 |
 |
 |
| |
POPPK MODELING OF Q2M IM RPV LA FOR
MANAGING DOSING INTERRUPTIONS IN HIV-1 PATIENTS
|
| |
| |
CROI 2021 March 6 Reported by Jules Levin
Stefaan Rossenu , Martine Neyens , Rodica Van Solingen-Ristea , Bryan Baugh ,Herta Crauwels1
Janssen, Beerse, Belgium, Janssen Pharmaceutical,Research and Development, Titusville, FL
Background: Long-acting rilpivirine (RPV LA) is intended for coadministration with cabotegravir long-acting (CAB LA) as a complete 2-drug injectable regimen for HIV-1 treatment. Eight-weekly RPV LA plus CAB LA was noninferior to 4-weekly RPV LA plus CAB LA in maintaining HIV-1 suppression (ATLAS-2M; Overton TE et al. CROI 2020 Abstract 34).
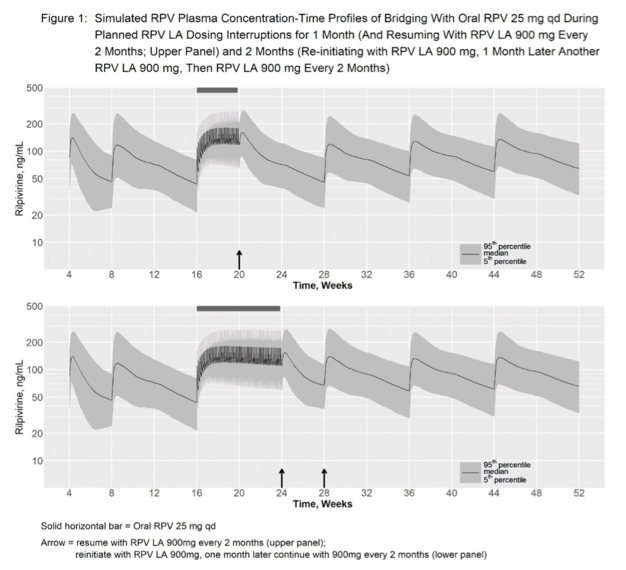
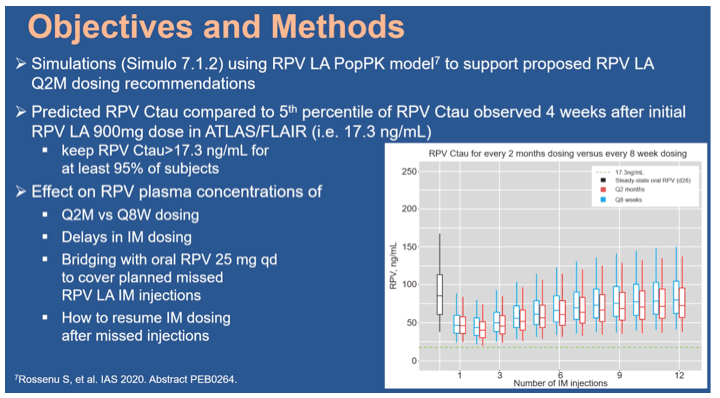
Methods: Every 2 months (Q2M) RPV LA consists of 1-month oral RPV 25mg once daily for tolerability assessment, two initiation RPV LA 900mg (3mL) intramuscular (IM) doses separated by 1 month and subsequent 900mg doses Q2M, to be administered with the Q2M CAB regimen. Population pharmacokinetic (PopPK) modeling and simulation was used to inform strategies for managing dosing interruptions, aimed at minimizing impact on the overall RPV LA PK profile. Simulations included effects on RPV concentrations of Q2M vs 8-weekly dosing, IM dosing delays, and bridging with oral RPV to cover a planned missed IM injection. Simulated RPV concentrations were compared to the 5th percentile of observed RPV concentrations 4 weeks after the initial RPV LA 900mg dose in ATLAS/FLAIR and to concentrations observed in the oral RPV development program, as done for monthly RPV LA (Rossenu S et al. AIDS 2020 PEB0264).
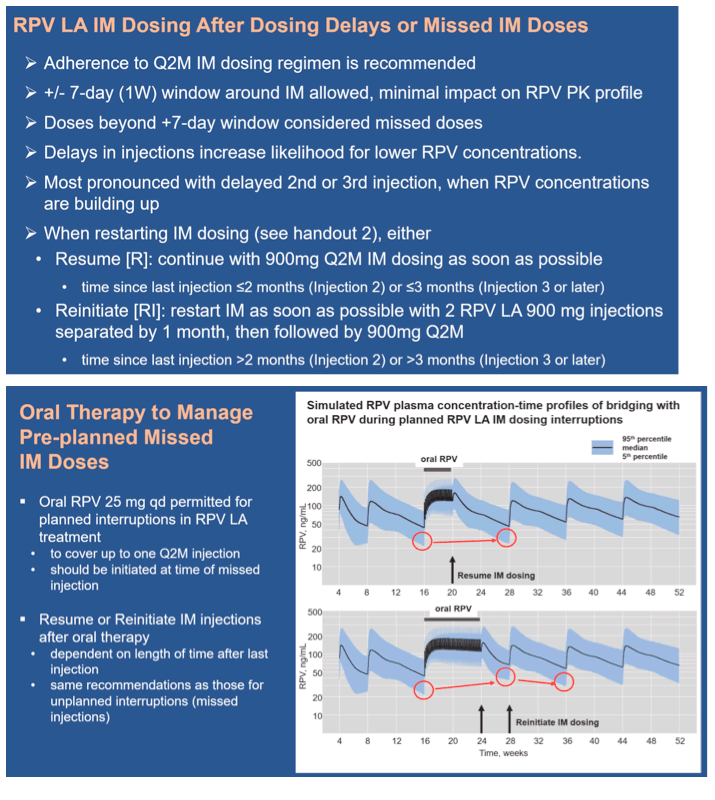
Results: Q2M vs 8-weekly dosing, with a 7-day window, has minimal impact on overall RPV PK profile. IM dosing delays of >7 days may have a larger impact, particularly in the first few months of therapy. If a patient plans to miss a scheduled injection by >7 days, oral RPV can provide coverage of up to 1 missed injection (Figure 1). Recommendations for resuming RPV LA after missed injections are: If time since last injection: - ≤2 months (Injection 2) or ≤3 months (Injection 3 or later): continue with 900mg dose (3mL) as soon as possible, then continue Q2M dosing. - >2 months (Injection 2) or >3 months (Injection 3 or later): re-initiate with 900mg dose (3mL) followed by a second 900mg dose (3mL) one month later, then continue Q2M dosing.
Conclusion: Adherence to the Q2M RPV LA injection schedule is strongly recommended. Oral therapy to cover planned dosing interruptions of RPV LA injections can provide exposures within ranges observed in clinical studies. Recommendations for managing dosing interruptions are aligned for RPV LA and CAB LA, to facilitate dosing for the complete regimen.

|
| |
|
 |
 |
|
|