 |
 |
 |
| |
HIGH RATE OF HEPATITIS C REINFECTION IN
AN ADULT CUSTODIAL SETTING IN AUSTRALIA
|
| |
| |

AASLD 2021 Nov 12-15
Andrew Teo1, Lyn Cadman2, Rajendra Prakash2, Hayley Thompson3, Cherie Bloomfield4, John Willett5, Paul Chapman6, Tony M. Rahman3 and Enoka S Gonsalkorala1, (1) Gastroenterology, Caboolture Hospital, (2)Medical, Woodford Correctional Centre, (3)The Prince Charles Hospital, (4) Coding and Analytics, Caboolture Hospital, (5)Pharmacy, Caboolture Hospital, (6)Infectious Disease, Royal Brisbane & Women's Hospital
Background: Chronic hepatitis C (HCV) infection is highly prevalent in custodial settings. The reported prevalence of anti-HCV in Australian prisons is 30% and is as high as 50% in people who inject drugs in prison. In the state of Queensland (20% of Australian population), 39% of all prescriptions for direct-acting antiviral (DAA) agents in 2019 were for people in custodial settings. Here we describe the Hepatitis C treatment program and its outcomes in a large maximum-security prison (population 1446) in Queensland, Australia.
Methods: HCV treatment information is prospectively recorded in a secure database at the Woodford Correctional Centre. We retrospectively reviewed the database to assess the outcomes of people who were HCV polymerase chain reaction (PCR) positive between August 2018 and May 2021.
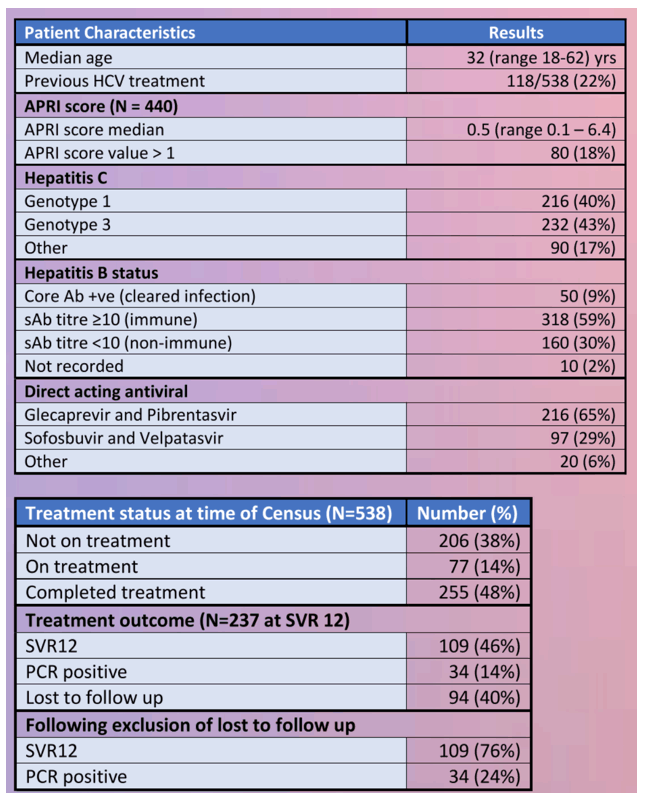
Results: 538 people with HCV mono infection (positive PCR) were included in the study. All were male and the median age was 32 years (range 18-62). A majority (83%) were infected with genotype 1 (N=216) or 3 (N=232). Hepatitis B status was cleared infection (cAb positive) 50/538 (9%), immune (sAb titre ≥10) 318/538 (59%), non-immune 160/538 (30%) and unknown 10/538 (2%). AST to platelet ratio index (APRI) score was available for 442 people. Median score was 0.5 (range 0.05-14.2) and 82/442 (19%) had a score ≥ 1. At time of data census 206/538 (38%) were not on treatment, 77/538 (14%) were on DAA therapy and 255/538 (48%) had completed treatment. Majority were treated with glecaprevir and pibrentasvir (N=216, 65%) or sofosbuvir and velpatasvir (N=97, 29%). 212/255 were eligible for sustained virological response (SVR) testing. 69/212 (33%) were lost to follow up, 109/212 (51%) have achieved SVR and 34/212 (16%) were PCR positive 12 weeks following end of treatment (EOT). On intention to treat analysis (lost to follow up excluded) 76% (109/143) achieved SVR and 24% (34/143) were PCR positive 12 weeks after EOT.
Conclusion: In our cohort a significant proportion of people did not achieve SVR. Taking in to account the age of the cohort, median APRI score and efficacy of current DAA regimens, we conclude that the failure to achieve SVR is due to reinfection and not due to treatment failure. Attention to all aspects of the hepatitis C care cascade, including harm minimization strategies and treatment scale-up strategies, is essential if we are to achieve the World Health Organization targets for HCV infection by 2030.


|
| |
|
 |
 |
|
|