 |
 |
 |
| |
ACTIVATING PKC-ε INDUCES HIV EXPRESSION WITH IMPROVED TOLERABILITY
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Alivelu M . Irrinki1, Jasmine Kaur1, Bally Randhawa1, Hongxia Li1, Ryan McFadden1, Chelsea Snyder1, Hoa Truong1, Daniel Soohoo1, Chad Greco1, Eric Hu1, Helen Yu1, Bernard Murray1, Wade Blair1, Tomas Cihlar1, Jeffrey Murry1
1Gilead Sciences, Inc, Foster City, CA, USA
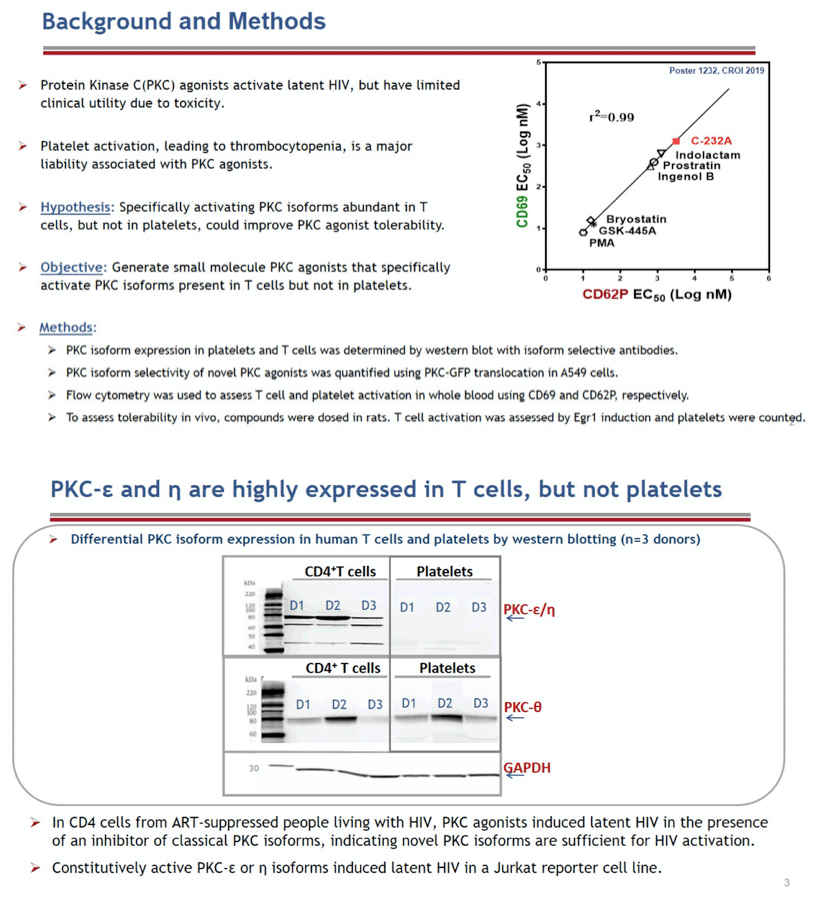
Background: Activation of latent HIV could increase elimination of infected cells by therapeutics that directly target infected cells or stimulate antiviral immunity, potentially leading to long-term remission or cure. Protein kinase C (PKC) agonists are highly effective at activating latent HIV in vitro. We previously found that platelet activation is associated with toxicity of novel PKC agonist C-232A and that this critical safety liability is broadly associated with known classes of PKC agonists. We hypothesized that specific targeting of PKC isoforms abundant in T cells but not in platelets could improve tolerability of PKC agonists that activate HIV.
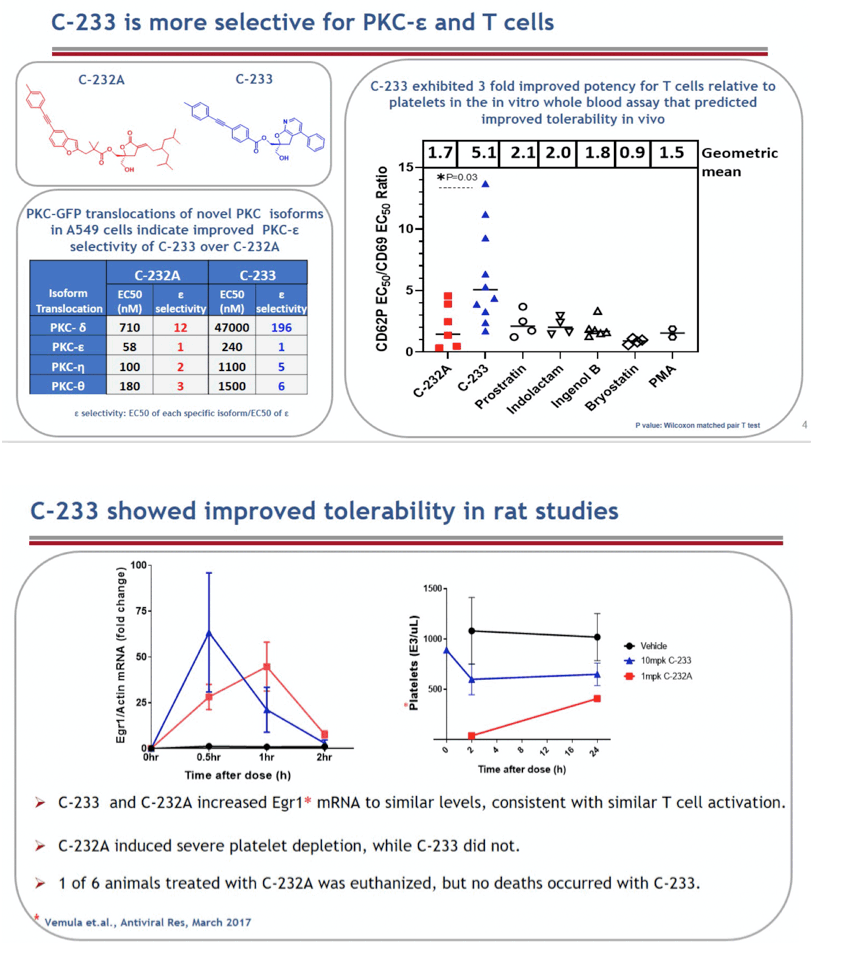
Methods: PKC isoform expression in T cells and platelets was measured by western blot to identify differentially expressed isoforms. Isoform-selective agonists were identified by testing compounds for individual PKC isoform translocation. HIV RNA was assessed after treatment in cells isolated from ART- suppressed people living with HIV. In latently infected Jurkat cells, HIV induction was assessed by flow cytometry after expression of constitutively active PKC isoforms. In vivo platelet activation was assessed by hematology counts and T cell activation by EGR1 and CD69 mRNA quantification from whole blood in rats. Results: Prostratin induction of HIV was unaffected by Gö6976, an inhibitor of classical PKC isoforms (PKC-α/β/γ), indicating that novel PKC isoforms (PKC-δ/ε/ η/Θ) are sufficient for HIV activation. Expression analysis revealed high levels of PKC-Θ and δ in platelets and T cells while PKC-η and ε were only abundant in T cells. In Jurkat cells, PKC-ε was sufficient for HIV activation, supporting selective targeting of PKC-ε to reduce toxicity. Systematic modification of C-232A led to the identification of C-233, a novel PKC agonist 2-fold and 10-fold more selective for PKC-ε over PKC-Θ and PKC-δ, respectively. In rats, C-233 and C-232A increased Egr1 mRNA associated with T cell activation to similar levels, but C-232A affected platelet levels more severely than C-233. One of 6 animals treated with C-232A was euthanized, but no deaths occurred with C-233.
Conclusion: Platelet activation is a critical safety liability associated with non-selective PKC agonists and should be carefully monitored in preclinical and clinical studies. These results indicate that specifically activating PKC-ε might improve safety in vivo and support continuing structure-based design of selective novel PKC agonists for safe activation of HIV reservoirs.

|
| |
|
 |
 |
|
|