 |
 |
 |
| |
Acute Kidney Injury Less Frequent With Remdesivir Than Standard of Care
|
| |
| |
CROI 2021, Conference on Retroviruses and Opportunistic Infections, March 6-10, 2021
Mark Mascolini
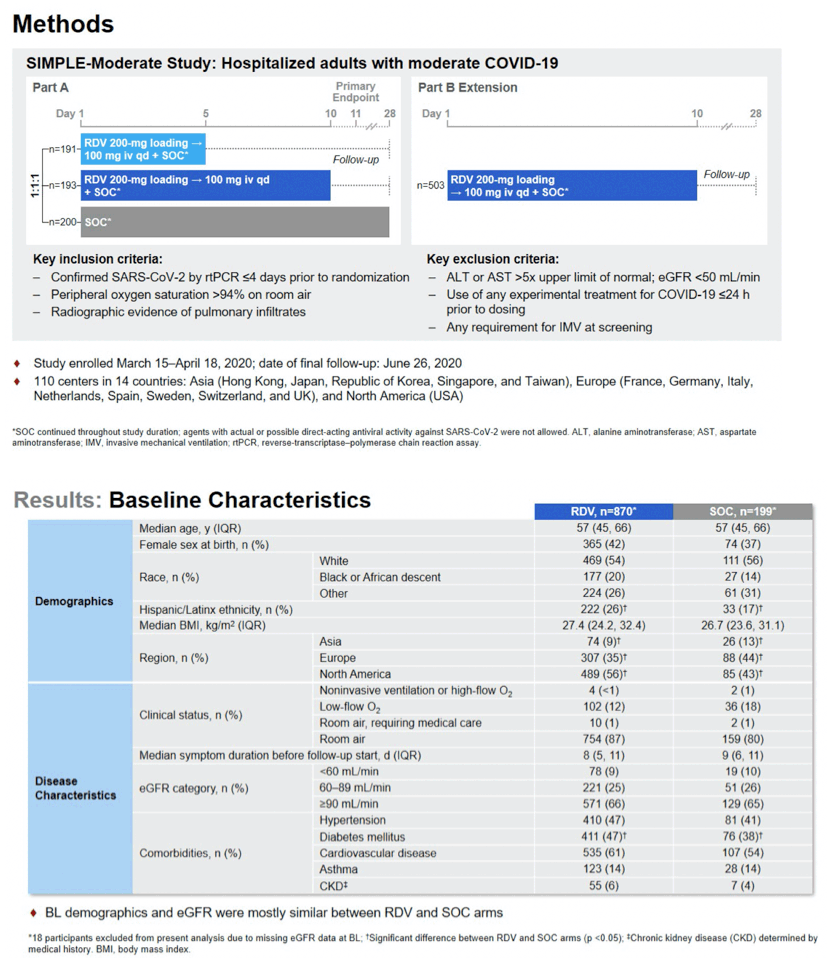
Acute kidney injury (AKI) proved less frequent in people receiving remdesivir than in those getting standard care in the SIMPLE-Moderate study of inpatients with moderate COVID-19 [1]. The remdesivir group and the standard-care group did not differ in a key kidney function marker (estimated glomerular filtration rate, eGFR) over time; nor did they differ in kidney adverse events during the study period.
Because the kidneys clear both remdesivir and its excipient (carrying agent) cyclodextrin from the body, one might expect people getting daily infusions of this drug would suffer AKI more often than similar people not getting remdesivir. To assess the impact of remdesivir on AKI, researchers analyzed data from the SIMPLE-Moderate trial, which compared 5- or 10-day remdesivir infusions with standard care in inpatients with COVID-19 [2]. They found that people getting remdesivir for 5 days (but not 10 days) had higher odds of improved clinical status than did the standard-care group, "but the difference was of uncertain clinical importance" [2].
The AKI analysis involved 870 people receiving remdesivir during the trial and 199 receiving standard care. The remdesivir and standard-care groups were similar in median age (57 in both groups), in proportions of women (42% and 37%), in racial distribution, and in body mass index (27.4 and 26.7 kg/m2). Everyone had confirmed SARS-CoV-2 infection, oxygen saturation above 94% on room air, radiographic evidence of pulmonary infiltrates, and eGFR at or above 50 mL/min. The researchers defined AKI as a creatinine increase from the baseline (initial) value classified as "stage 1 (increase > 0.3 and % change ≤25%, or % change >25% and ≤ 100%), stage 2 (% change >100% and ≤200%), and stage 3 (% change >200%)."
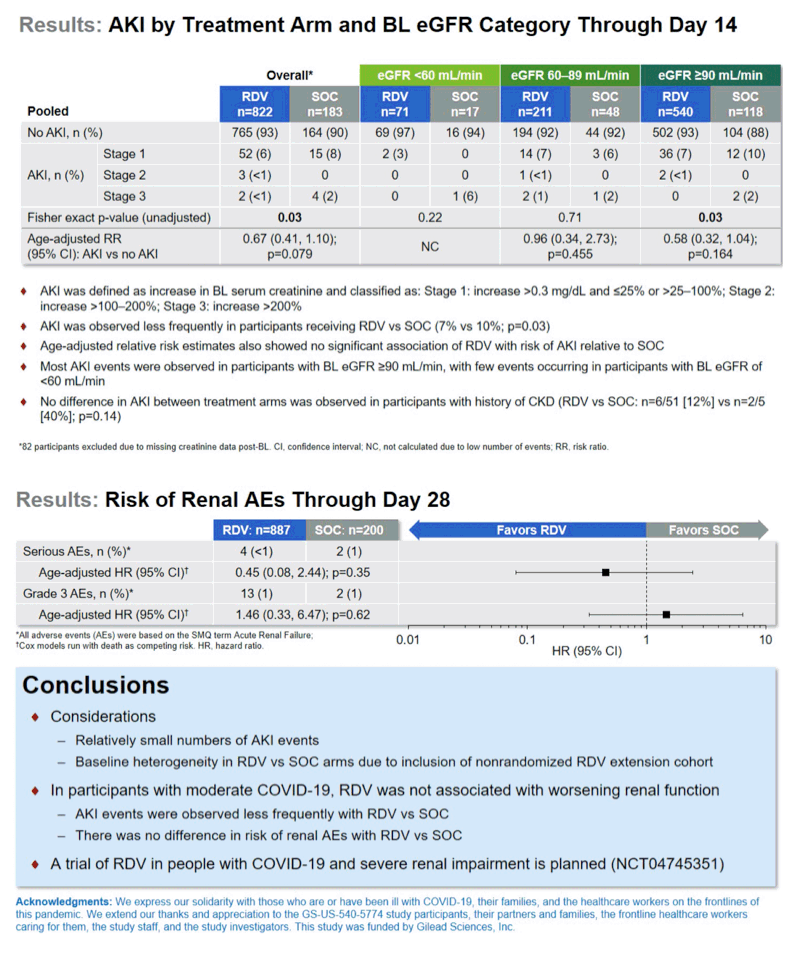
Overall the researchers saw AKI develop in 7% of trial participants randomized to remdesivir and 10% randomized to standard care, a significant difference (P = 0.03). When the researchers adjusted AKI risk by age (because older people run a higher risk of poor kidney function), they found no significant link between remdesivir use and AKI risk compared with standard care (adjusted relative risk 0.67, 95% confidence interval 0.41 to 1.10, P = 0.079).
When the research team calculated AKI rates in people who started the trial with a history of chronic kidney disease (CKD), they found no significant difference in AKI risk with remdesivir versus standard care (12% with remdesivir vs 40% with standard care, P = 0.14).
Most AKIs occurred in people with good kidney function when entering the trial (eGFR above 90 mL/min), while few AKIs arose in people with an eGFR below 60 mL/min. And eGFR did not differ significantly over 14 days between the remdesivir group and the standard-care group. Statistical analysis adjusted for age determined that taking remdesivir did not affect chances of a serious adverse event or a grade 3 adverse event during study period.
The researchers concluded that in these hospital inpatients with moderate COVID-19, taking remdesivir was not linked to worsening kidney function. They added that a placebo-controlled trial of remdesivir in people with COVID-19 and severe kidney impairment is being planned [3].
References
1. Ogbuagu O, Tashima KT, Gunthard HF, et al. Acute kidney injury in patients with moderate COVID-19 treated with remdesivir versus SoC. CROI 2021, Conference on Retroviruses and Opportunistic Infections, March 6-10, 2021. Abstract 395.
2. Spinner CD, Gottlieb RL, Criner GJ, et al. Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial. JAMA. 2020;324:1048-1057. doi: 10.1001/jama.2020.16349.
3. ClinicalTrials.gov. Study to evaluate the efficacy and safety of remdesivir in participants with severely reduced kidney function who are hospitalized for coronavirus disease 2019 (COVID-19). ClinicalTrials.gov identifier NCT04745351.

|
| |
|
 |
 |
|
|