 |
 |
 |
| |
PHASE IIA PROOF-OF-CONCEPT TRIAL OF NEXT-GENERATION MATURATION INHIBITOR GSK3640254
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Christoph Spinner , Franco B. Felizarta , Giuliano Rizzardini , Patrick Philibert, Essack Mitha , Pere Domingo , Christoph Stephan , Michelle DeGrosky , Veronica Bainbridge , Joyce Zhan, Teodora Pene Dumitrescu, Samit R. Joshi, Max Lataillade, Jerry L. Jeffrey, for 208132 Study Team
1TechnicalUniversityofMunich,Munich,Germany, OfficeofFrancoFelizarta, 2Bakersfield,CA,USA, ASST Fatebenefratelli, Milan, Italy, Hôpital Européende 3Marseille, Marseille, France, Newtown Clinical Research, Johannesburg, South Africa, Hospital Santa Creuy Sant Pau, Barcelona, Spain, 7Universitäts klinikum Frankfurt,Frankfurt,Germany, 8ViiV Healthcare, Branford,CT,USA, GlaxoSmithKline, Uxbridge, UK, 10GlaxoSmithKline, Collegeville, PA, USA, ViiV Healthcare, Research Triangle Park, NC, USA
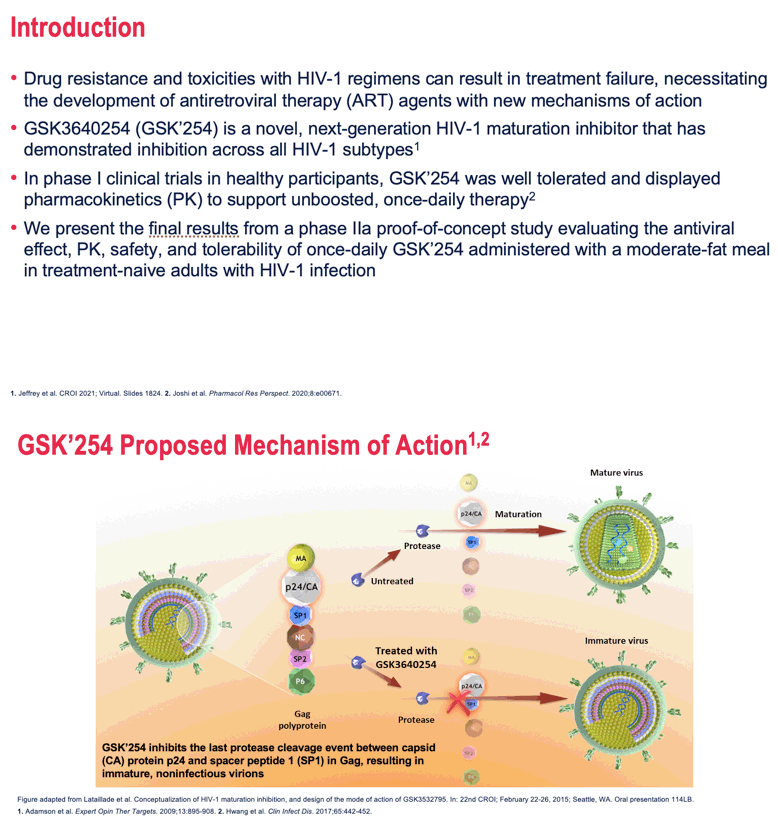
Background: Drug resistance and toxicities with HIV-1 regimens can result in treatment failure, so antiretroviral agents with new mechanisms of action are needed. GSK3640254 (GSK'254) is a novel, next-generation HIV-1 maturation inhibitor with pharmacokinetics (PK) to support unboosted, once-daily therapy and a desirable drug-drug interaction profile.
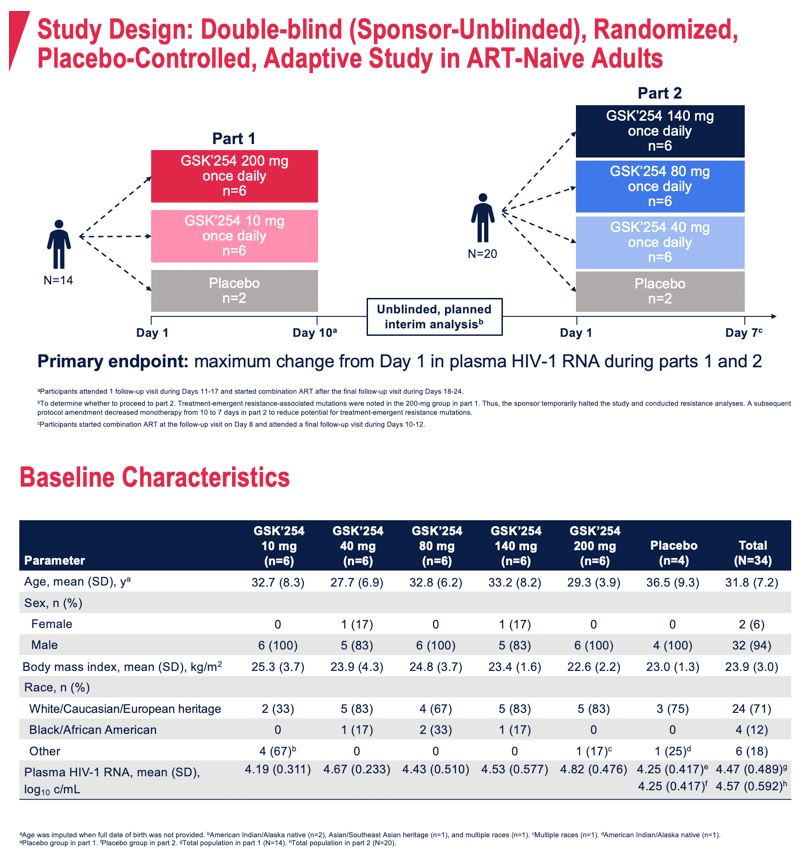
Methods: This phase IIa double-blind (sponsor unblinded), randomized, placebo-controlled, adaptive study evaluated the antiviral effect, safety, tolerability, and PK of once-daily GSK'254 administered with a moderate-fat meal in treatment-naive adults infected with HIV-1. In part 1, participants received GSK'254 10 or 200 mg for 10 days. In part 2, participants received GSK'254 40, 80, or 140 mg for 7 days, modified from 10 days by a protocol amendment to decrease potential for treatment-emergent resistance- associated mutations (RAMs) observed after 7 days in part 1. Primary endpoint was maximum change in plasma HIV-1 RNA during parts 1 and 2. Secondary endpoints were safety, tolerability, and PK parameters.
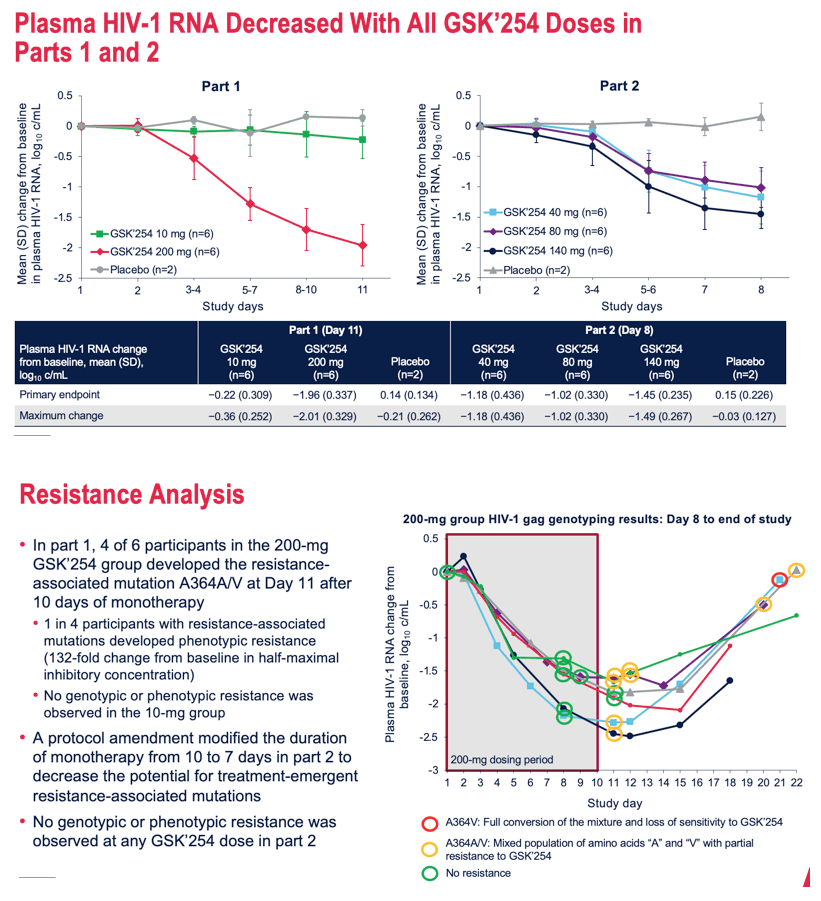
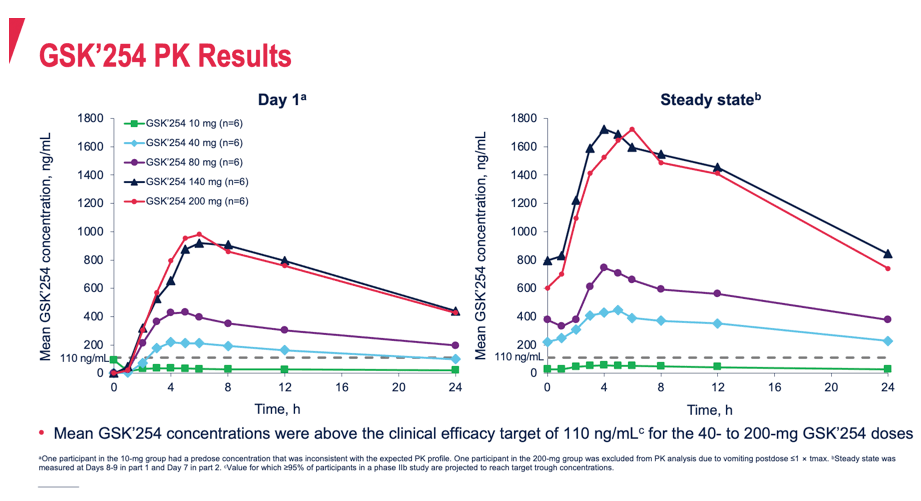
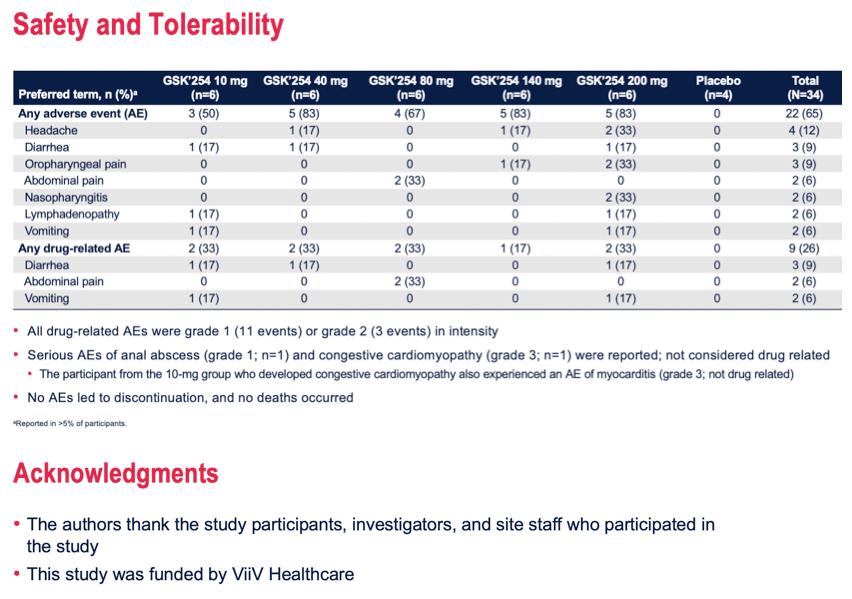
Results: Of 34 participants who received GSK'254 (n=6/dose) or placebo (n=4), 94% were men; mean age was 31.8 years. Mean changes in plasma HIV-1 RNA ranged from -2.0 to 0.2 log10; the largest decreases of 2.0- and 1.5 log10 occurred in the 200- and 140-mg groups, respectively (Table). PK results were generally dose proportional and consistent with those observed in non-HIV-infected individuals. 4/6 participants in the 200-mg group (part 1) developed RAMs on Day 11; 1 of these 4 developed phenotypic resistance. No RAMs were observed at any dose in part 2. No deaths were reported. Two non-drug-related serious adverse events (AEs) of congestive cardiomyopathy and anal abscess occurred. No AEs led to discontinuation. AEs were reported by 22 (65%) participants, with the most common being headache (n=4). Drug-related gastrointestinal AEs, including diarrhea (n=3), abdominal pain (n=2), and vomiting (n=2), occurred in 6 (18%) participants. All AEs and serious AEs were mild to moderate in intensity, except for 1 participant who developed congestive cardiomyopathy and myocarditis (both g
Conclusion: This short-term monotherapy study established a dose-antiviral response relationship. Regardless of dosing duration, GSK'254 140- and 200-mg doses demonstrated greatest declines in plasma HIV-1 RNA. No safety or tolerability concerns were noted. These results support the ongoing phase IIb study (ClinicalTrials.gov identifier: NCT04493216).


|
| |
|
 |
 |
|
|