 |
 |
 |
| |
HCV CASCADE OF CARE [Big Gaps] AND NEXT STEPS FOR HCV ELIMINATION IN THE UNITED STATES FOLLOWING THE COVID-19 PANDEMIC - new updated improved HCV screening guidelines needed including for pregnant women & high-risk populations
|
| |
| |
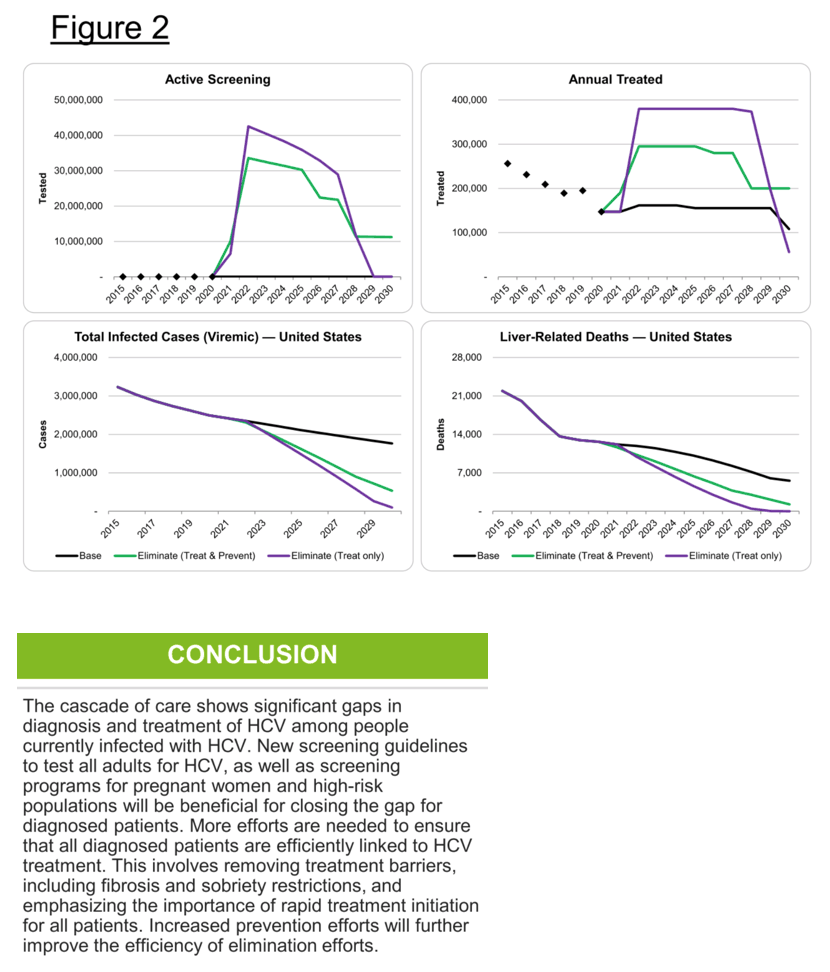
Conclusion: The cascade of care shows significant gaps in diagnosis and treatment of HCV among people currently infected (HCV-RNA+) with HCV. New screening guidelines to test all adults for HCV, as well as screening programs for pregnant women and high-risk populations will be beneficial for closing the gap for diagnosed patients. However, more efforts are needed to ensure that all diagnosed patients are efficiently linked to HCV treatment. This involves removing treatment barriers, including fibrosis and sobriety restrictions, and emphasizing the importance of rapid treatment initiation for all patients.

AASLD 2021 Nov 12-15
Sarah Blach, Chris Estes and Homie Razavi, Center for Disease Analysis Foundation
Background: According to the CDC's 2019 National Progress Report, the United States (US) was on track to achieve three of the five hepatitis C virus (HCV) related targets by the year 2025. However, the COVID-19 pandemic introduced disruptions to health departments, community-based organizations, and healthcare services that have continued into 2021. These disruptions threaten past progress and introduce new challenges, as well as new opportunities, for the coming years. Our objective was to evaluate the HCV prevalence and cascade of care in the US, as well as strategies to achieve national elimination goals by 2025 and 2030.
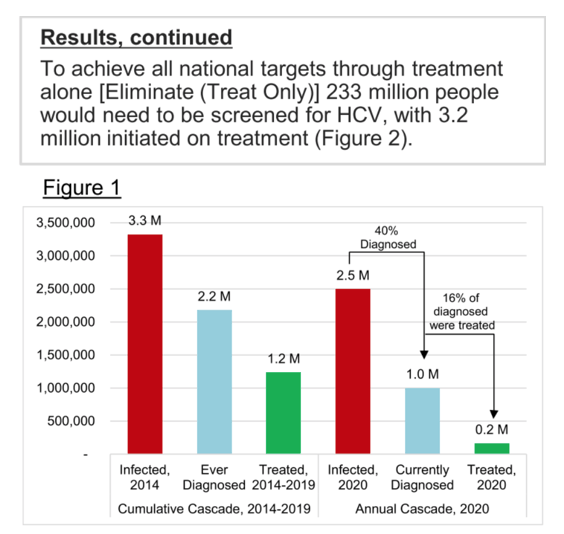
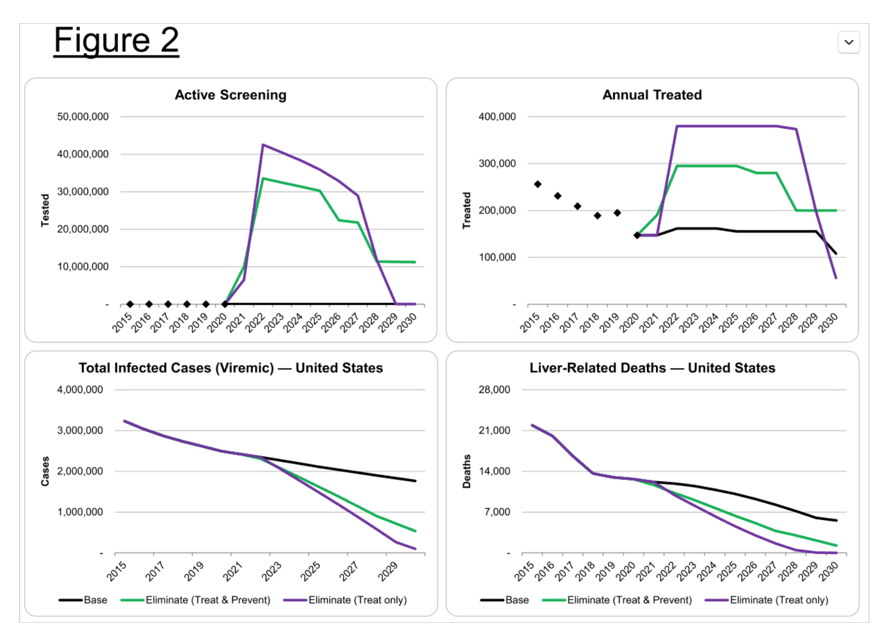
Methods: We updated a previously validated Markov model to estimate HCV-related morbidity and mortality in the US. HCV epidemiological data were based on published data, with annual treatment data from medicine sales data through 2020. Next, a scenario was developed to achieve the national elimination goals of a reduction in acute HCV (20% by 2025; 90% by 2030) and mortality (25%; 65%), as well as an HCV viral clearance rate of 58% by 2025 and 80% by 2030.
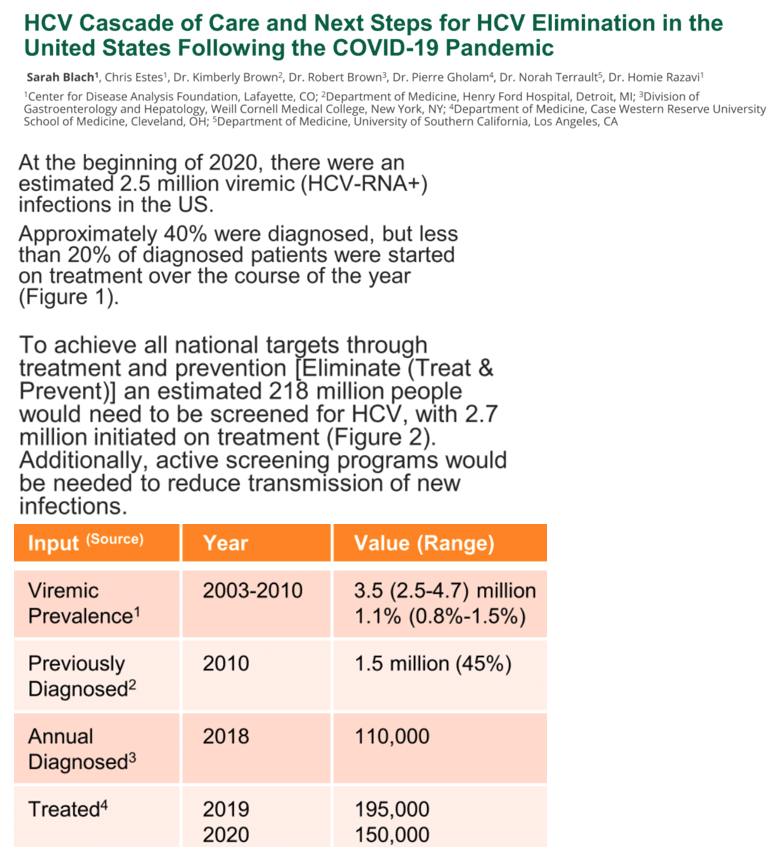
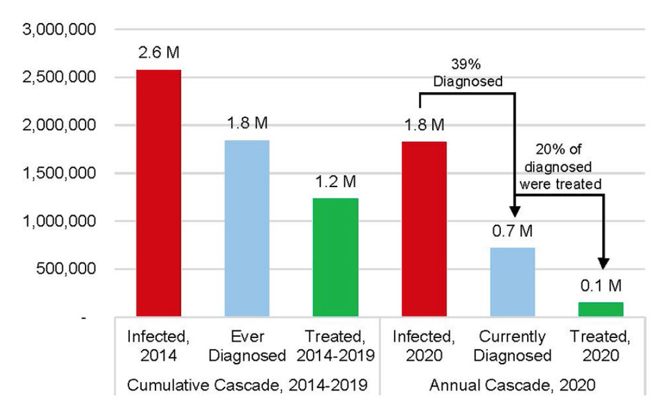
Results: At the beginning of 2020, there were an estimated 1.8 million viremic (HCV-RNA+) infections in the US. Approximately 39% were diagnosed, but only 20% of diagnosed patients were started on treatment over the course of the year. Since the launch of direct acting antiviral (DAA) therapies in the US in 2014, more than 1.2 million patients have been started on treatment. Combined with incident infections and mortality, this has resulted in a net decline in total viremic infections of more than 800,000. To achieve all national targets by 2025 and 2030, 1.1 million people would need to be diagnosed, with 1.4 million people initiated on treatment before 2025.
Conclusion: The cascade of care shows significant gaps in diagnosis and treatment of HCV among people currently infected (HCV-RNA+) with HCV. New screening guidelines to test all adults for HCV, as well as screening programs for pregnant women and high-risk populations will be beneficial for closing the gap for diagnosed patients. However, more efforts are needed to ensure that all diagnosed patients are efficiently linked to HCV treatment. This involves removing treatment barriers, including fibrosis and sobriety restrictions, and emphasizing the importance of rapid treatment initiation for all patients.



|
| |
|
 |
 |
|
|