 |
 |
 |
| |
HIV WITH TRANSMITTED DRUG RESISTANCE
IS DURABLY SUPPRESSED BY B/F/TAF AT WEEK 144
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Rima K . Acosta , Grace Q. Chen , Silvia Chang , Ross Martin , Xinxin Wang , Hailin Huang , Diana Brainard , Jason Hindman , Sean E. Collins , Hal Martin , Kirsten L. White1
1Gilead Sciences, Inc, Foster City, CA, USA
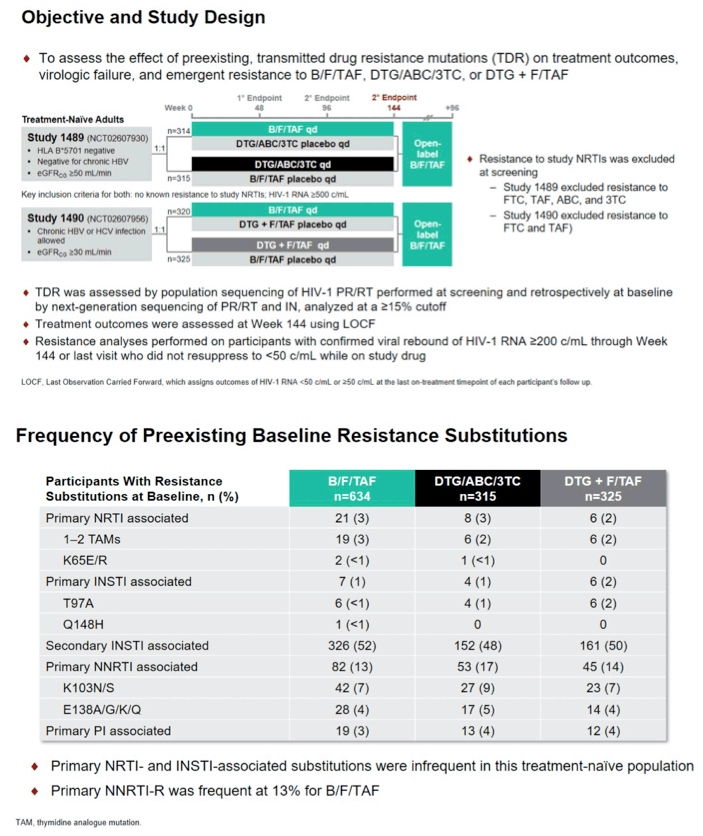
Background: Two phase 3, randomized, double-blind, active-controlled studies of initial HIV-1 treatment demonstrated that bictegravir/emtricitabine/ tenofovir alafenamide (B/F/TAF) was non-inferior to dolutegravir/abacavir/ lamivudine (DTG/ABC/3TC, Study 1489) or to DTG+F/TAF (Study 1490) through 144 weeks. In both studies, there was no emergent resistance to study drugs. Here, we describe the effect of baseline transmitted drug resistance (TDR) on treatment response over 3 years.
Methods: Population sequencing of HIV-1 protease and reverse transcriptase (RT) was performed at screening; resistance to study nucleos(t)ide reverse transcriptase inhibitors (NRTIs) was excluded. Retrospective baseline next generation sequencing of protease, RT, and integrase (IN) was analyzed at a ≥15% cutoff. Treatment outcomes were assessed at Week 144 using last on-treatment observation carried forward (LOCF). Resistance analyses were performed on participants with confirmed viral rebound of HIV-1 RNA ≥200 copies/mL through Week 144 or last visit who did not resuppress to <50 copies/ mL while on study drug.
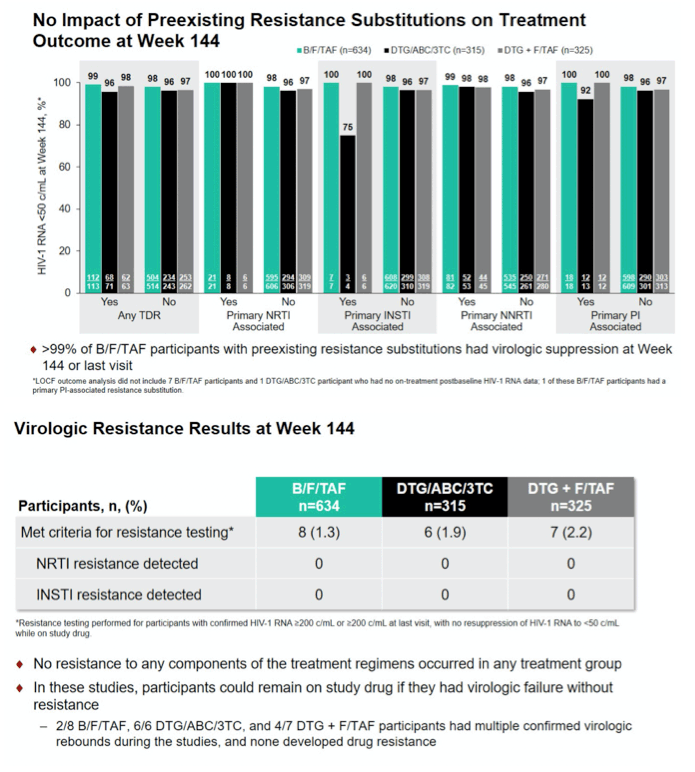
Results: Of 1421 PLWH screened for both studies, only 3 (0.2%) were excluded due to TDR to FTC, TAF, ABC, or 3TC. TDR was present in 19.5% (248/1274) of enrolled participants and consisted of InSTI resistance (-R) in 1.3% (17/1270 with data), NRTI-R in 2.7% (35/1274), NNRTI-R in 14.1% (179/1274), and PI-R in 3.5% (44/1274). Treatment outcomes by LOCF at Week 144 of participants with or without TDR were comparable (98% of those with primary TDR had HIV RNA <50 copies/mL vs. 97% of those without TDR) (Table), indicating that preexisting TDR did not affect treatment outcomes. One participant had preexisting Q148H+G140S in IN and K70R and K103N in RT at baseline. This participant was randomized to B/F/TAF, had HIV-1 RNA <50 copies/mL at Week 4, and maintained HIV-1 RNA <50 copies/mL through Week 144. In total, 21 participants qualified for post-baseline resistance testing (1.3% [8/634] B/F/ TAF; 1.9% [6/315] DTG/ABC/3TC; 2.2% [7/325] DTG+F/TAF); of those, 2/8 B/F/ TAF, 6/6 DTG/ABC/3TC, and 4/7 DTG+F/TAF participants had multiple confirmed virologic rebounds during the studies. No participant had emergent resistance to study drugs.
Conclusion: Initial HIV-1 treatment with B/F/TAF, DTG/ABC/3TC, or DTG+F/ TAF achieved high, durable rates of virologic suppression. The presence of TDR did not affect treatment outcomes, and there was no treatment-emergent resistance through 144 weeks.

|
| |
|
 |
 |
|
|