 |
 |
 |
| |
A PLACEBO-CONTROLLED ATI TRIAL OF HTI VACCINES IN EARLY TREATED HIV INFECTION
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Lucia Bailon1, Anuska Llano2, Samandhy Cedeno2, Miriam B. Lopez1, Yovaninna Alarcon1, Pep Coll2, Angel Rivero3, Anne R. Leselbaum4, Ian McGowan5, Devi SenGupta6, Bonaventura Clotet2, Christian Brander2, Jose Molto1, Beatriz Mothe2, for the AELIX-002 Trial Group
1Fundació Lluita Contra la Sida, Badalona, Spain, 2IrsiCaixa Institute for AIDS Research, Badalona, Spain, 3Projecte dels NOMS-Hispanosida, BCN Checkpoint, Barcelona, Spain, 4AELIX Therapeutics S.L., Barcelona, Spain, 5University of Pittsburgh, Pittsburgh, PA, USA, 6Gilead Sciences, Inc, Foster City, CA, USA
Background: HTI is a novel HIV vaccine immunogen designed at redirecting cellular immune responses to HIV targets associated with viral control.
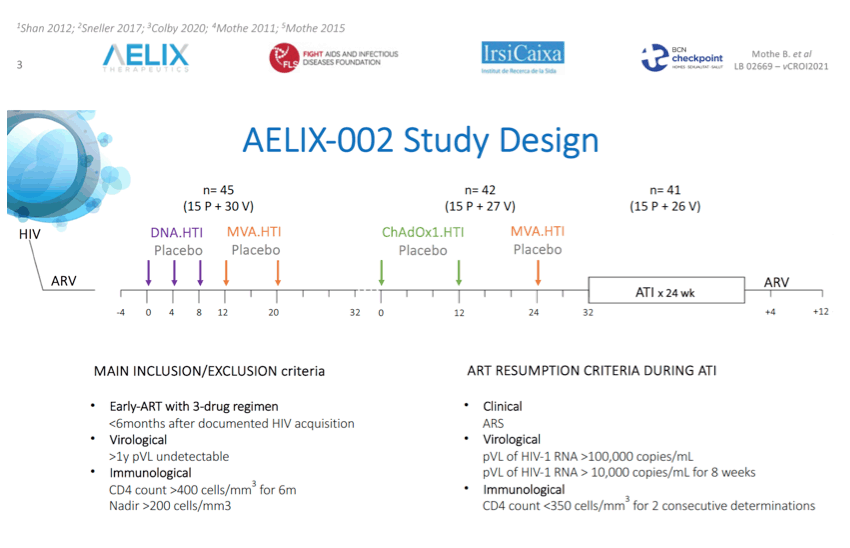
Methods: The AELIX-002 trial (NCT03204617) was a randomized, single- center, placebo-controlled trial to evaluate the safety, immunogenicity and antiviral effect of DNA.HTI (D), MVA.HTI (M) and ChAdOx1.HTI (C) vaccines after discontinuation of ART in early-treated people living with HIV (PLWH). 45 participants were randomized (2:1) to receive heterologous prime-boost vaccination regimens consisting of DDDMM followed by CCM, or matched placebo (P). During a 24-week analytical treatment interruption (ATI), plasma viral load (pVL) was monitored weekly and ART was resumed if pVL >100,000 copies/mL, or >10,000 copies/mL over 8 weeks, and/or CD4<350.
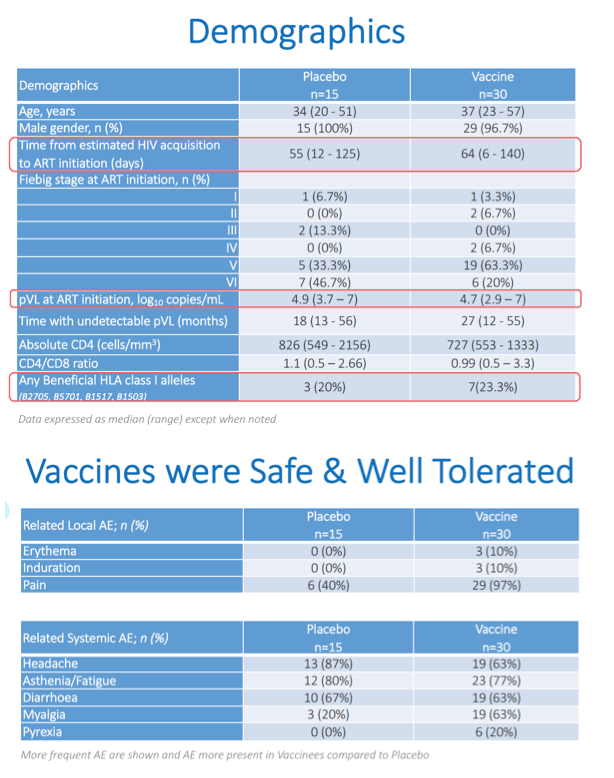
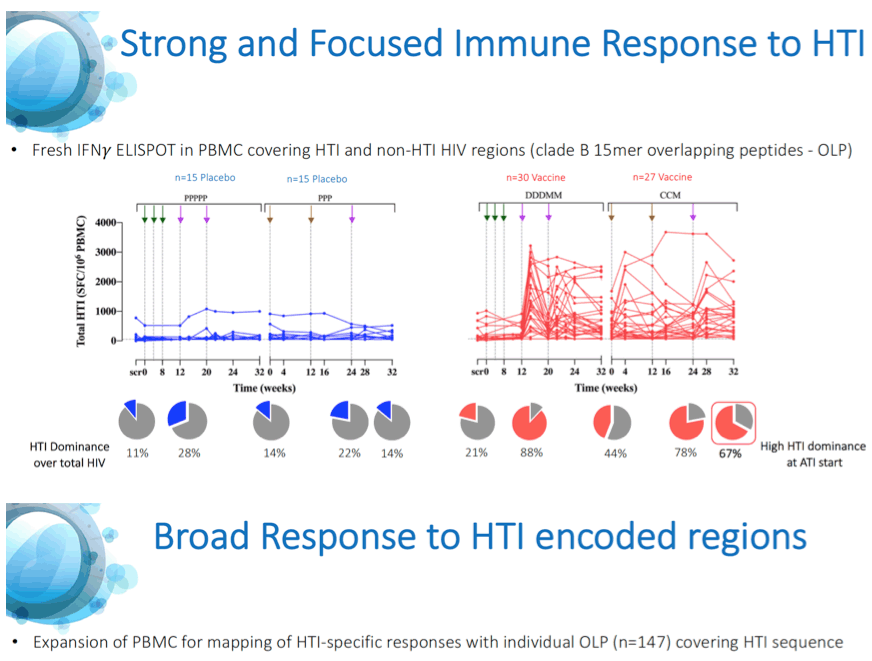
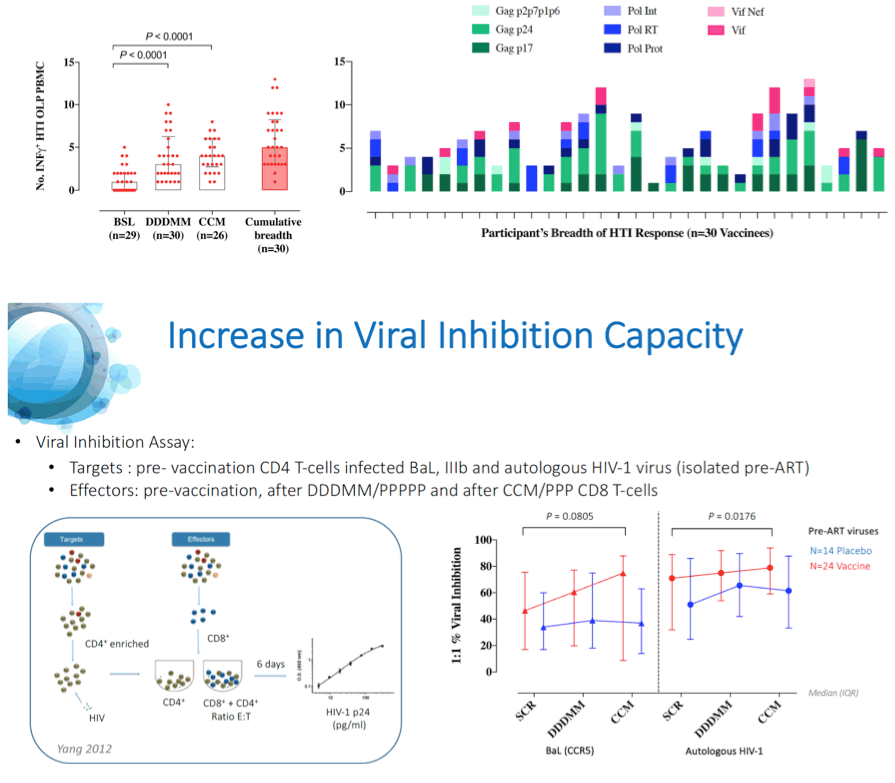
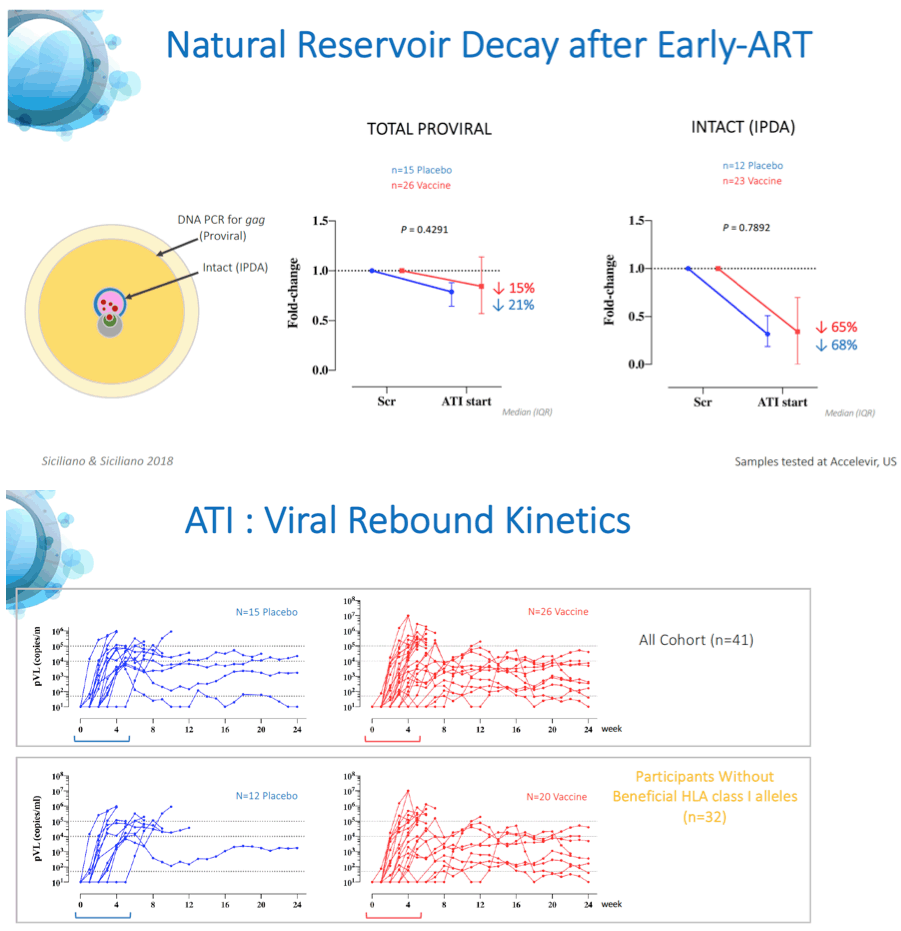
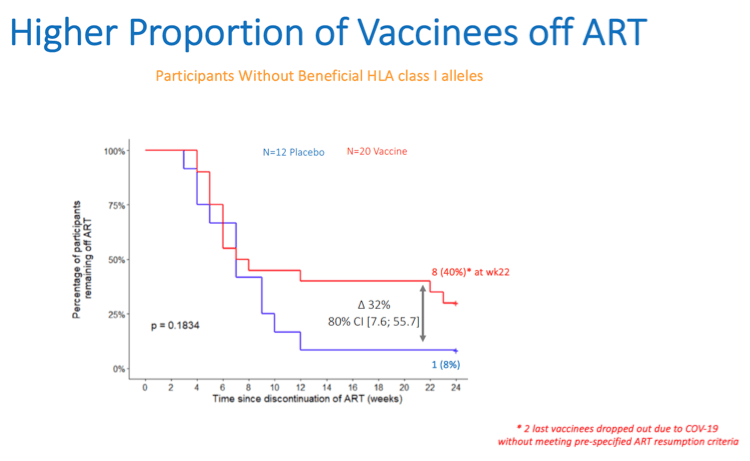
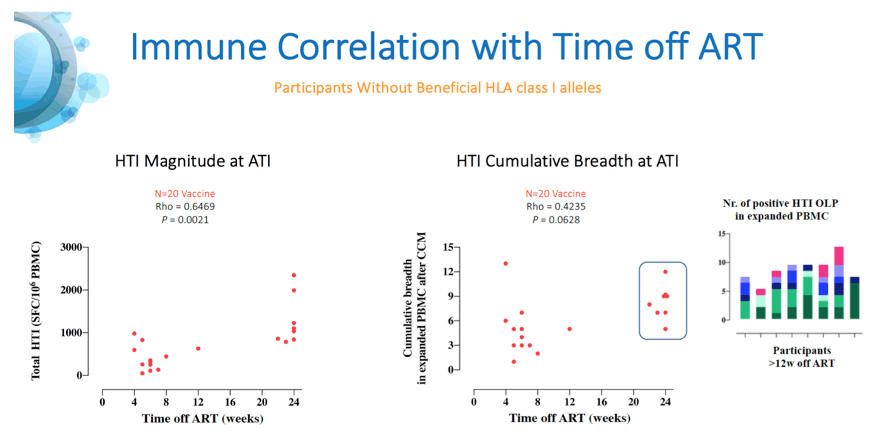
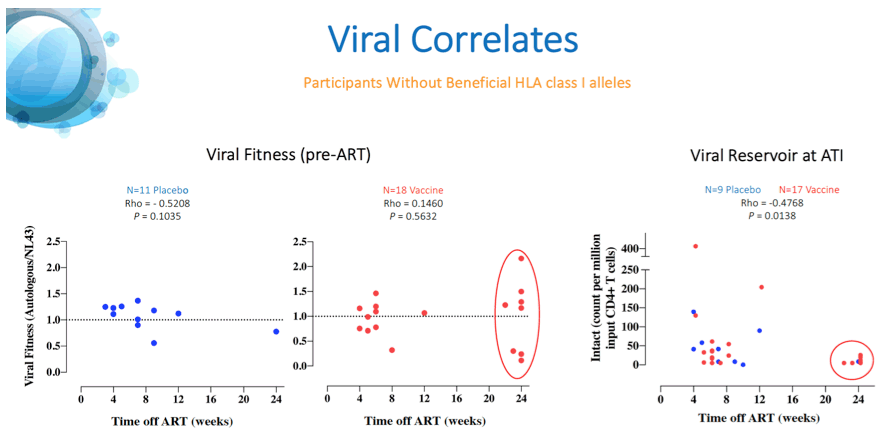
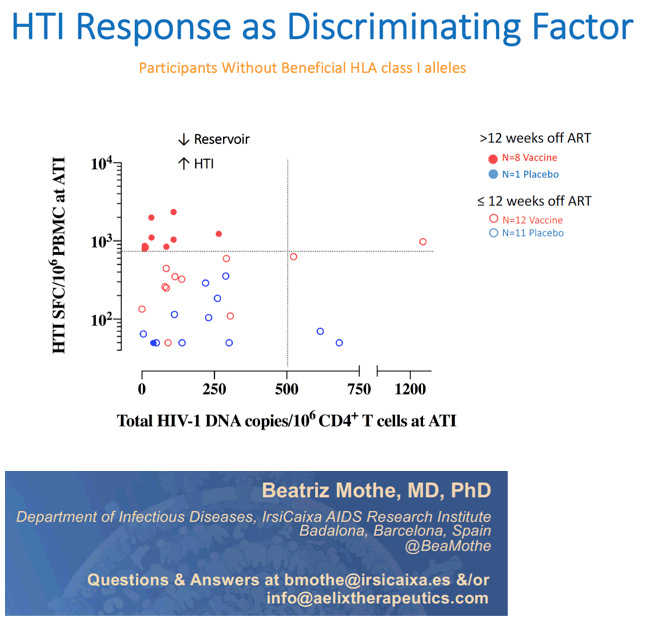
Results: A total of 45 participants received DDDMM (n=30) or PPPPP (n=15). Of the 45 participants, 41 further completed the CCM (n=26) or PPP (n=15) regimen and entered the ATI. Immunizations were well tolerated, with no SAEs, and were immunogenic in 97% of vaccine recipients (defined by a >2-fold increase in HTI-specific T cell responses compared to baseline). Median (range) increase in total frequencies of HTI-specific T cells from baseline was 1,499 (120 to 3,150) SFC/million PBMC. At time of ATI start, 71% (0 to 100) of the total anti- HIV-1 T-cell response was HTI-specific. For participants without any potentially beneficial HLA class I alleles (32 of the 41), 8 (40%) of the vaccinees and 1 (8%) of the placebo recipients were able to remain off ART for 22 weeks (∇ 32%, 80%CI [7.6; 55.7]); with pVL <2,000 copies/mL being observed in 5 and 1 vaccine and placebo recipients, respectively. Magnitude of HTI-specific responses at the time of ATI start positively correlated with time off ART in vaccinees (Rho 0.65, p < 0.01). Decay in total or intact HIV proviral DNA from baseline to ATI was similar between vaccine and placebo arms.
Conclusion: HTI vaccines were safe and highly immunogenic in early-treated PLWH with a prolonged time off ART seen in vaccinees with non-beneficial HLA class I alleles. Time off ART positively correlated with vaccine-induced HTI-specific T cell responses at ATI start. Multivariate analysis for other correlates of response is ongoing. These encouraging data strongly support the use of HTI- based vaccines as the backbone of combination cure regimens such as with the TLR7 agonist vesatolimod, which is currently being evaluated in the AELIX-003 study (NCT04364035).


|
| |
|
 |
 |
|
|