 |
 |
 |
| |
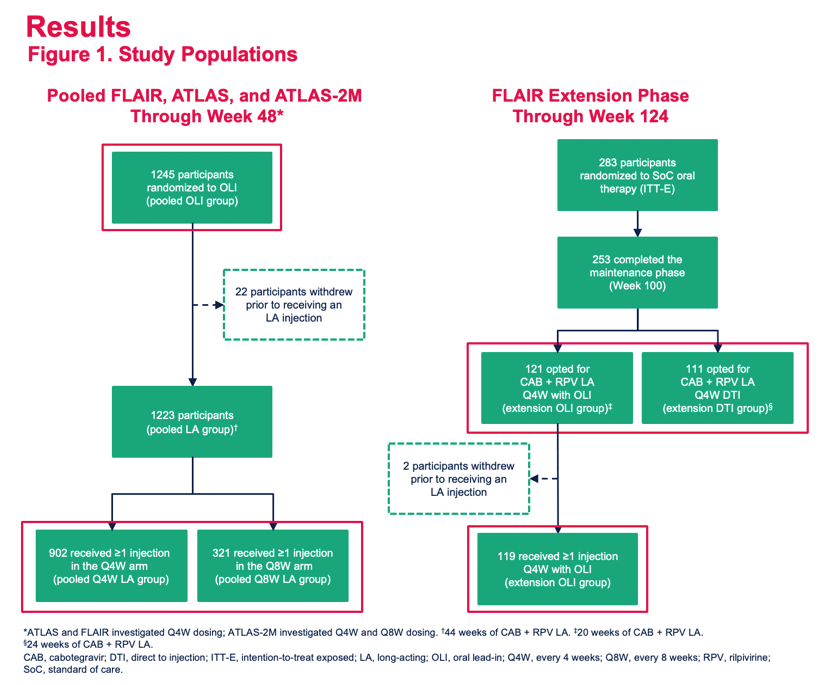
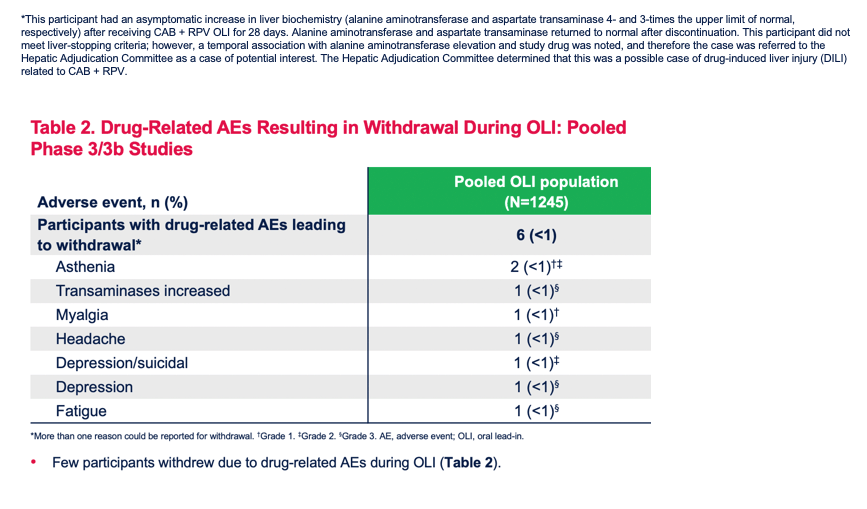
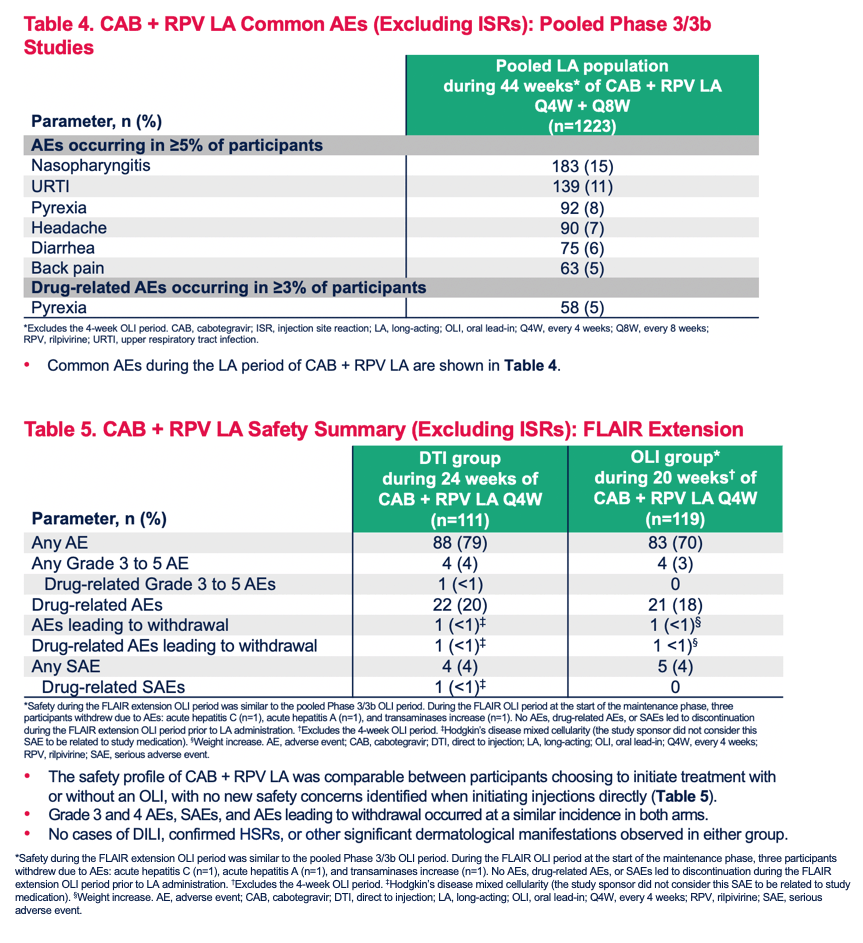
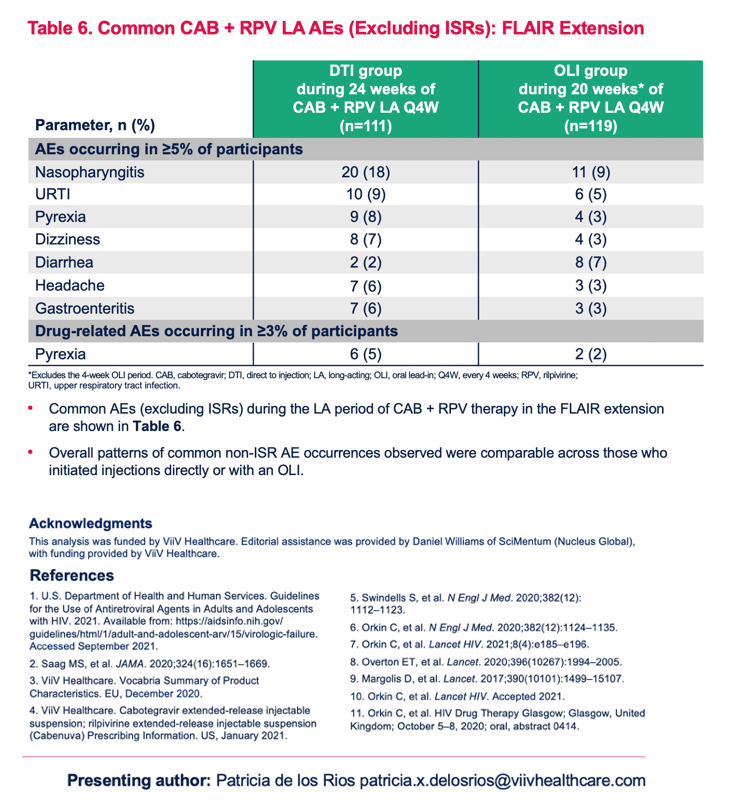
Safety Profile of Cabotegravir + Rilpivirine During Oral Lead-In and Through Long-Acting Therapy: Pooled Analysis of the Phase 3 FLAIR, ATLAS, and ATLAS-2M Studies
|
| |
| |
EACS 2021 Oct 27-30
Patricia de los Rios1, Parul Patel1, Jenny Huang2, Conn Harrington1, Ronald D'Amico1, Shanker Thiagarajah3, Eileen Birmingham4, Rodica Van Solingen-Ristea5, William R. Spreen1, Bryan Baugh6, Matt Bosse7
1ViiV Healthcare, Research Triangle Park, NC, United States; 2GlaxoSmithKline, Mississauga, Ontario, Canada; 3GlaxoSmithKline, London, United Kingdom; 4Janssen Research & Development LLC, Raritan, NJ, United States;
5Janssen Research & Development, Beerse, Belgium; 6Janssen Pharmaceutical, Research & Development, Titusville, NJ, United States; 7ViiV Healthcare, San Diego, CA, United States




|
| |
|
 |
 |
|
|