 |
 |
 |
| |
INTEGRASE INHIBITORS TARGET MITOCHONDRIA IN
BROWN ADIPOCYTES DISRUPTING THERMOGENESIS
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Ikrak Jung1, Sunghee Jin1, Becky Tu-Sekine1, Fredrick Anokye-Danso1, Todd Brown1, Sangwon F. Kim1
1Johns Hopkins University School of Medicine, Baltimore, MD, USA
Background: Antiretroviral therapy (ART) containing integrase strand transfer inhibitors (InSTI) has been associated with weight gain in both ART-initiation and switch studies, especially in women, but the underlying mechanisms are unclear. Estrogen promotes brown adipocyte differentiation while suppressing white adipose differentiation. Hence, we hypothesized that InSTIs may interrupt adipose function via inhibition of estrogen action.
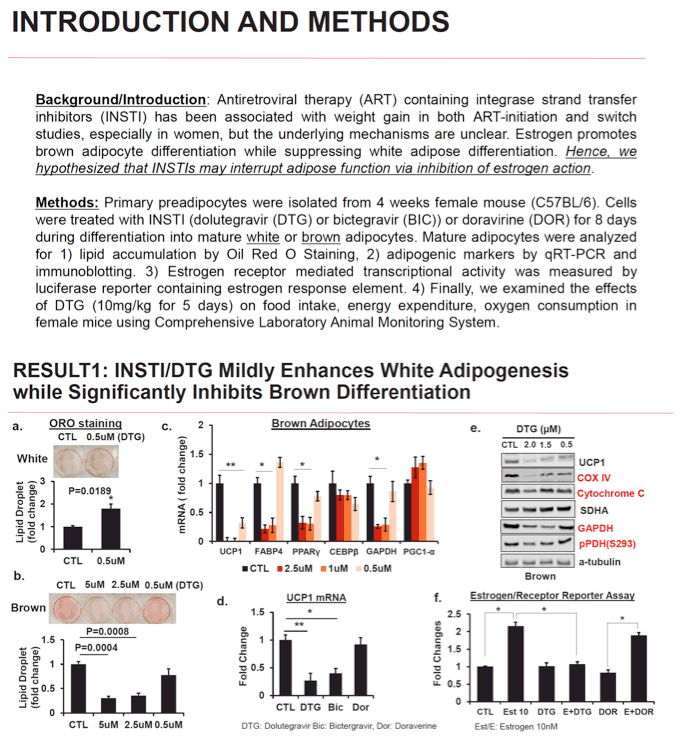
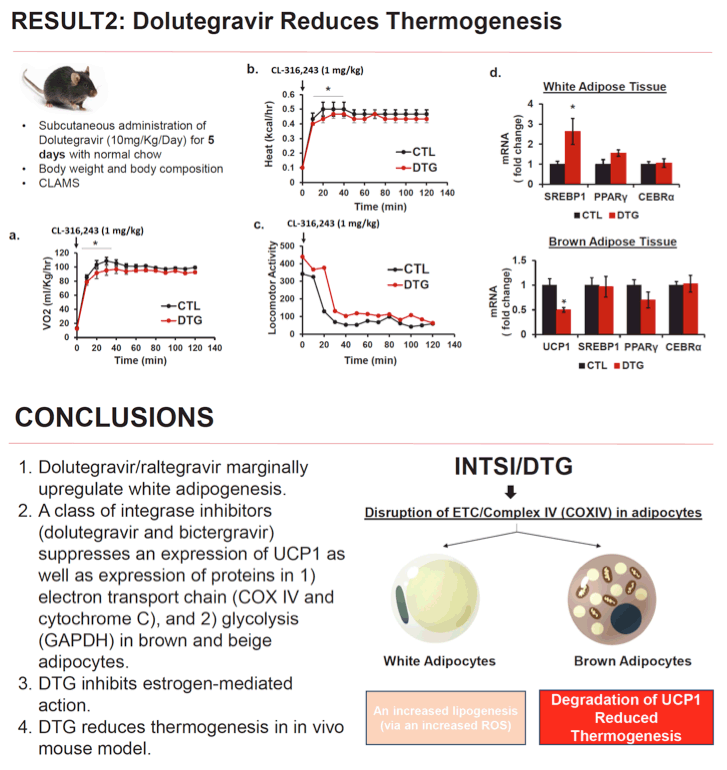
Methods: Primary preadipocytes were isolated from 4 weeks female mouse (C57BL/6). Cells were treated with InSTI (dolutegravir (DTG) or bictegravir (BIC)) or doravirine (DOR) for 8 days during differentiation into mature white or brown adipocytes. Mature adipocytes were analyzed for lipid accumulation by Oil Red O Staining, adipogenic markers by qRT-PCR and immunoblotting. Estrogen receptor mediated transcriptional activity was measured by luciferase reporter containing estrogen response element. Finally, we examined the effects of DTG (10mg/kg for 5 days) on food intake, energy expenditure, oxygen consumption in female mice using Comprehensive Laboratory Animal Monitoring System.
Results: We found that DTG and BIC mildly induced white adipocyte differentiation measured by white adipogenic markers (SREBP, CEBPα, and PPARγ) and lipid accumulation. In contrast, brown adipogenic markers (CEBPβ, PGC1αa and FABP4) were significantly reduced by DTG or BIC exposure (50-80%). Uncoupling protein1 (UCP1), which is an essential for a thermogenic process in brown/beige adipocytes, was downregulated by more than 90% compared to no treatment group. In addition, a decrease in UCP1 in brown adipocytes was accompanied by a decrease in cytochrome oxidase complex IV (COX IV) in mitochondria as well as GAPDH, a key glycolytic enzyme. Moreover, estrogen receptor-reporter assay revealed that estrogen-mediated pathway was blocked by DTG by 70%. DOR had no effect on fat differentiation, UCP1 expression, or mitochondrial enzyme activity. In vivo, DTG administration to female mice inhibited oxygen consumption and energy expenditure by 15% without affecting food consumption.
Conclusion: In in vitro models, InSTI exposure had opposite effects on the differentiation of white and brown fat. In brown adipocytes, the inhibition of brown thermogenic function by DTG was associated with interruption of mitochondrial proteins (e.g. COX IV and UCP1), which may be mediated through estrogen receptor. These findings suggest a novel mechanism by which InSTIs may lead to weight gain, especially in women.


|
| |
|
 |
 |
|
|