 |
 |
 |
| |
DoLPHIN2 FINAL RESULTS DOLUTEGRAVIR VS EFAVIRENZ IN LATE PREGNANCY TO 72W POSTPARTUM
|
| |
| |
DoLPHIN2 FINAL RESULTS DOLUTEGRAVIR VS EFAVIRENZ IN LATE PREGNANCY TO 72W POSTPARTUM
CROI 2021 March 6-10 Reported by Jules Levin
Thokozile R . Malaba1, Irene Nakatudde2, Kenneth Kintu2, Tao Chen3, Sabrina Bakeera-Kitaka2, Lucy Read3, Helen Reynolds4, Angela Colbers5, Kelly Byrne3, Duolao Wang3, Catriona Waitt4, Catherine Orrell1, Mohammed Lamorde2, Landon Myer1, Saye Khoo4
1University of Cape Town, Cape Town, South Africa, 2Makerere University, Kampala, Uganda, 3Liverpool School of Tropical Medicine, Liverpool, UK, 4University of Liverpool, Liverpool, UK, 5Radboud University, Nijmegen, Netherlands
program abstract
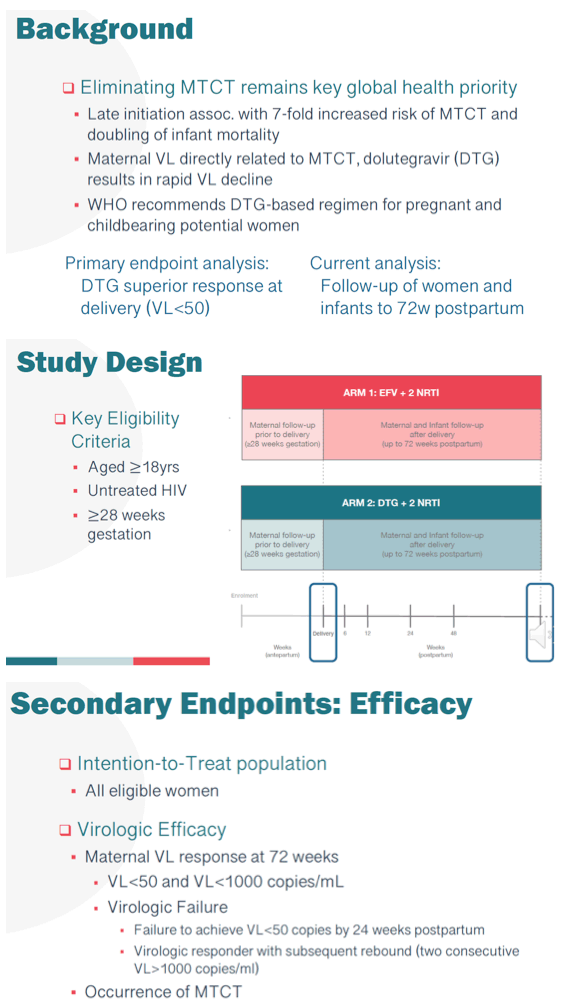
Background: Delayed ART initiation in pregnancy is associated with failure to achieve viral suppression and increased risk of MTCT. DolPHIN-2 (NCT03249181) randomized pregnant women initiating treatment in the third trimester to either dolutegravir (DTG) or efavirenz (EFV) based regimens in South Africa and Uganda. Preliminary analysis of the primary endpoint (viral load (VL) <50 copies at delivery) have been published.
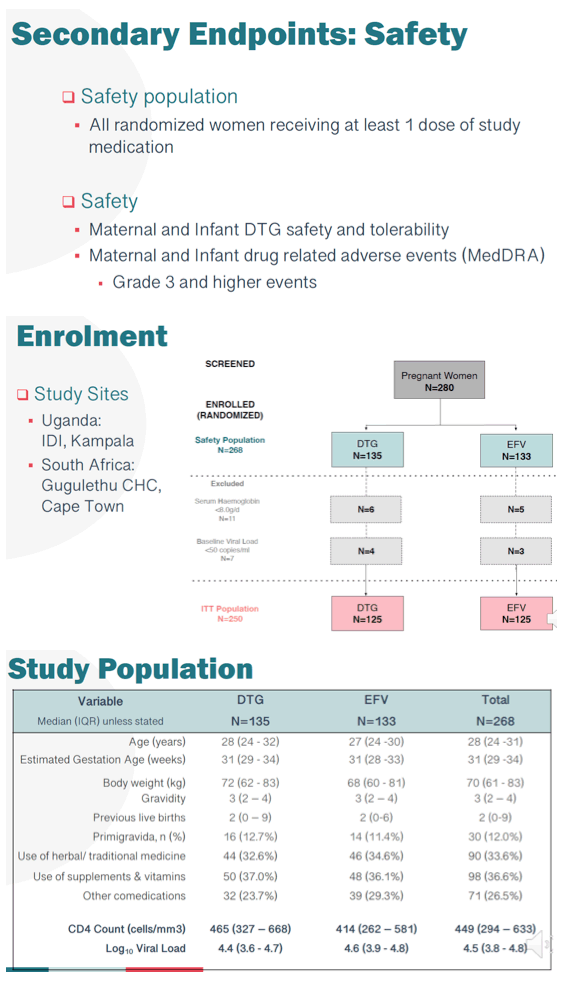
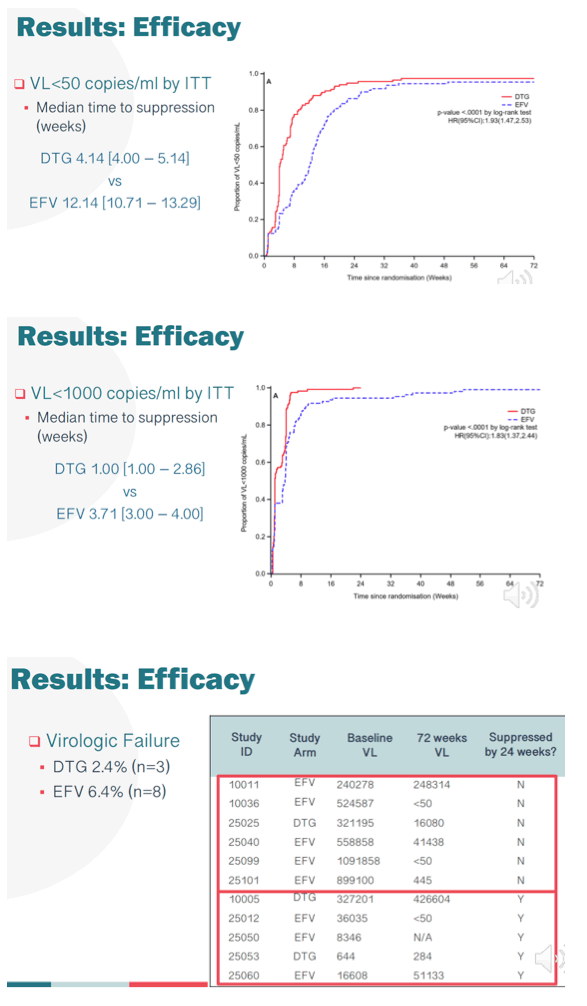
Methods: Between Jan-Aug 2018, 268 mothers (safety cohort) were randomized to receive EFV (133) or DTG (135), of whom 250 (EFV-125, DTG-125, intention-to-treat cohort) were evaluable for efficacy. In addition to measurement in pregnancy, VL was also measured at 6, 12, 24, 48 and 72w postpartum (PP). The primary endpoints were VL<50 copies/mL for efficacy; and the occurrence of maternal and/or infant drug related serious adverse events (SAE) for safety. Here we present final data with follow-up of mothers and infants to 72w PP.
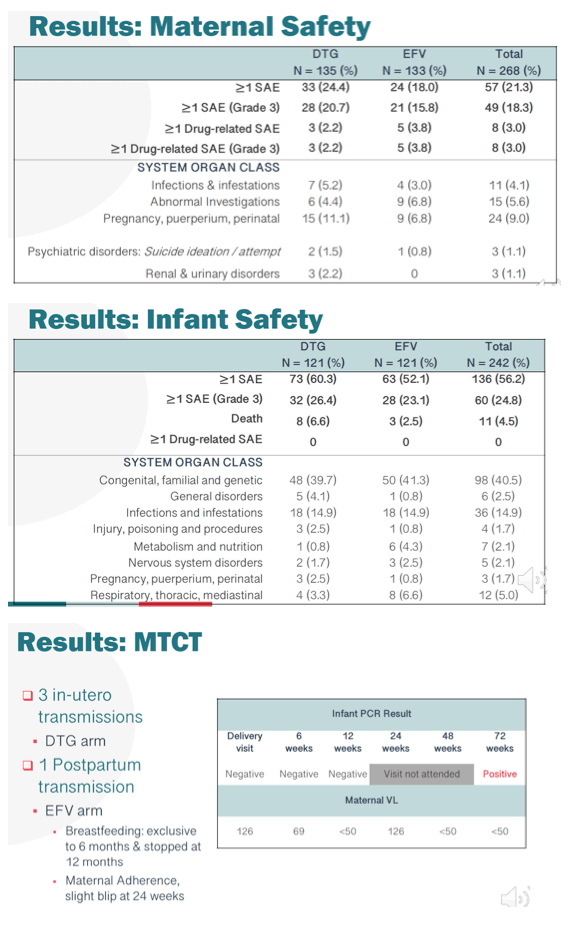
Results: As previously reported, DTG was associated with superior responses (VL<50) in the first 26w of therapy. At 72w, 116/125 mothers receiving DTG achieved VL<50 with a median time of 4.14 (IQR4.00, 5.14) weeks. In contrast, among 114/125 mothers randomized to the EFV arm, suppression was achieved at a median time of 12.14 (IQR 10.71, 13.29) weeks (adjusted HR 1.93 (95% CI 1.47, 2.53)) (figure). By 72w PP, 21.3% of mothers and 56.2% of infants experienced an SAE, however in mothers only 3% was related to study drug, with no infant drug related events. DTG was well tolerated with a lower frequency of maternal drug related AE (DTG 2.2% vs EFV 3.8%). Overall, the mean change in maternal weight from delivery to 72w PP was -1.2kg, with nonsignificant differences observed by arm in weight retention (DTG -0.7kg vs EFV -1.6kg). No differences in maternal glycosuria or infant hyperglycaemia were observed by arm. Overall 4 infant HIV infections were detected; 3 at delivery in DTG arm, with a new transmission detected at 72w PP in EFV arm despite optimal maternal suppression (VL<50) from delivery and serial negative tests in the child
Conclusion: Maternal DTG-based ART was safe and well tolerated. Women randomized to DTG had more rapid viral suppression after initiation of ART and they maintained virologic suppression through the breastfeeding period. The infant HIV infection in the EFV arm highlights the potential for transmission during breastfeeding in mothers despite evidence of virologic suppression.

|
| |
|
 |
 |
|
|