 |
 |
 |
| |
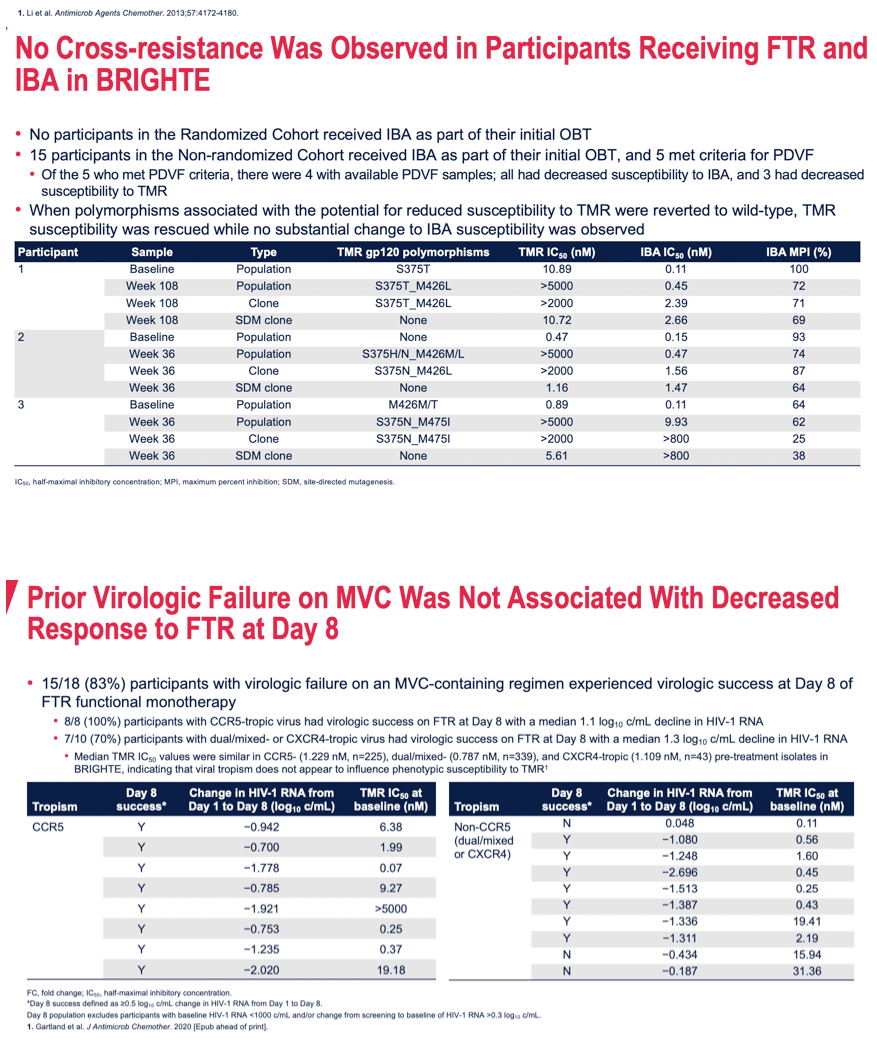
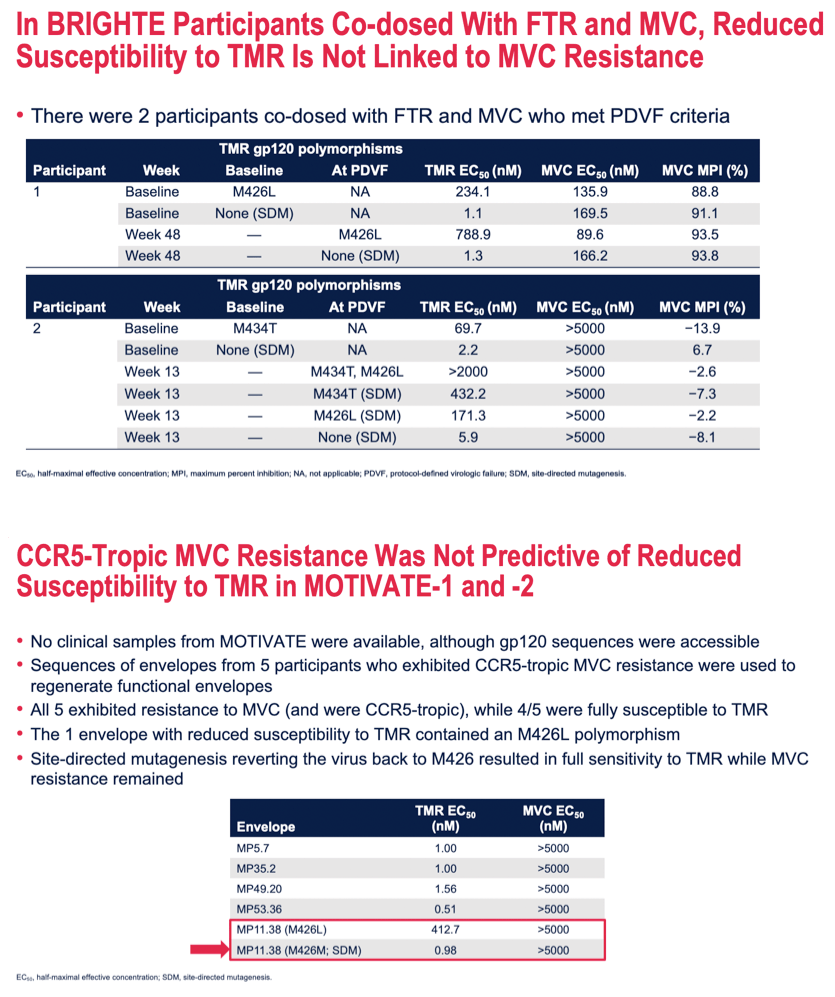
REDUCED SUSCEPTIBILITY TO TEMSAVIR IS NOT LINKED TO IBA OR MVC RESISTANCE
|
| |
| |
CROI 202 March 6-10 Reported by Jules Levin
Burt Rose1, Margaret Gartland2, Eugene Stewart3, Mark Cockett1, Peter Ackerman1, Max Lataillade1, Cyril Llamoso1, Mark Krystal1
1ViiV Healthcare, Branford, CT, USA, 2ViiV Healthcare, Research Triangle Park, NC, USA, 3GlaxoSmithKline, Collegeville, PA, USA
Background: Temsavir (TMR), the active agent of the gp120-directed attachment inhibitor fostemsavir (FTR), the CD4-directed attachment inhibitor ibalizumab (IBA) and the CCR5 antagonist maraviroc (MVC) are antiretroviral (ARV) agents that target steps in HIV-1 viral entry. Although the mechanisms of inhibition of the 3 agents are different, it is important to understand whether there is any potential for cross-resistance between these agents since all involve interactions with gp120.
Methods: Six envelopes derived from plasma samples from participants in the BRIGHTE study who experienced protocol-defined virologic failure (PDVF) and were co-dosed with FTR and either IBA or MVC (3 each) were generated and analyzed for susceptibility to the agents. In addition, 5 R5-tropic MVC-resistant envelopes from the MOTIVATE trials were regenerated based on their gp120 sequences. Site-directed mutagenesis of these envelopes was performed to understand whether susceptibility to multiple agents was linked.
Results: The envelopes from participants experiencing PDVF in BRIGHTE exhibited reduced susceptibility to TMR and resistance to the co-dosed agent (either IBA or MVC). At PDVF, emergent (compared to baseline) or pre-existing amino acids were present in all envelopes at one or more gp120 positions of interest (375, 426 or 475) that are known to have the potential to adversely affect susceptibility to TMR. When these positions were reverted to the consensus sequence (S375S, M426M or M475M), full susceptibility to TMR was restored in all cases without affecting resistance to the co-dosed agent. In addition, 5 regenerated envelopes from the MOTIVATE studies all exhibited R5-tropic MVC resistance. Only 1 exhibited reduced susceptibility to TMR as it contained an M426L polymorphism. When mutated to L426M, this envelope reverted to full sensitivity for TMR, but remained MVC resistant.
Conclusion: Using envelopes obtained from clinical studies, the data clearly show that decreased susceptibility to TMR and resistance to IBA or MVC are not linked and that there is no cross-resistance with these latter 2 agents and FTR.


|
| |
|
 |
 |
|
|