 |
 |
 |
| |
CASIRIVIMAB WITH IMDEVIMAB ANTIBODY COCKTAIL FOR COVID-19 PREVENTION: INTERIM RESULTS
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Meagan P . O'Brien1, Eduardo Forleo Neto1, Kuo-Chen Chen1, Flonza Isa1, Ingeborg Heirman1, Neena Sarkar1, Divya Ramesh1, Myron S. Cohen2, Ruanne Barnabas3, Christopher B. Hurt2, Dan H. Barouch4, Katharine J. Bar5, Gary Herman1, George D. Yancopoulos1, David M. Weinreich1
1Regeneron Pharmaceuticals, Inc., Tarrytown, NY, USA, 2University of North Carolina at Chapel Hill, Chapel Hill, NC, USA, 3University of Washington, Seattle, WA, USA, 4Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA, 5University of Pennsylvania, Philadelphia, PA, USA
Background: Passive immunization has a long history for infection prevention following exposure. We report results of a descriptive interim analysis from
a study of an antibody "cocktail" of casirivimab with imdevimab (cas/imdev; formerly REGN-COV2) designed to bind non-competing epitopes of the viral spike protein, as a potential passive vaccine for the prevention of COVID-19 in people at risk of infection from household contact.
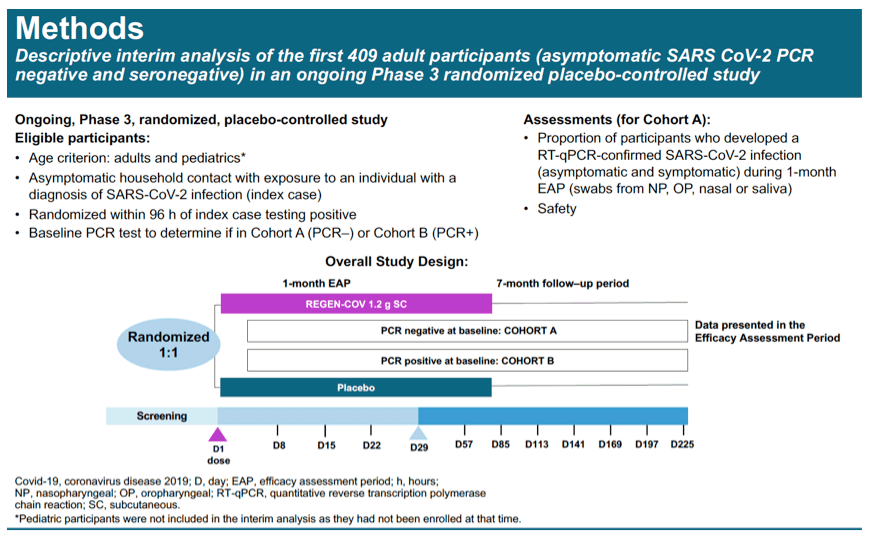
Methods: In this ongoing Phase 3 study, asymptomatic participants exposed to a COVID-19-infected household member were randomized 1:1 to placebo or 1200 mg cas/imdev (600 mg of each antibody administered subcutaneously) within 96 hours of their household member testing positive. The analysis included participants who tested negative for SARS-CoV-2 by nasal, saliva, or nasopharyngeal swab and who were seronegative to SARS-CoV-2 antibodies at baseline. The proportion of participants who developed an RT-PCR-confirmed SARS-CoV-2 infection (asymptomatic or symptomatic) during the 1-month efficacy assessment period was summarized.
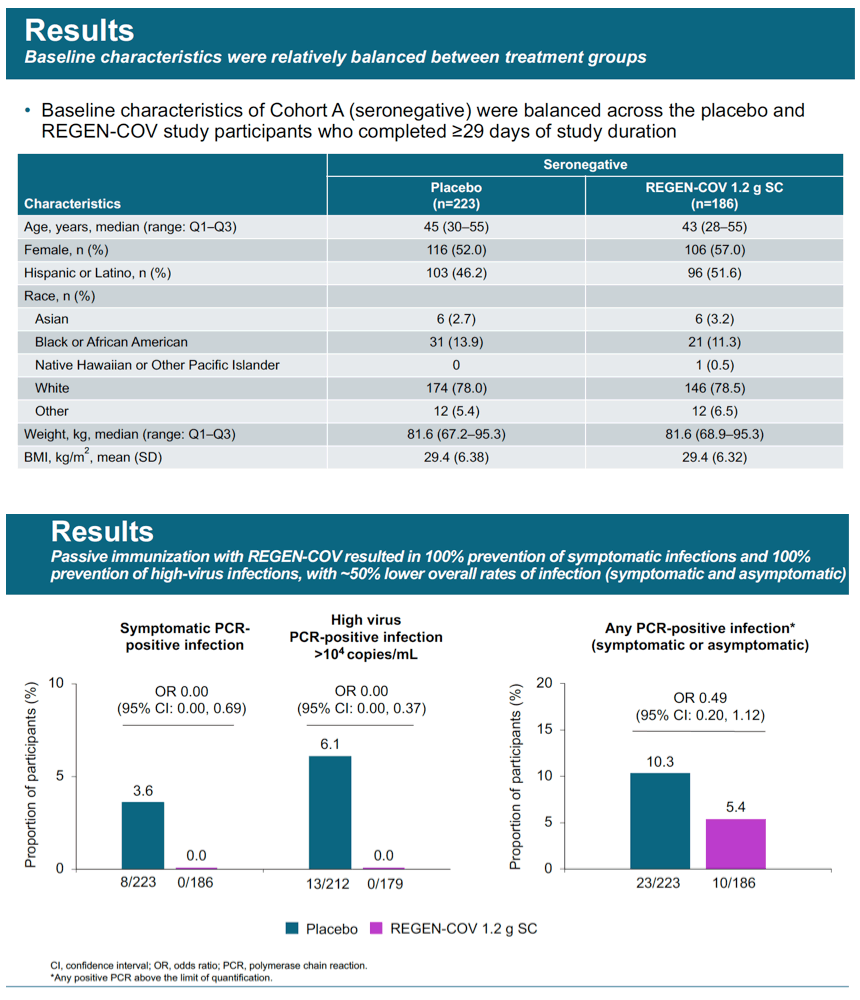
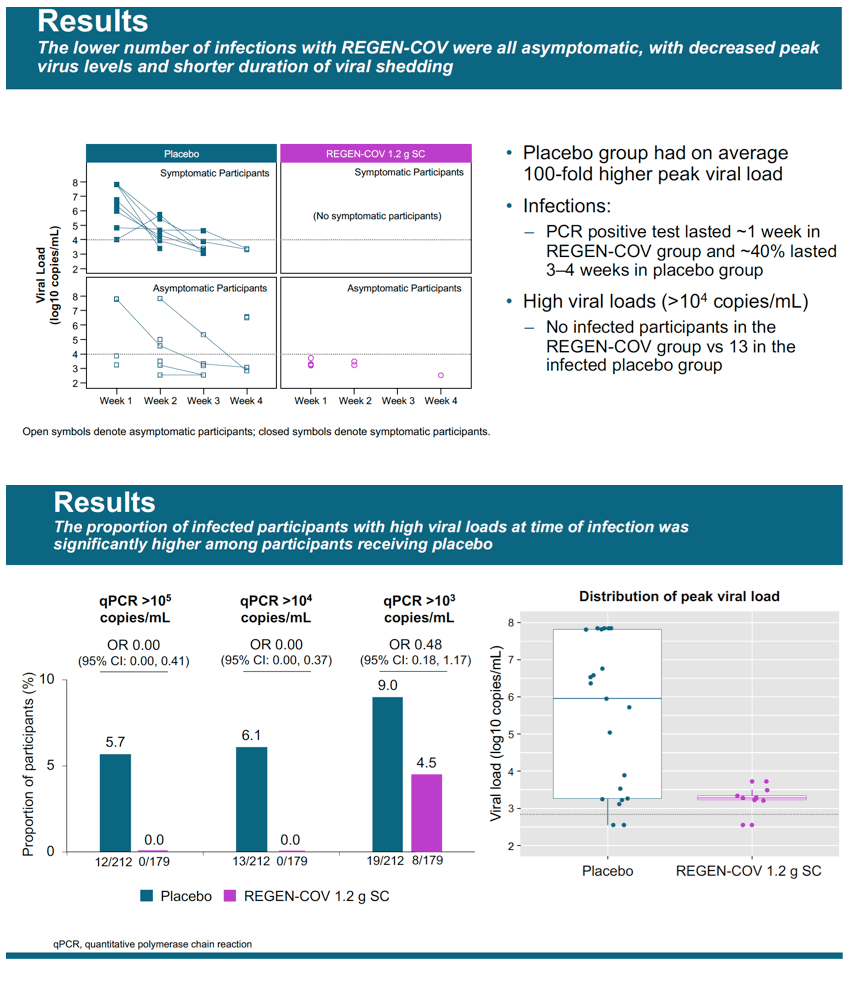
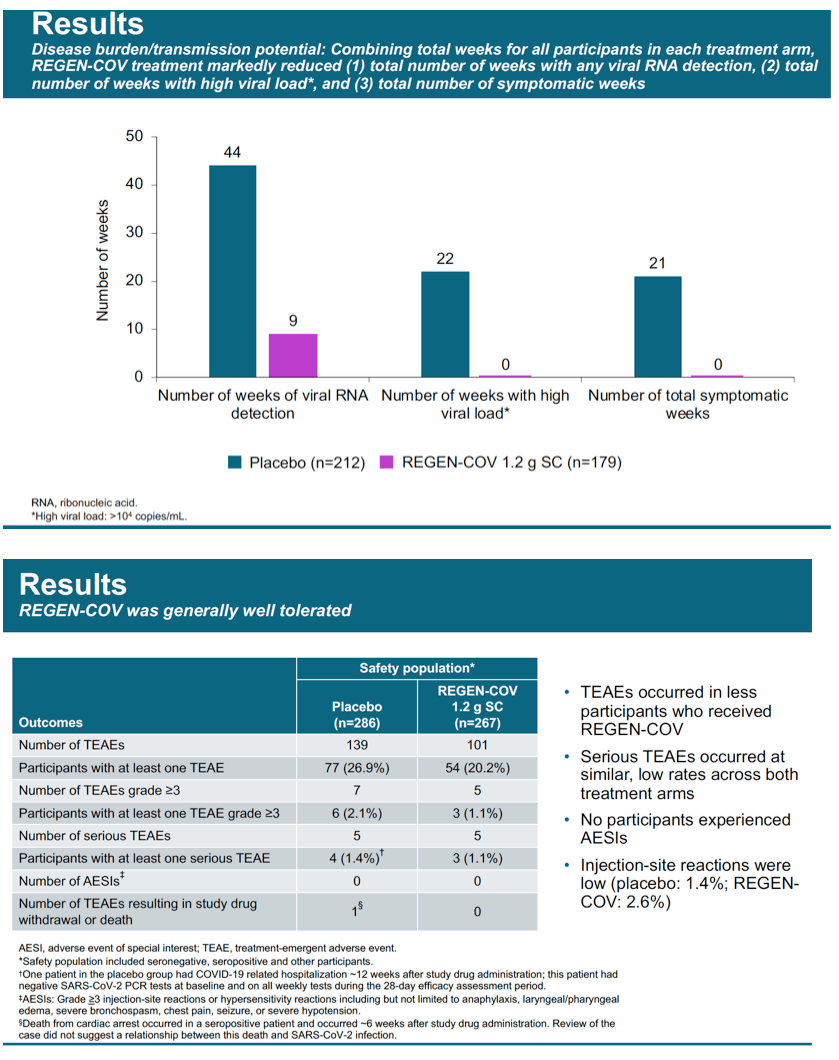
Results: Initial results from the first evaluable 223 placebo and 186 cas/imdev participants who completed ≥29 days of the study are reported. Reduction in PCR-positive symptomatic disease was 100% (0/186 cas/imdev vs 8/223 placebo; OR 0.00 [CI 0.00, 0.69]). Reduction in any PCR-positive infection (symptomatic or asymptomatic) was 48% (10/186 vs 23/223; OR 0.49 [CI 0.20, 1.12]). Placebo-group participants had on average 100-fold higher peak viral load. In the cas/imdev group, viral RNA was not detected for longer than 1 week but was detected for 3-4 weeks in approximately 40% of placebo participants (Fig. 1). The proportions of infected participants with high viral loads (>10^4 copies/mL) were 13/21 placebo vs 0/9 cas/imdev. Total weeks of viral RNA detection and high viral load were 44 and 22 weeks in the placebo group vs 9 and 0 in the cas/imdev group. Total symptomatic weeks were 21 for placebo vs 0 for cas/imdev. A similar proportion of participants experienced at least 1 serious adverse event: placebo, 3/222 and cas/imdev, 1/186; none were deemed related to study treatment. Injection site reactions were similar: placebo, 1.4%; cas/ imdev, 2.6%.
Conclusion: In this descriptive interim analysis of participants at risk of SARS- CoV-2 infection from household transmission, a subcutaneous dose of the cas/ imdev antibody cocktail prevented symptomatic infection, reduced overall infection, and decreased viral load and duration of viral RNA detection.



|
| |
|
 |
 |
|
|