 |
 |
 |
| |
WEEK 96 EFFICACY AND SAFETY OF CABOTEGRAVIR + RILPIVIRINE EVERY 2 MONTHS: ATLAS-2M
|
| |
| |
CROI 2021 March 6
Reported by Jules Levin
ViiV Healthcare presents data for long-acting cabotegravir and rilpivirine for the treatment of HIV showing continued virologic suppression to 96 weeks
webcast: http://www.croiwebcasts.org/console/player/47431?mediaType=slideVideo&
Hans Jaeger1, E. Turner Overton2, Gary Richmond3, Giuliano Rizzardini4, Jaime Federico Andrade-Villanueva5, Rosie Mngqibisa6, Antonio Ocampo Hermida7, Anders Thalme8, Paul Benn9, Yuanyuan Wang10, Krischan J. Hudson11, David M. Margolis11, Christine Talarico11, Kati Vandermeulen12, William R. Spreen11
1MVZ Karlsplatz HIV Research and Clinical Care Center, Munich, Germany, 2University of Alabama at Birmingham, Birmingham, AL, USA, 3Nova Southeastern University, Fort Lauderdale, FL, USA, 4Fatebenefratelli Sacco Hospital, Milan, Italy, 5Hospital Civil Fray Antonio Alcalde, Guadalajara, Mexico, 6Durban International Clinical Research Site, Durban, South Africa, 7Complejo Hospitalario Universitario de Vigo, Vigo, Spain, 8Karolinska University Hospital, Stockholm, Sweden, 9ViiV Healthcare, Brentford, UK, 10GlaxoSmithKline, Collegeville, PA, USA, 11ViiV Healthcare, Research Triangle Park, NC, USA, 12Janssen, Beerse, Belgium
Background: The dosing frequency of cabotegravir (CAB) and rilpivirine (RPV) long-acting (LA) administered every 1 or 2 months may address challenges associated with daily oral ART, such as adherence, pill burden, and stigma.
The Week (W) 48 results from ATLAS-2M (NCT03299049) demonstrated noninferiority of CAB+RPV LA administered every 8 weeks (Q8W) compared with every 4 weeks (Q4W). Here, we report the W96 results.
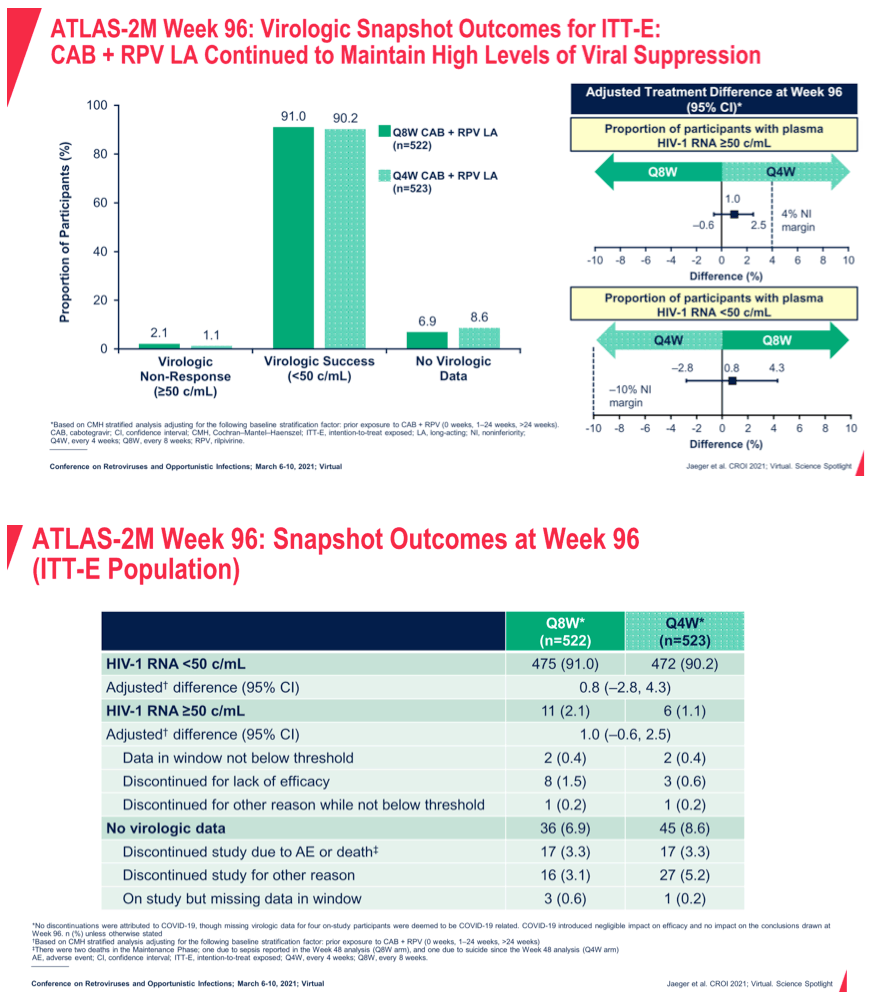
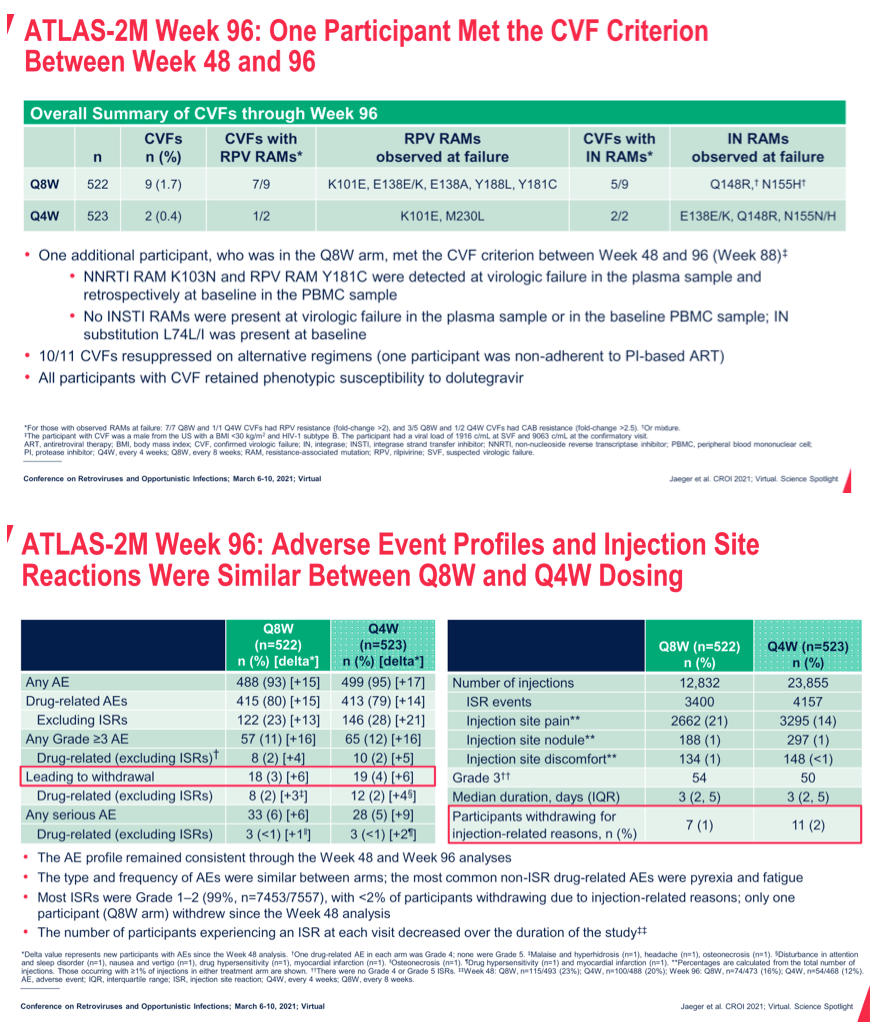
Methods: ATLAS-2M is an ongoing Phase 3b, randomized, multicenter study assessing the efficacy and safety of CAB+RPV LA Q8W vs. Q4W. Virologically suppressed individuals receiving CAB+RPV LA Q4W (ATLAS [NCT02951052] study rollover) or oral SoC were randomized 1:1 to receive CAB+RPV LA Q8W or Q4W. The primary endpoint at W48 was the proportion of participants with plasma HIV-1 RNA ≥50 c/mL (FDA Snapshot, ITT-E; 4% noninferiority margin). Endpoints assessed at W96 include proportion of participants with plasma HIV-1 RNA ≥50 c/mL and HIV-1 RNA <50 c/mL, incidence of confirmed virologic failure (CVF; two consecutive plasma HIV-1 RNA ≥200 c/mL), safety, and tolerability. Results: 1045 participants received CAB+RPV LA (Q8W, n=522; Q4W, n=523). The median (range) age was 42y (19-83); 27% were female (sex at birth), and 73% were white. At W96, CAB+RPV LA Q8W confirmed noninferior efficacy to Q4W dosing, with 2.1% (n=11) and 1.1% (n=6) of participants having HIV-1 RNA ≥50 c/mL in each arm, respectively (Table 1). High levels of virologic suppression were observed across both arms, with 90-91% of participants maintaining HIV-1 RNA <50 c/mL at W96. Through W96, 9 (1.7%) and 2 (0.4%) participants in the Q8W and Q4W arms had CVF, respectively; 1 occurred between W48 and W96 (participant in Q8W arm with baseline RPV resistance-associated mutation [RAM] Y181C and no InSTI RAMs). Safety profiles were comparable between arms, with no new safety signals identified since the W48 analysis. Injection site reactions (ISRs) were the most common adverse event and led to 1 withdrawal between W48 and W96 (Q8W arm); most were mild or moderate in severity (98.6%), with a median duration of 3 days. The frequency of ISRs decreased over time (W48: Q8W, n=115/493 [23%]; Q4W, n=100/488 [20%]; W96: Q8W, n=74/473 [16%]; Q4W, n= 54/468 [12%]).
Conclusion: Efficacy of CAB+RPV LA Q8W continued to be noninferior to Q4W at W96, with both regimens maintaining high levels of virologic suppression. These longer-term efficacy, safety, and tolerability data further support the therapeutic potential of CAB+RPV LA.




|
| |
|
 |
 |
|
|