 |
 |
 |
| |
LONG-ACTING HIV CAPSID INHIBITOR EFFECTIVE
AS PrEP IN A SHIV RHESUS MACAQUE MODEL
|
| |
| |
CROI 2021 March 6 Reported by Jules Levin
Elena Bekerman , Samuel J. Vidal , Derek Hansen , Bing Lu , Kelly Wang , Abishek
Chandrashekar , Jim Zheng , William Rowe , Darryl Kato , Christian Callebaut , Wade Blair, Tomas Cihlar , Stephen Yant , Romas Geleziunas , Dan Barouch2
1Gilead Sciences, Inc, Foster City, CA, USA, 2Beth Israel Deaconess Medical Center, Boston, MA, USA
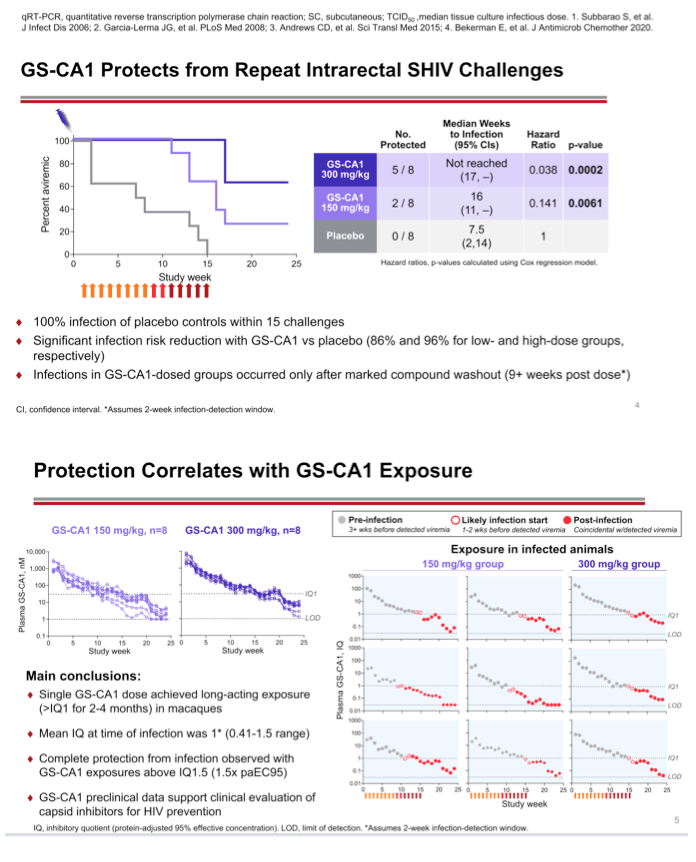
Background: The ability of daily oral preexposure prophylaxis (PrEP) to effectively reduce the risk of contracting HIV is strongly dependent on high medication adherence, which is not uniformly achieved among people at risk for HIV. Long-acting agents can circumvent the need for daily dosing while providing long-term protection. Lenacapavir (LEN), an investigational small molecule inhibitor of HIV capsid function with picomolar antiviral potency and long-acting subcutaneous (s.c.) dosing potential (twice yearly), is in clinical development for HIV treatment. Here, we evaluated GS-CA1, a LEN analog active against both HIV and SIV, as a long-acting PrEP agent in a macaque rectal SHIV challenge model.
Methods: Eight naïve Indian rhesus macaques per group received a single s.c. injection of either vehicle control (placebo), 150 mg/kg (low dose) or 300 mg/kg (high dose) GS-CA1, followed by weekly escalating titer SHIV challenges starting 1-week post-dosing. Blood was collected weekly for the evaluation of plasma drug levels, viral loads, and serology. Animals were considered protected if they remained SHIV-negative by plasma PCR assay and seronegative by enzyme immunoassay throughout the 15-week challenge phase and the 10-week follow-up.
Results: Following a single injection, plasma concentrations of GS-CA1 exceeded its serum protein binding adjusted (pa) EC95 value (30.1 nM) for 8-15 weeks and 15-17 weeks in the low and high dose groups, respectively. After a total of 15 challenges 8/8 animals became infected in the placebo group, whereas 2/8 and 5/8 animals remained protected in the low and high GS-CA1 dose groups, respectively. The median time-to-infection was 7.5 weeks in the placebo group, 16 weeks in the low-dose GS-CA1 group, and not reached due to insufficient number of infections in the high dose group. Relative to the placebo group, the low and high dose treatment groups demonstrated an 86% (p=0.0061) and 96% (p=0.0002) infection risk reduction, respectively, as determined by Cox regression analysis. Notably, based on a 2-week infection- to-detection window, treatment group infections occurred only after plasma GS-CA1 concentrations fell below 2X paEC95.
Conclusion: These preclinical data provide a proof of concept for the prophylactic efficacy of a long-acting capsid inhibitor in a nonhuman primate SHIV challenge model and support the clinical development of LEN for HIV prevention.


|
| |
|
 |
 |
|
|