 |
 |
 |
| |
THE PHARMACOKINETIC AND SAFETY PROFILES OF RO7445482 siRNA
[RG6346] ARE SIMILAR BETWEEN ASIAN AND NON-ASIAN HEALTHY
VOLUNTEERS AND CHRONIC HEPATITIS B PATIENTS IN A PHASE 1 STUDY
|
| |
| |
AASLD 2021 Nov 12-15

Edward J. Gane1, Nelson Guerreiro2, Simon Buatois2, Won Kim3, Jung-Hwan Yoon4, Tien Huey Lim5, William Sievert6, Alexander Thompson7, Wattana Sukeepaisarnjaroen8, Pisit Tangkijvanich9, Christian Schwabe10, Bob D. Brown11, Miriam Triyatni2, Qingyan Bo2, Hardean Achneck11 and Man-Fung Yuen12, (1)New Zealand Liver Transplant Unit, Auckland City Hospital and University of Auckland, Auckland, New Zealand, (2)Roche, (3)Department of Internal Medicine, Seoul National University College of Medicine Seoul Metropolitan Government Seoul National University Boramae Medical Center, (4) Department of Internal Medicine and Liver Research Institute, Seoul National University College of Medicine, Seoul, Korea,(5)Middlemore Hospital, New Zealand, (6)Monash Health and Monash University, Melbourne, Australia, (7)Department of Gastroenterology, St Vincent's Hospital Melbourne, (8) Srinagarind Hospital, Khon Kaen University, Thailand, (9) King Chulalongkorn Memorial Hospital, Bangkok, (10)New Zealand Clinical Research, (11)Dicerna Pharmaceuticals, Inc., (12)Queen Mary Hospital, the University of Hong Kong
Background: RO7445482 (f.k.a. RG6346) is a short-interfering RNA designed to inhibit hepatitis B viral gene expression through targeting of the S open reading frame of the viral genome. RO7445482 is conjugated to GalNAc, enabling liver targeting through binding to the asialoglycoprotein receptor. The present study compared the pharmacokinetic (PK) and safety profile of RO7445482 in Asian and non-Asian subjects.
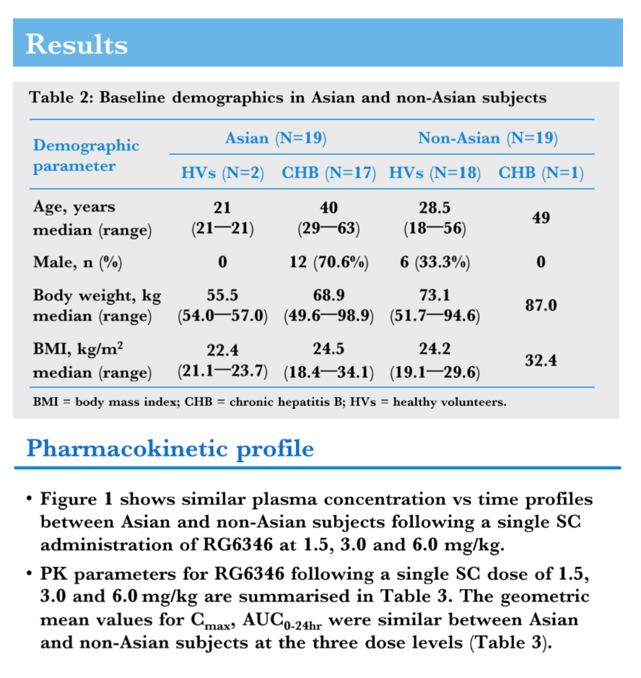
Methods: Data from a Phase 1 trial of a single subcutaneous (SC) dose in healthy volunteers (HV), and a single or 4 monthly SC doses in patients with Chronic Hepatitis B (CHB) were analyzed for PK and safety. Asian (2 HV/17 CHB, age: 21-63, 7 female/12 male) and Non-Asian (18 HV/1 CHB, age: 18-56, 13 female/6 male) subjects were included. PK parameters of RO7445482 were calculated by non-compartmental analysis from plasma concentration data following the 1st dose of 1.5, 3.0 or 6.0 mg/kg. A population PK model was developed to infer the quantity of RO7445482 in liver and assess the ethnicity effect on plasma and liver exposure using a non-linear mixed-effect approach. All available plasma and urine concentration data were used for modeling. Safety was monitored using standard assessments.
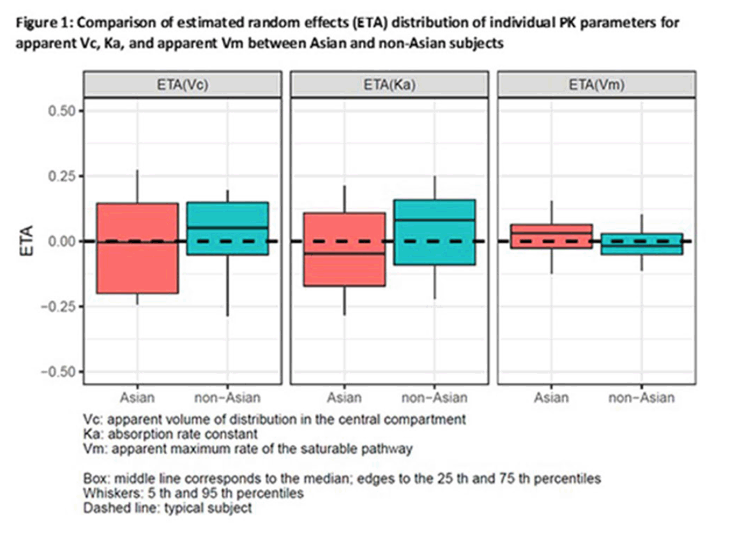
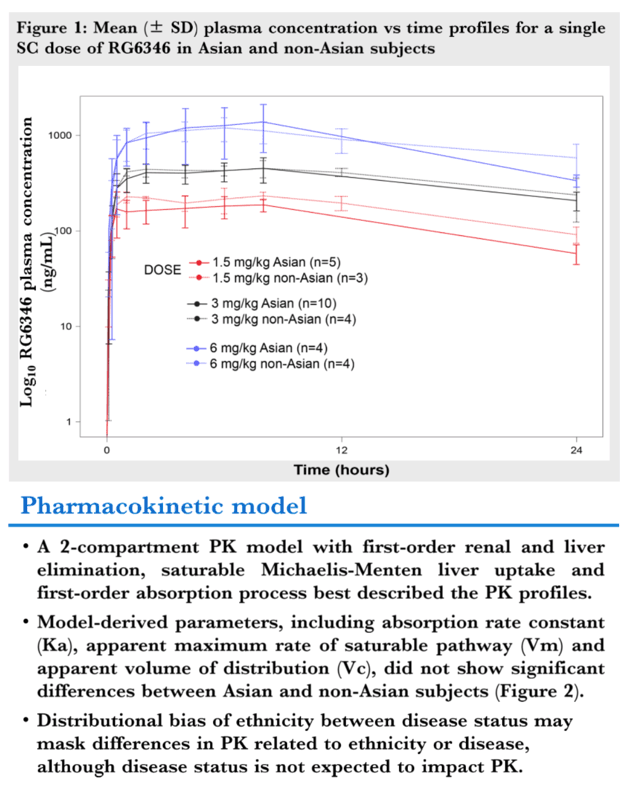
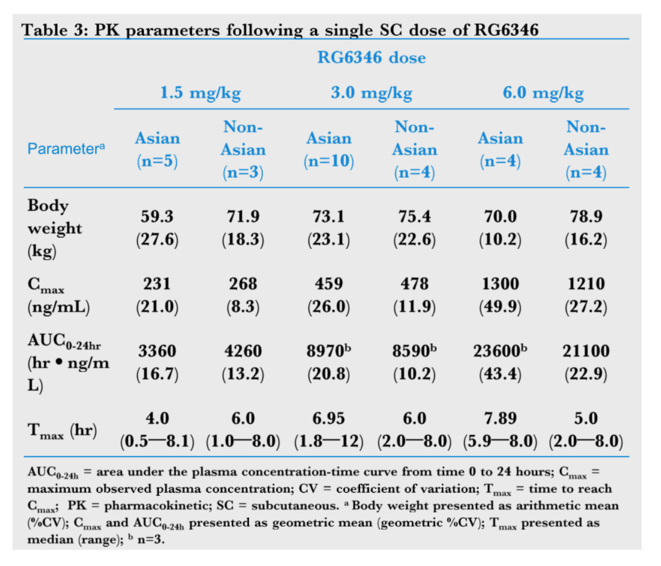
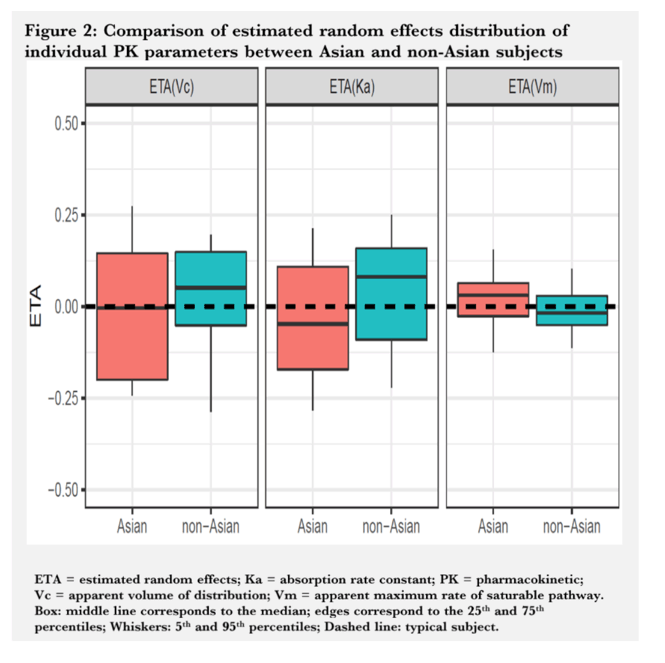
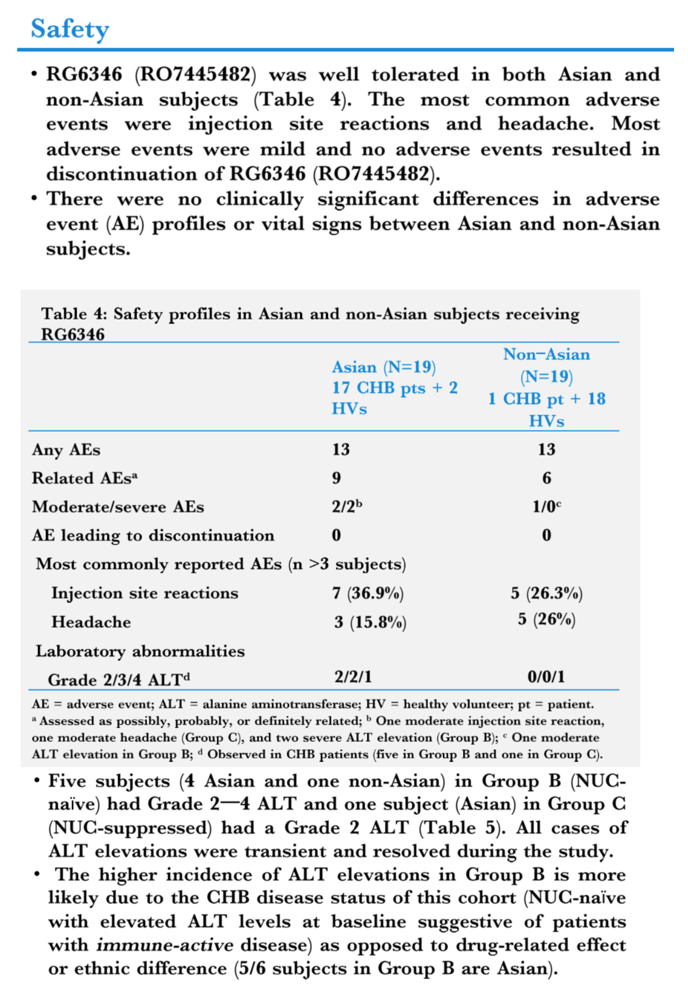
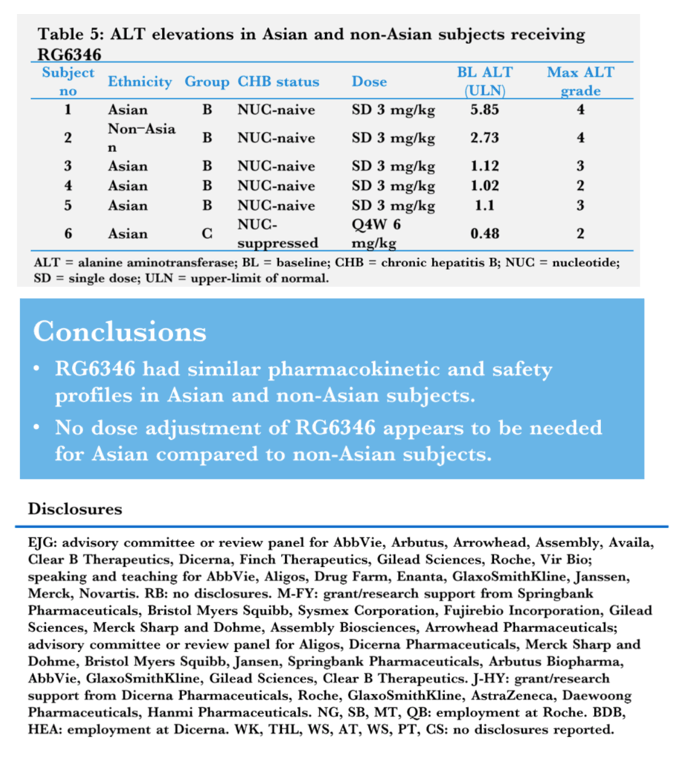
Results: A single SC administration of RO7445482, at the therapeutic dose of 3.0 mg/kg, resulted in similar PK parameter values between the 2 ethnic groups, with geometric mean AUC0-24h of 8970 hr•ng/mL (CV% 20.8) vs. 8590 (CV% 10.2) and geometric mean peak plasma concentration of 459 ng/mL (CV% 26.0) vs. 478 (CV% 11.9) in Asian compared to non-Asian subjects. A 2-compartment PK model with first-order renal and liver elimination, saturable Michaelis-Menten liver uptake and first-order absorption process best described the PK profiles. Model-derived parameters, including Ka and apparent Vm and Vc, did not show significant differences between Asian and non-Asian subjects (Fig. 1). Distributional bias of ethnicity between disease status may mask differences in PK related to ethnicity or disease, although disease status is not expected to impact PK. RO7445482 was well tolerated in both Asian and non-Asian groups. There were no clinically significant differences in adverse event profile, clinical laboratory results or vital signs between the groups.
Conclusion: The current investigation demonstrated similar PK and safety profiles of RO7445482 in Asian and non-Asian subjects. No dose adjustment appears to be needed for Asian compared to non-Asian subjects.



|
| |
|
 |
 |
|
|