 |
 |
 |
| |
SWITCHING TO DTG/3TC FDC IS NONINFERIOR TO TBR FOR 96 WEEKS: TANGO SUBGROUP ANALYSES
|
| |
| |
CROI 2021 March 6 Reported by Jules Levin
Paul Benson1, Clifford A. Kinder2, María Jesús Pérez-Elías3, Don E. Smith4, Stefan H. Scholten5, Mounir Ait-Khaled6, Keith A. Pappa7, Ruolan Wang7, Jonathan Wright8, Brian Wynne7, Michael Aboud6, Jean A. Van Wyk6, Kimberly Smith7
1Be Well Medical Center, Berkley, MI, USA, 2AIDS Healthcare Foundation - The Kinder Medical Group, Miami, FL, USA, 3Hospital Universitario Ramón y Cajal, Madrid, Spain, 4Albion Centre, Sydney, Australia, 5Praxis Hohenstaufenring, Cologne, Germany, 6ViiV Healthcare, Brentford, UK, 7ViiV Healthcare, Research Triangle Park, NC, USA, 8GlaxoSmithKline, Uxbridge, UK
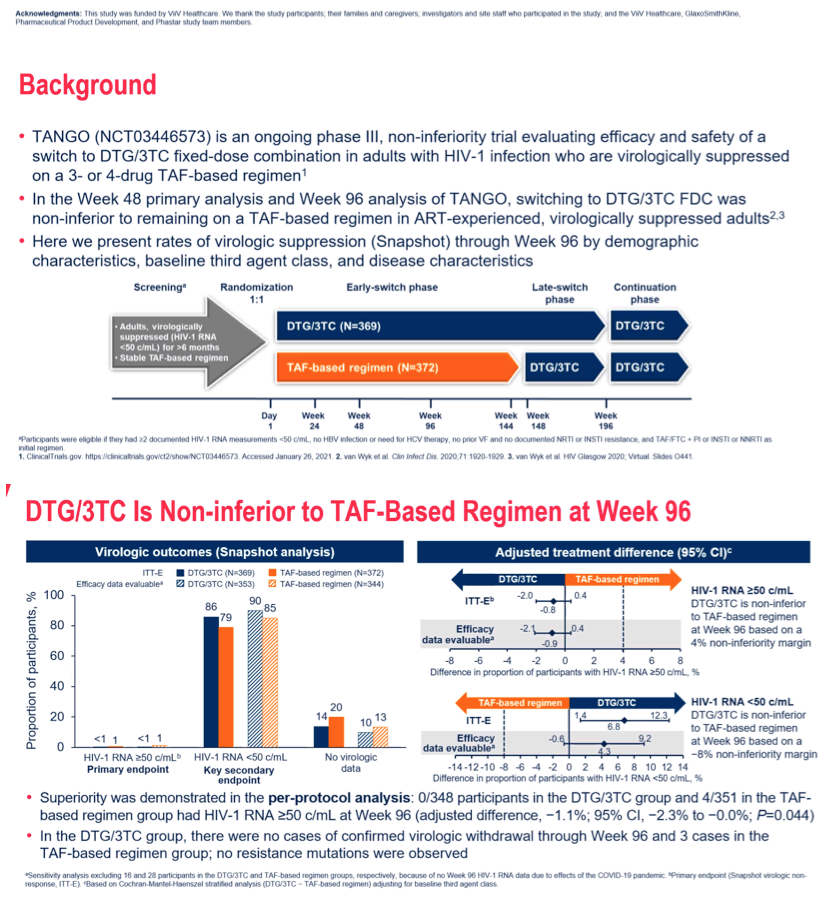
Background: The 2-drug regimen (2DR) of DTG/3TC reduces the number of antiretroviral agents taken by individuals treated for HIV-1 infection, when compared to traditional 3DRs. DTG/3TC is non-inferior to DTG+TDF/FTC in HIV-1 infected ART-naïve adults (GEMINI) through Week 144 and in ART-experienced, virologically suppressed participants switching from a TAF-based 3/4DR (TANGO) through Week 96. Here we present a key Week 96 secondary endpoint from the TANGO study: Snapshot virologic success by baseline regimen third agent class, disease and demographic characteristics.
Methods: TANGO is a randomized, open-label, multicenter, non-inferiority Phase III study evaluating the efficacy and safety of switching to DTG/3TC once daily versus remaining on a current TAF-based regimen in. HIV-1 infected adults, with HIV-1 RNA<50c/mL for >6 months, on a TAF-based regimen for at least 3 months and without prior virologic failure or historical NRTI or InSTI major resistance mutations, were eligible to participate. Randomization was stratified by baseline 3rd agent class: PI, NNRTI, InSTI. The primary endpoint was the proportion of participants with plasma HIV-1 RNA≥50c/mL at Week 48 (FDA Snapshot algorithm, Intention To Treat-Exposed [ITT-E] population) with secondary analyses at Week 96.
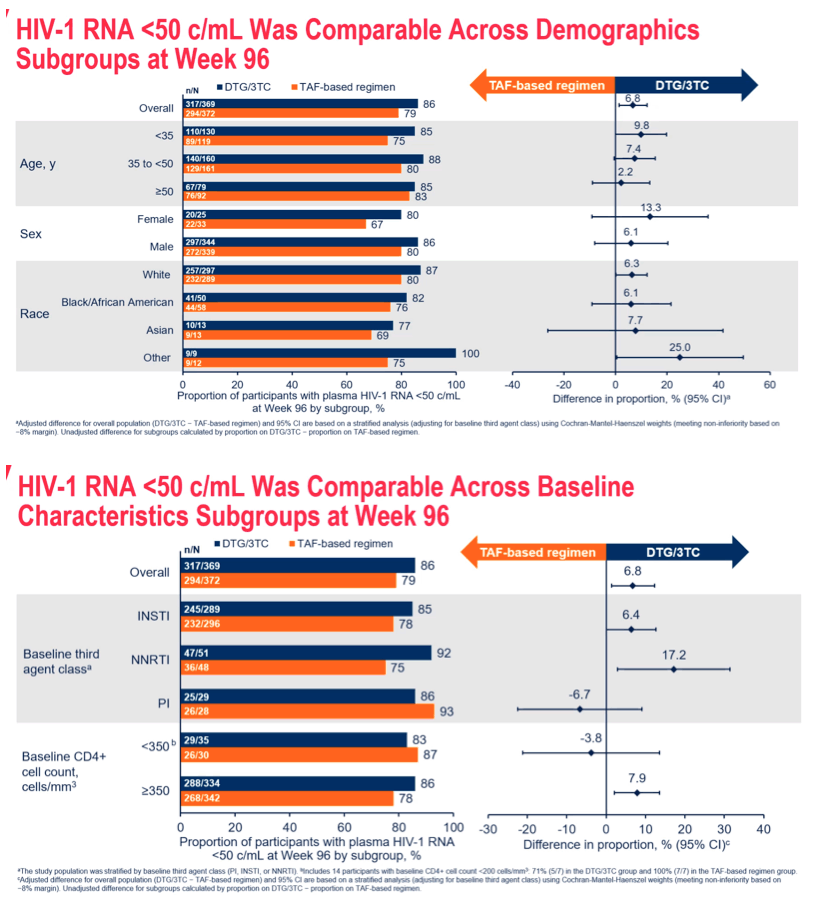
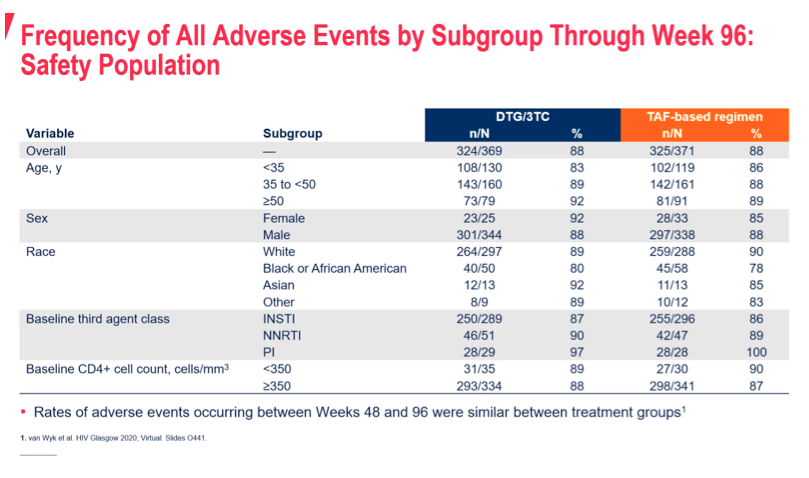
Results: 741 randomized/exposed participants (DTG/3TC: 369; TBR: 372) were included. Snapshot success rates across subgroups were generally consistent with the overall TANGO Week 96 study results and were similar between arms (Figure). Zero participants on DTG/3TC and 3 participants (<1%) on TBR met confirmed virologic withdrawal criteria with no resistance mutations observed at failure.
Conclusion: Switching to DTG/3TC FDC was non-inferior to continuing a TAF-based 3DR in maintaining virologic suppression in HIV-1 infected ART- experienced adults through Week 96. Efficacy by subgroups was consistent with overall Week 96 study results, demonstrating that switching from TAF-based regimens to DTG/3TC is effective at maintaining virologic suppression regardless of baseline regimen, patient or disease characteristics.

|
| |
|
 |
 |
|
|