (27%) with GT3 and 49 (7%) with GT4 infection were included, the median sampling point after EOT was 7.6 months (0.0-63.0). The patients had not responded to LDV/SOF (39%, 289/749), DCV/SOF (17%, 124/749), PrO±D (13%, 95/749), VEL/SOF (12%, 93/749), GZR/EBR (8%, 61/749), SMV/SOF (6%, 48/749) or G/P (5%, 39/749). The frequencies of NS3 RASs were 70-90% after EOT in protease inhibitor-experienced patients and RASs disappeared rapidly in GT1b, GT3 and GT4 after follow-up month 3 (FU3), which was largely due to the loss of variants at position 168. RASs in GT1a were stable due to Q80K. In NS5B, nucleotide RASs were very rare and S282T only occurred in with GT3a. NS5A RASs were very common in NS5A inhibitor-experienced patients across all GT (90-95% after EOT) and were detectable in 70% of patients even after FU24. L31M and Y93H were most common in GT1b. In GT1a and GT4 different RASs at positions 28, 30, 31 and 93 were detected. A30K and Y93H were frequent in GT3. While NS5A RASs remained stable in GT1b, RASs slightly decreased in GT1a and GT3 after FU24 (GT1a, 71%, GT1b, 95%, GT3, 73%), which was mainly caused by the decline of Y93H. For GT4, data sets are not yet competed at later time points, but there was also a trend towards an Y93H decrease.
Conclusion: NS3 and NS5B RASs quickly disappeared, whereas high rates of persistent NS5A RASs were observed more than two years after end of DAA treatment in all GT with only slow decline in certain HCV GTs. This may have an impact on retreatment with first generation DAAs in resource-limited settings and for the achievement of the global HCV elimination aims.


|
| |
|
 |
 |
|
|