 |
 |
 |
| |
RARE HCV GENOTYPES: PREVALENCE AND RESISTANCE-ASSOCIATED
SUBSTITUTIONS IN DAA-NAÏVE AND DAA-EXPERIENCED PATIENTS
|
| |
| |
AASLD 2021 Nov 12-15
Julia Dietz1,2,3,4, Christiana Graf1, Beat Muellhaupt5, Peter Buggisch6, Kerstin Port7, Jorn M Schattenberg8, Kai-Henrik Peiffer9, Georg Dultz1, Christophe Moreno10, Christoph Berg11, Thomas Berg12, Stefan Zeuzem13 and Christoph Sarrazin1,14, (1) Department of Internal Medicine I, Goethe University Hospital Frankfurt, Germany, (2)Department of Internal Medicine I, Goethe-University Hospital, (3)Department of Internal Medicine I, University Hospital Frankfurt, (4)Department of Internal Medicine I, University Hospital Frankfurt, Germany, (5) University Hospital Zürich, (6)Ifi-Institute for Interdisciplinary Medicine, (7)Department of Gastroenterology, Hepatology and Endocrinology, Hannover Medical School, Hannover, Germany, (8)Metabolic Liver Research Program, University Medical Center, (9)Internal Medicine I, Hospital of the Goethe University Frankfurt, (10)Université Libre De Bruxelles, (11) University Hospital Tübingen, (12)Department of Medicine II - Division of Hepatology, Leipzig University Medical Centre,(13)Department of Internal Medicine I, Goethe University Hospital, (14)St. Josefs-Hospital Wiesbaden, Germany
Background: High cure rates of hepatitis C virus (HCV) infection with short courses of treatment with direct acting antivirals (DAAs) make global eradication of HCV feasible. However, second generation DAAs are not available in resource-limited settings. Recent reports suggest that rare HCV genotypes (GT) exist in certain countries and are associated with lower SVR rates. This study investigated the prevalence of rare HCV GT and resistance-associated substitutions (RASs) among European DAA-naÏve and DAA-failure patients. Methods: We have collected samples from 7300 patients in the European resistance database in Frankfurt, of whom 4499 were DAA-naÏve and 1258 had failed to an IFN-free DAA treatment. NS3, NS5A and NS5B population sequencing was conducted to determine the HCV GT and RASs conferring a >2-fold increased DAA susceptibility were analyzed.
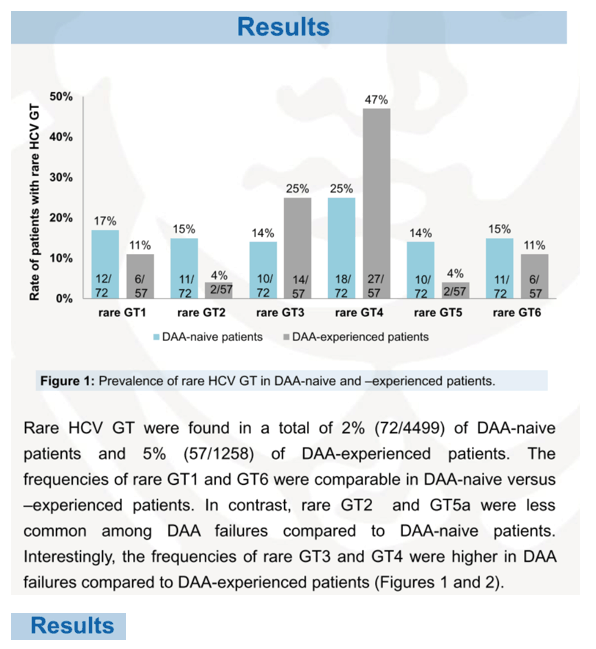
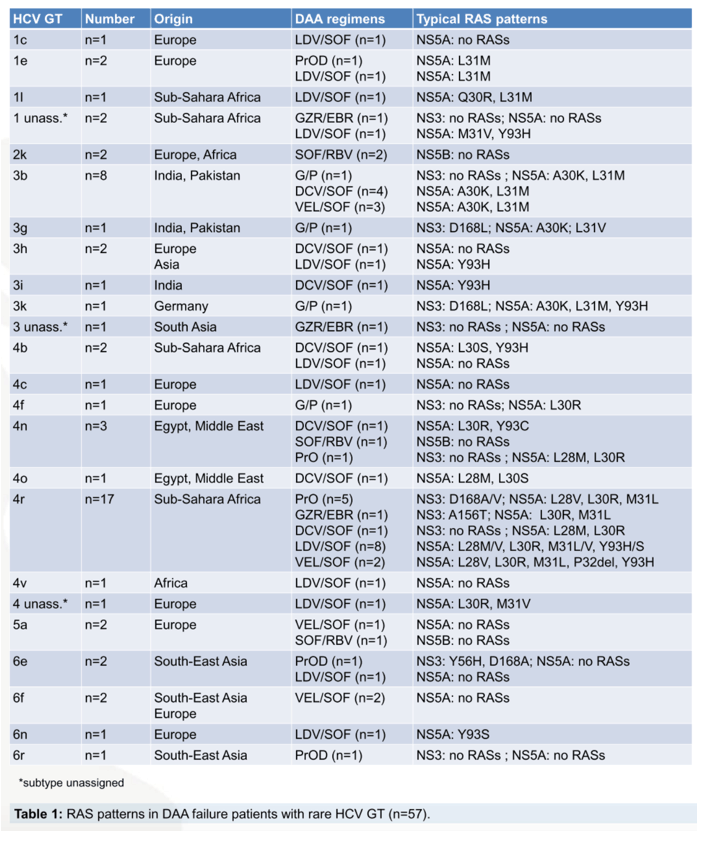
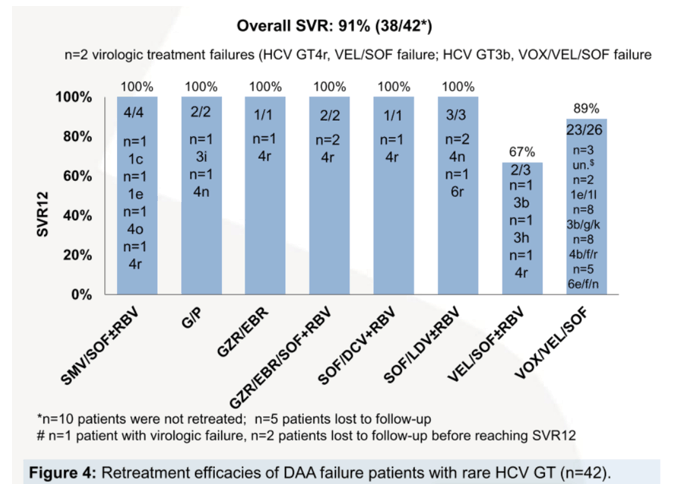
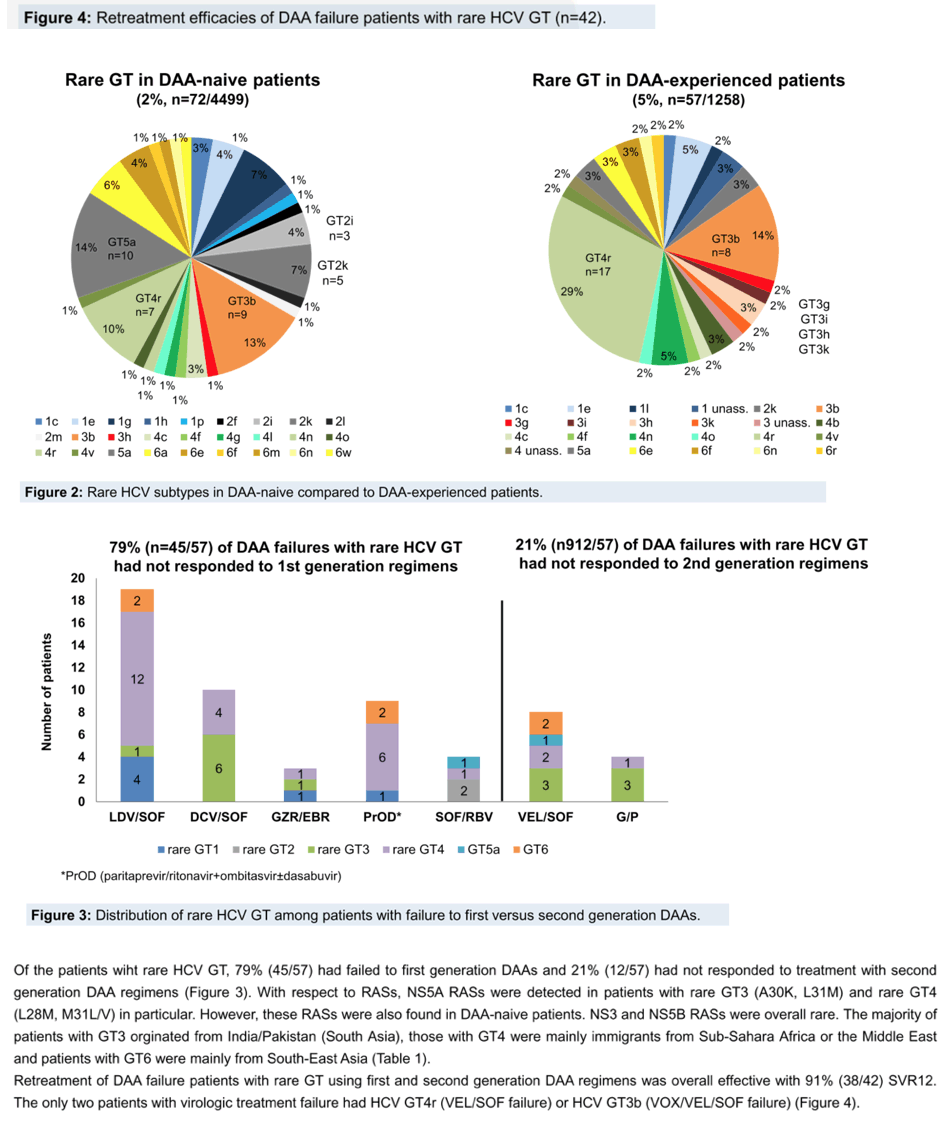
Results: The prevalence of rare HCV GT was 2% (72/4499) in DAA-naÏve and 5% (57/1258) in DAA-experienced patients and the frequencies of rare GT1 (1c, 1e, 1g, 1h, 1l, 1p) and GT6 (6a, 6e, 6f, 6m, 6n, 6r) were comparable in both groups (GT1, 17% and 11%; GT6, 15% and 11%). Rare GT2 (2i, 2k, 2l, 2m) and GT5a were less common among DAA failures (4% each) compared to DAA naÏve patients (15% each). However, the frequencies of rare GT3 (3b, 3g, 3h, 3i, 3k) and GT4 (4b, 4c, 4f, 4m, 4n, 4o, 4r, 4v) were higher in DAA failures (GT3, 25%; GT4, 47%) compared to DAA-naÏve patients (GT3, 14%; GT4, 25%). Among DAA failures, rare GT were more common after failure to first generation regimens (LDV/SOF, DCV/SOF, GZR/EBR, 3D) (79%, 45/57) compared to second generation pangenotypic regimens (VEL/SOF, G/P) (21%, 12/57). Characteristic NS5A RASs were particularly found in rare GT4 (L28M, M31L/V) and rare GT3 (A30K, L31M) and Y93H was rare. Patients with rare GT1, GT2 or GT4 mainly originated from Africa, those with rare GT3 from India/Pakistan, GT5a from South Africa and patients with rare GT6 came from South-East Asia. Retreatment of DAA failures using first and second generation DAAs was effective with 91% SVR (38/42).
Conclusion: Rare HCV GT were more common in DAA failures (5%) compared to DAA-naÏve patients (2%) especially for certain HCV subtypes and were more frequent after failure to first generation compared to second generation DAA regimens. Patients originated mainly from Africa or Asia and first-generation DAAs available in these countries may not be effective enough to achieve the global HCV elimination goals.

|
| |
|
 |
 |
|
|