 |
 |
 |
| |
HIV/HCV COINFECTION TRENDS IN SPAIN (2015-2019)
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Chiara Fanciulli1, Juan Berenguer1, Carmen Busca Arenzana2, Maria Jesús Vivancos3, Maria Jesús Tellez4, Lourdes Dominguez5, Pere Domingo6, Jordi Navarro7, Jesús Santos8, José A. Iribarren9, Luis Morano10, Marta De Miguel11, Inmaculada Jarrin12, Juan González-Garcia13, for the GeSIDA 8514 Study Group 1Hospital General Universitario Gregorio Marañón, Madrid, Spain, 2Hospital La Paz Institute for Health Research, Madrid, Spain, 3Hospital Ramón y Cajal, Madrid, Spain, 4Hospital Clinico San Carlos, Madrid, Spain, 5Hospital Universitario 12 de Octubre, Madrid, Spain, 6Hospital Sant Pau, Barcelona, Spain, 7Hospital Universitario de la Vall d’Hebron, Barcelona, Spain, 8Hospital Universitario Virgen de la Victoria, Malaga, Spain, 9Hospital Donostia, San Sebastián, Spain, 10Hospital Universitario Alvaro Cunqueiro, Vigo, Spain, 11Fundación SEIMC-GeSIDA, Madrid, Spain, 12Instituto de Salud Carlos III, Madrid, Spain, 13La Paz University Hospital, Madrid, Spain
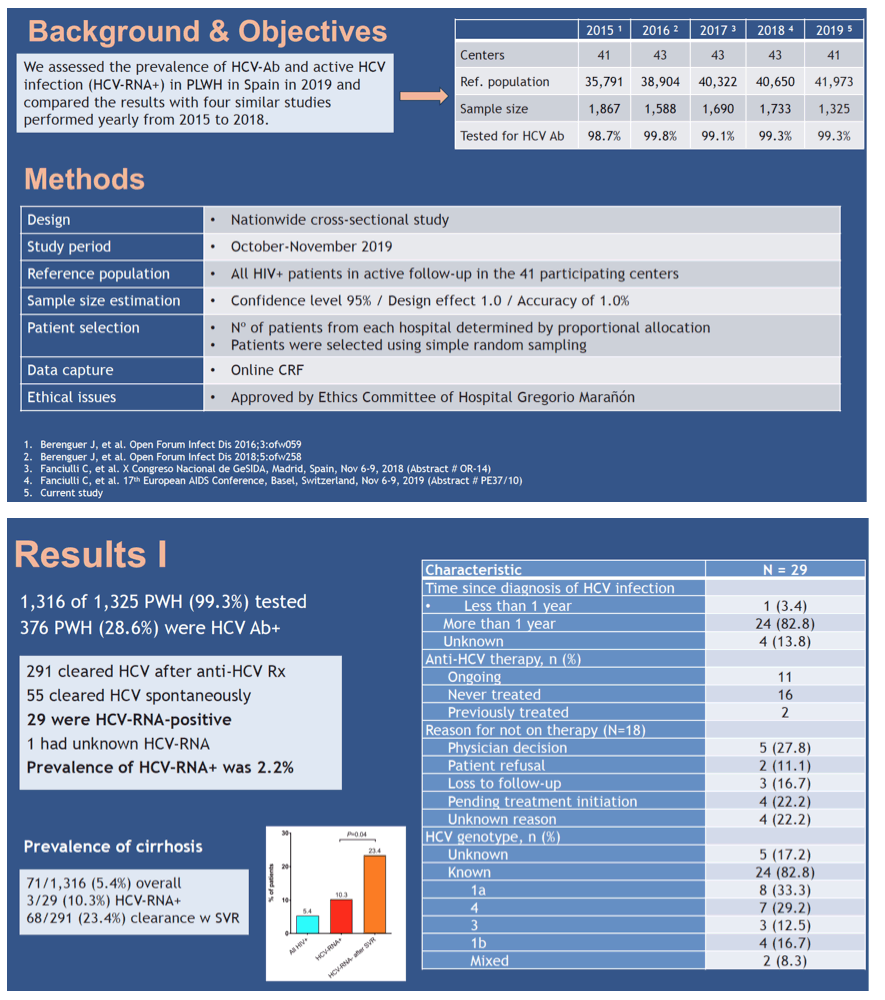
Background: We assessed the prevalence of anti-HCV antibodies (HCV-Ab) and active HCV infection (HCV-RNA-positive) in people living with HIV (PLWH) in Spain in 2019 and compared the results with four similar studies performed yearly from 2015 to 2018.
Methods: The study was performed in 41 centers (October-November, 2019). The sample size was estimated for an accuracy of 1.0%, the number of patients from each hospital was determined by proportional allocation, and patients were selected using simple random sampling. All oral DAA-based therapy has been available in Spain since the third trimester of 2014. Since June 2017, free access to treatment has been available to all HCV-infected individuals.
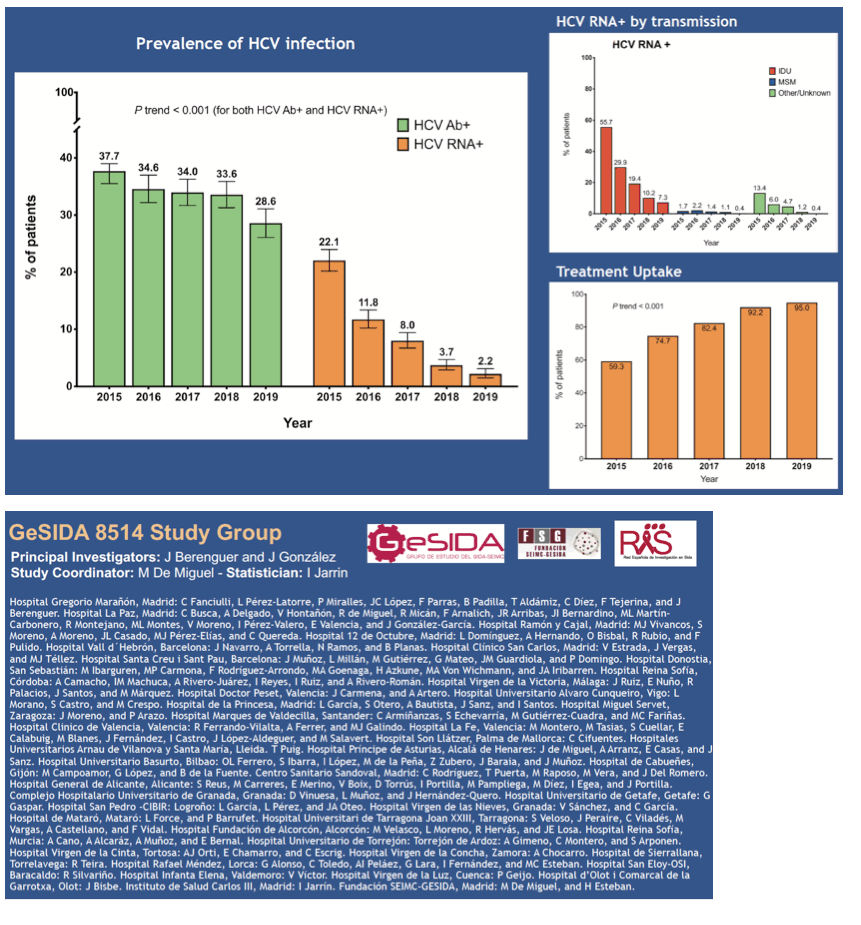
Results: The reference population comprised 41,973 PLWH, and the sample size was 1,325. HCV serostatus was known in 1,316 (99.3%), and 376 (28.6%) were HCV Ab-positive (78.7% PWID and 7.7% MSM). Of the 376 HCV Ab- positive patients, 291 cleared HCV after anti-HCV therapy, 55 cleared HCV spontaneously, 29 were HCV-RNA-positive, and 1 had unknown HCV-RNA. The prevalence of HCV-RNA-positive was, therefore, 2.2%. As 11 of 29 patients were receiving DAAs, and assuming treatment effectiveness of 95%, the HCV-RNA- positive prevalence could be considered to be 1.4%. Reasons for not receiving anti-HCV therapy in 18 patients included physician decision (N=5), loss to follow-up (N=3), patient refusal (N=2), and unknown reasons (N=4). Of the 29 HCV-RNA-positive patients, the infection was chronic in 24, acute/recent in 1, and unknown duration in 4. HCV-related liver cirrhosis was present in 71 (5.4%) PLWH overall, 3 (10.3%) HCV-RNA-positives, and 68 (23.4%) of those who cleared HCV after anti-HCV therapy (P=.04). The prevalence of HCV Ab decreased steadily from 37.7% in 2015 to 28.6% in 2019 (P <.001). Likewise, HCV-RNA prevalence decreased from 22.1% in 2015 to 2.2% in 2019 (P <.001). Anti-HCV treatment uptake increased from 53.9% in 2015 to 95.0% in 2019 (P <.001) (Table).
Conclusion: Active HCV infection among PLWH in Spain at the end of 2019 was 2.2%, that is, 90.0% lower than in 2015, meaning that elimination of HCV infection among PLWH in Spain is an achievable goal shortly. Increased exposure to DAAs was likely the main reason for this sharp decrease. Despite the high coverage and effectiveness of DAA-based treatment, HCV-related cirrhosis among those successfully treated for HCV remains significant in this population.

|
| |
|
 |
 |
|
|