 |
 |
 |
| |
WEEK 96 ANALYSIS OF VIRAL BLIPS FROM A PHASE 2B TRIAL OF ISLATRAVIR AND DORAVIRINE
|
| |
| |
CROI 2021 March 6 Reported by Jules Levin
Chloe Orkin1, Jean-Michel Molina2, Yazdan Yazdanpanah3, Carolina Chahin Anania4, Joseph J. Eron5, Stephanie O. Klopfer6, Karen A. Eves6, Deborah A. Hepler6, Carey Hwang6, Todd A. Correll6 1Queen Mary University of London, London, UK, 2University of Paris Diderot, Paris, France, 3Hopital Bichat Claude Bernard, Paris, France, 4Hospital Hernan Henríquez Aravena de Temuco, Temuco, Chile, 5University of North Carolina at Chapel Hill, Chapel Hill, NC, USA, 6Merck & Co, Inc, Kenilworth, NJ, USA
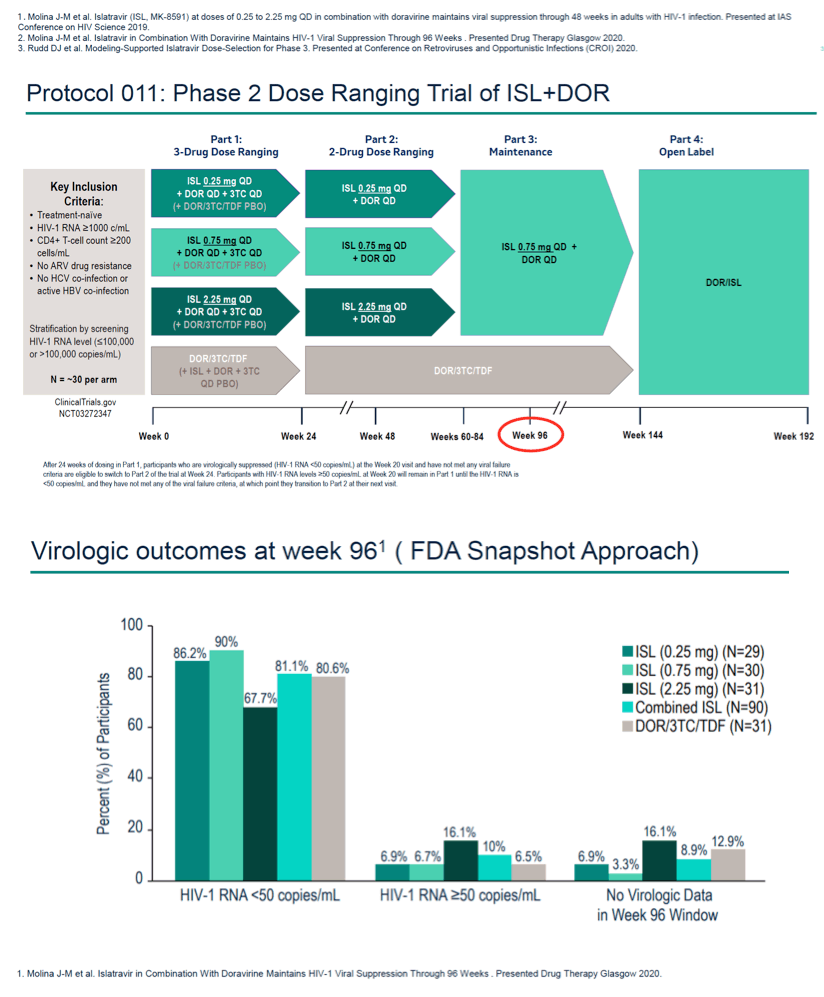
Background: Islatravir (ISL, MK-8591) is the first nucleoside reverse transcriptase translocation inhibitor (NRTTI) in development for the treatment and prevention of HIV-1 infection. Previously we showed that ISL+DOR demonstrated efficacy in maintaining viral suppression and was well tolerated through week 96 in a Phase 2b trial. Rates of protocol defined virologic failure (PDVF) were low across all groups. Here we report on an analysis of blip frequency--a sensitivity marker for efficacy.
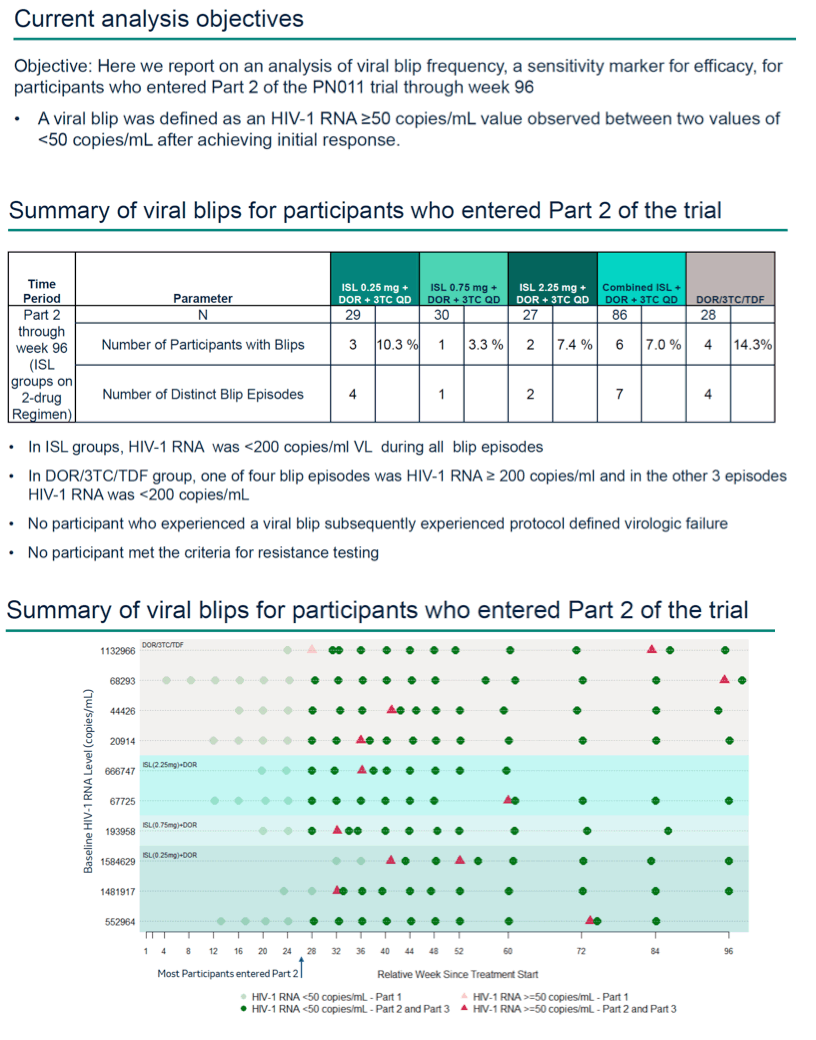
Methods: In a Phase 2b trial in treatment-naïve adults with HIV-1, participants were randomized to receive ISL (0.25, 0.75 or 2.25 mg) + DOR (100 mg) and lamivudine (3TC, 300 mg) QD, or a fixed-dose combination of DOR, 3TC and tenofovir disoproxil fumarate (DOR/3TC/TDF). Participants receiving ISL achieving HIV-1 RNA <50 copies/mL at week 20 or later stopped 3TC at the next study visit to transition to the two-drug regimen for Part 2 of the trial. For this current analysis we analyzed viral blip frequency for participants who entered Part 2 of the trial through week 96. A viral blip was defined as an HIV-1 RNA ≥50 copies/mL value observed between two values of <50 copies/mL after achieving initial response.
Results: 114 participants entered Part 2 of the trial and were included in the analysis. During Part 2 of the trial through week 96, a higher percentage of participants on the three-drug regimen in the DOR/3TC/TDF group experienced viral blips as compared to participants on the two-drug regimen in the combined ISL groups; 4 blip episodes occurred in 4 out of 28 participants (14.3%) in the DOR/3TC/TDF group as compared to 8 blip episodes in 7 of 86 participants (8.1%) in the combined ISL groups (Table 1). Of the participants with viral blips, 5 of 7 participants in the combined ISL groups and 0 out of 4 participants in the DOR/3TC/TDF group had baseline HIV-1 RNA >100,000 c/mL. All participants with viral blips, including those with high baseline HIV-1 RNA levels, re- suppressed by the next study visit and remained suppressed through week 96. None of the participants with viral blips had subsequent viral rebound or PDVF.
Conclusion: Viral blips were relatively rare for all treatment groups and were not associated with a loss of virologic suppression or PDVF.


|
| |
|
 |
 |
|
|