 |
 |
 |
| |
NEURON DAMAGE AND RESERVOIR ARE SECONDARY TO
HIV TRANSCRIPTS DESPITE SUPPRESSIVE ART
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Kazuo Suzuki1, John Zaunders1, Angelique Levert1, Shannen Butterly1, Zhixin Liu2, Takaomi Ishida3, Chin-Shiou Huang4, Thomas Gates1, Caroline Rae5, Lauriane Jugé5, Lucette A. Cysique1, Bruce Brew1
1St Vincent's Hospital, Sydney, Australia, 2University of New South Wales, Randwick, Australia, 3DENKA Life Innovation Research Institute, Tokyo, Japan, 4PlexBio Research Development, Taipei, Taiwan, 5Neuroscience Research Australia, Randwick, Australia
these findings explain HAND in the context of viral suppression as current cART does not stop viral components being made. The obvious example is tat - it is not just tat but the other components that are being produced eg nef vpr and env......should flip our thinking re comorbidities as being confounds to one where they are just another manifestation of HIV viral components eg tat nef etc causing damage..... lifestyle issues including accelerated ageing etc. are relevant, but I think HIV per se is more important.
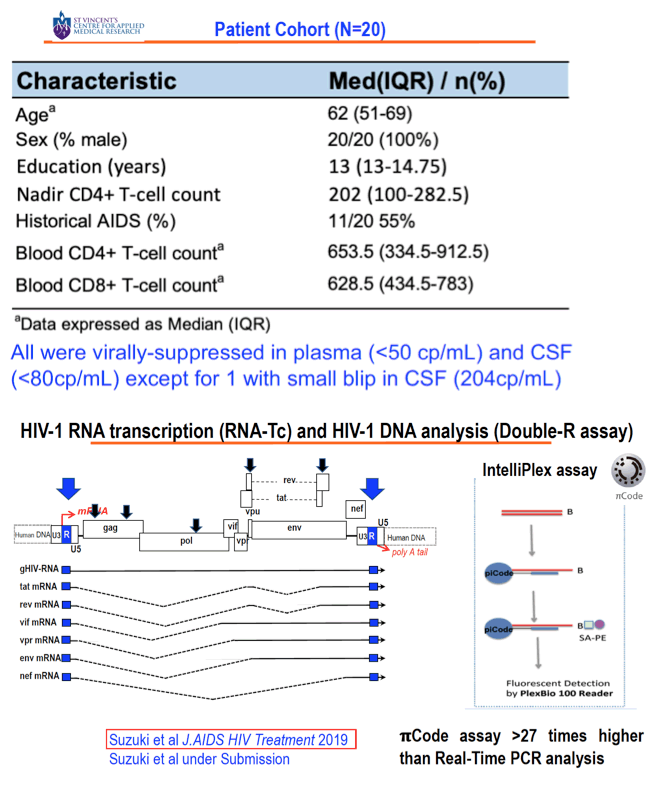
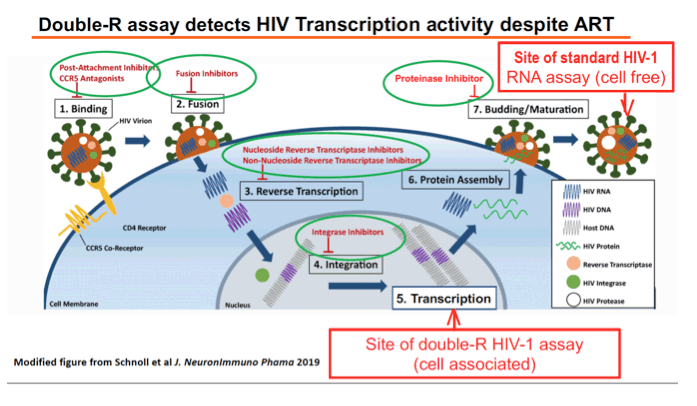
Background: HAND persists despite suppressive cART for reasons that are unclear. We have previously shown that transcription without whole virus may explain HAND and contribute to the CSF HIV reservoir. Here we confirm and extend these findings with double the number of patients using our highly sensitive Double-R assay of HIV RNA/DNA and flow cytometry for cellular origin.
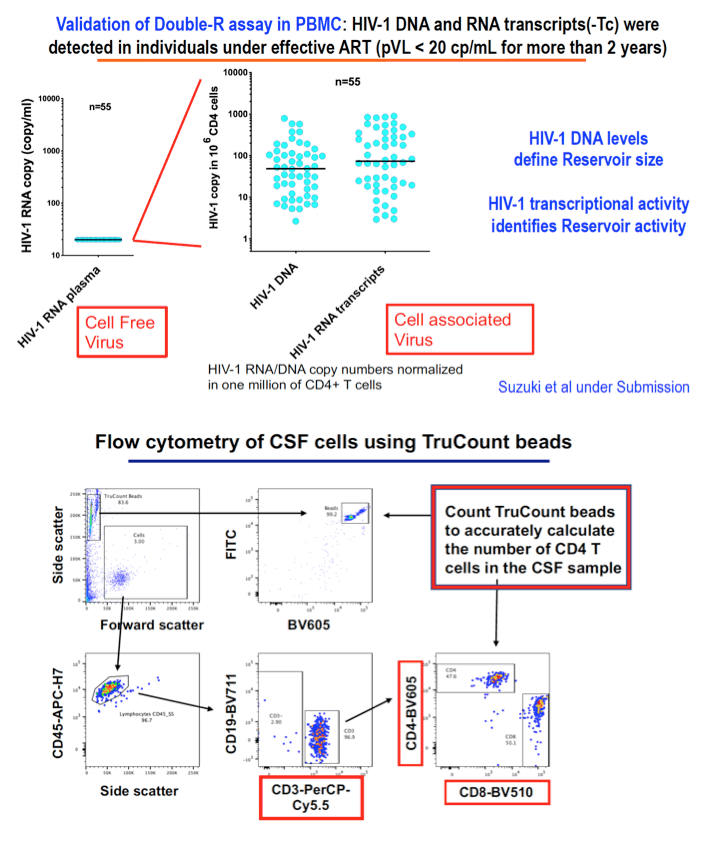
Methods: DNA and RNA were extracted from cells in 20 paired samples of CSF and blood taken from HIV+ subjects (4 with current HAND and 2 with past HAND) on cART with both plasma and CSF HIV RNA (Roche) <50 copies/ml. HIV-1 transcripts and DNA levels were determined by the previously described Double-R assay based on the extremely sensitive πCode MicroDiscs platform. Immunological profiles of CSF cells and PBMC were compared by 18-colour flow cytometry. In vivo brain injury was assessed with MR spectroscopy in the frontal white matter (FWM), posterior cingulate cortex (PCC), and caudate area.
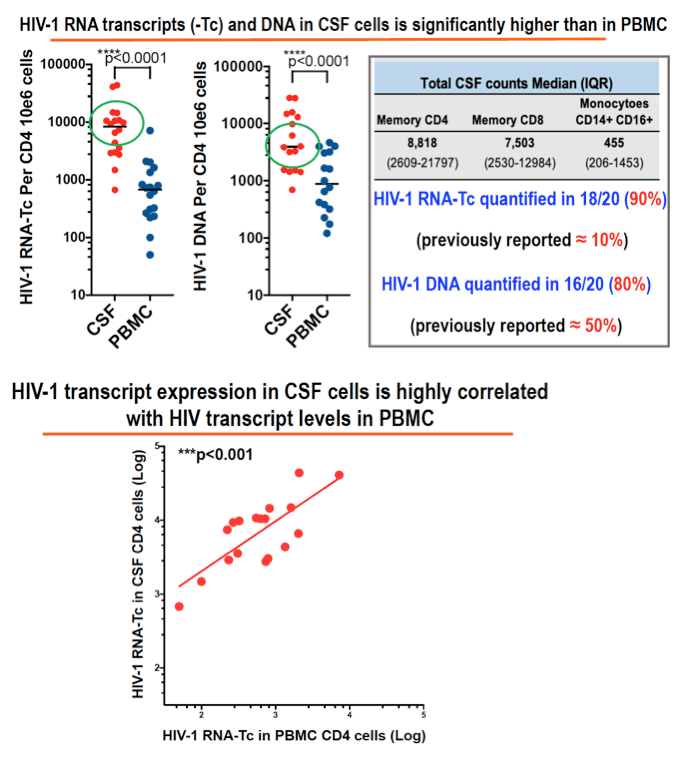
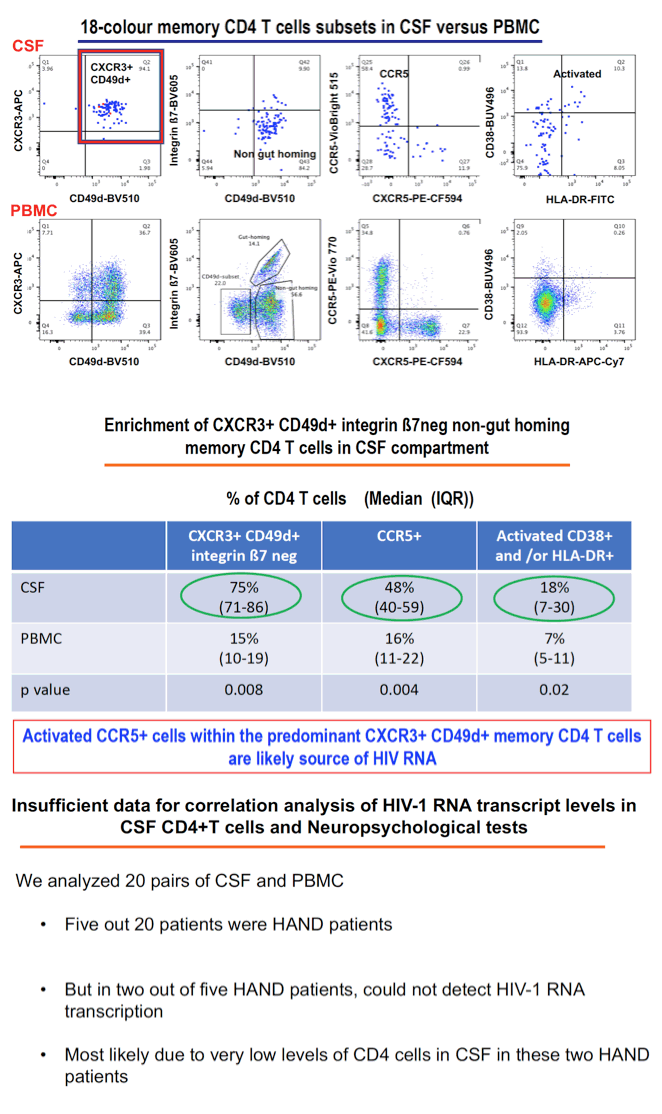
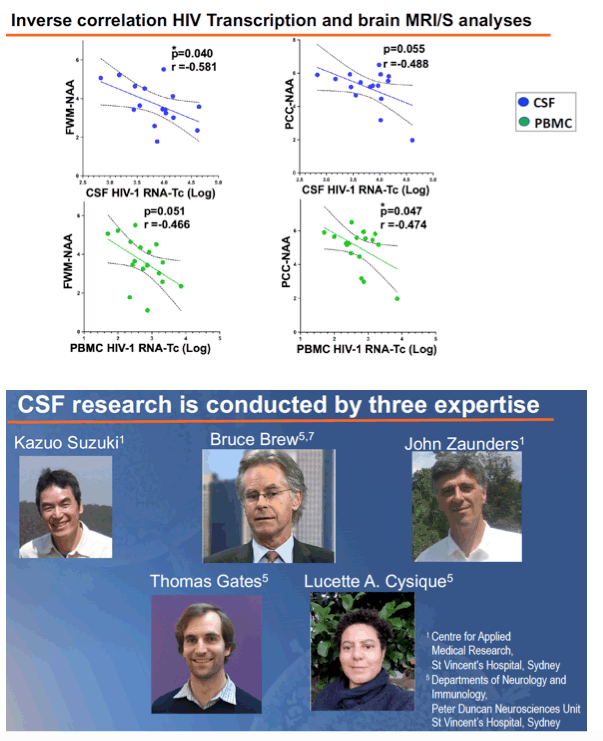
Results: 18/20 patients' CSF CD4+T cells had significantly higher levels of cell- associated HIV-1 RNA transcriptional activity vs PBMCs (median 8,331 copies/106 CD4+T cells, vs 680; p<0.0001), with significant correlation of transcriptional activity within CD4+T cells between CSF and PBMC (r=0.46; p=0.029). 16/20 patients also had significantly higher HIV-1 DNA levels in CSF CD4+T cells (median 3,940 copies/106 cells vs 885; p<0.0001). CSF transcriptional activity was inversely correlated with the neuronal integrity biomarker N-acetyl aspartate (NAA) in FWM (p=0.04) and PCC (p=0.055). Transcriptional activity in PBMCs showed similar results: NAA in FWM (p=0.051) and in PCC (p=0.047). CSF cells were 91% memory T cells, with roughly equal memory CD4 (median 3,605 cells) and CD8 T cells (3,507 cells). Other CSF cells were 3.1% CD14+CD16+ monocytes, 2.0% NK cells and 0.4 % B cells. CXCR3+ CD49d+ integrinß7- cells were 76% of CSF CD4 T cells compared with 17% of CD4 in PBMC; 51% were CCR5+ (vs 16% in PBMC); and 18% expressed CD38 and/or HLA-DR activation markers (vs 11% in PBMC).
Conclusion: CSF is an HIV reservoir with high transcription activity despite ART. It is biologically significant because of compromised neuron integrity likely mediated by transcription products (tat). The cellular source of HIV RNA is most likely the predominant CXCR3+ CD49d+ integrinß7- CCR5+ memory CD4+T cells; monocytes may be less important. Therapies targeting transcription should be developed.


|
| |
|
 |
 |
|
|