 |
 |
 |
| |
Maraviroc or Metformin Does Not Lower Liver Fat in People With HIV and NAFLD
|
| |
| |
18th European AIDS Conference, EACS 2021, October 27-30, 2021, London
Mark Mascolini
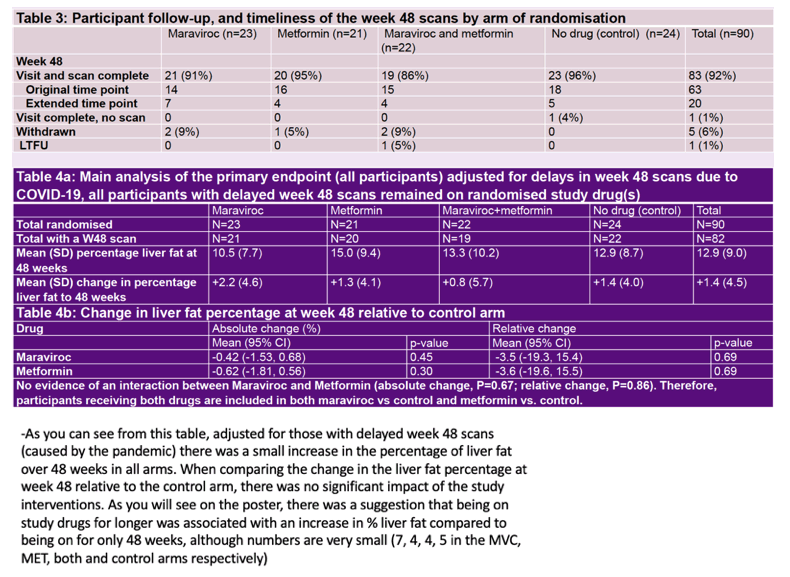
Maraviroc, metformin, or both drugs did not lower liver fat percentage in a randomized trial of 90 people with HIV infection and nonalcoholic fatty liver disease (NAFLD) [1]. In fact, people who took maraviroc or metformin longer than 48 weeks ended up with a higher liver fat percentage than people treated for only 48 weeks.
HIV-positive people have a high prevalence of NAFLD, but no medication is licensed to treat this condition. Researchers at University College London and collaborators at other centers suggested that maraviroc (an antiretroviral CCR5 antagonist) and metformin (a diabetes drug) may address NAFLD-maraviroc through its impact on inflammation of the liver and system-wide inflammation, and metformin through its impact on free fatty acid metabolism and insulin sensitization.
This multicenter, open-label, randomized, controlled trial recruited nondiabetic people with HIV taking a stable antiretroviral regimen and with evidence of NAFLD (waist >/= 94 cm in men, >/= 80 cm in women, or 1 or more abnormal liver function tests with no other cause, or radiologic or biopsy evidence). Participants had to be at least 35 years old, have HIV infection for at least 5 years, and have a viral load below 50 copies for at least 1 year. Researchers randomized them in equal proportions to twice-daily maraviroc (with dose adjusted to the antiretroviral regimen), 500 mg of metformin twice daily, maraviroc plus metformin, or antiretroviral therapy alone (the control arm).
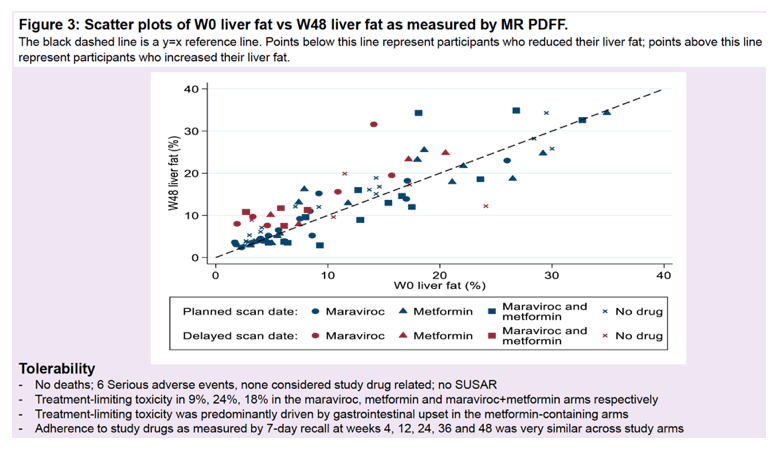
At weeks 0 and 48 the researchers used magnetic resonance proton density fat fraction (MR-PDFF) to assess fat content because it is winning favor as the gold standard for gauging liver fat and because results can be converted into the Clinical Research Network (CRN) steatosis grade. The primary endpoint was change in percentage of MR-PDFF-measured liver fat over the 48 weeks of treatment.
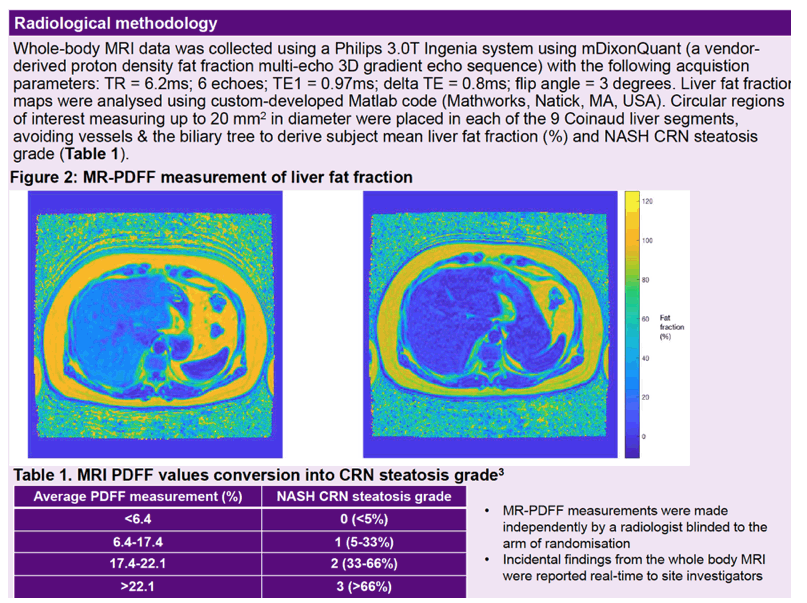
Ninety participants enrolled from 6 London sites. Similar proportions across the four study arms-and 70% overall-met entry requirements with a scan or biopsy diagnosis plus at least one other criterion. The study group was mostly male (93%) and white (81%) and had a median age of 52 (interquartile range [IQR] 47 to 57) and a median body mass index of 28 kg/m2 (IQR 26 to 33), which is in the overweight range. Median MR-PDFF liver fat fraction was only 8.9%, and 78% of participants had grade 1 or lower hepatic steatosis.
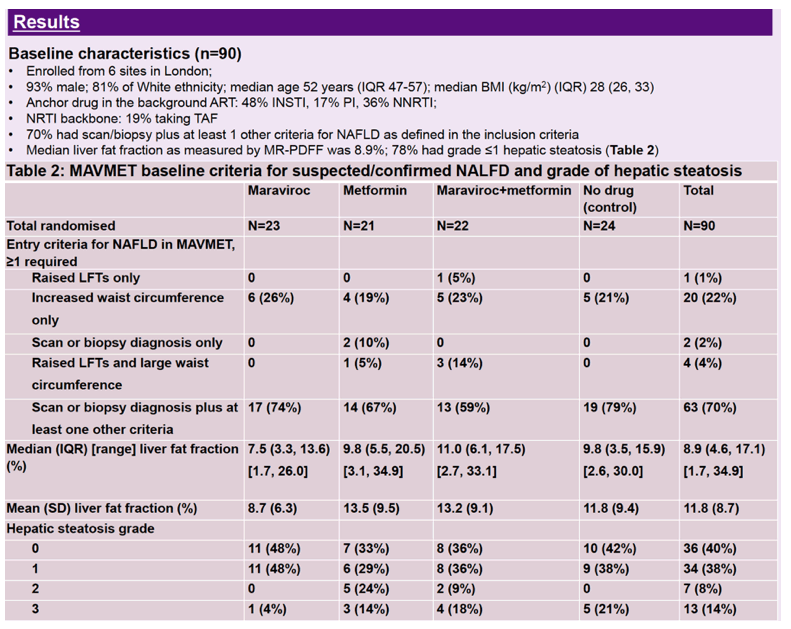
Linear regression analysis adjusted for pandemic-related delays in week-48 scans showed a small rise in percentage of liver fat in all study arms: 2.2% with maraviroc, 1.3% with metformin, 0.8% with both drugs, 1.4% with antiretroviral therapy alone, and 1.4% overall. The 48-week absolute change or relative change in liver fat percentage did not differ significantly between either the maraviroc arm or the metformin arm and the antiretroviral-only control arm. And there was no evidence of an interaction between maraviroc and metformin for absolute change or relative change in liver fat percentage. Hepatic steatosis increased in 25% of participants, stayed the same in 68%, and decreased in 7%.
For 20 participants treated more than 48 weeks, longer treatment appeared to increase liver fat percentage compared with only 48 weeks of treatment, but the small number of participants treated more than 48 weeks makes it difficult to draw any conclusions from that finding.
The lower-than-expected baseline liver fat percentage in this study group suggested to the researchers that surrogate markers of NAFLD they used were not sensitive to detecting people with higher liver fat percentages. In any event, 48 weeks of maraviroc, metformin, or both drugs did not trim liver fat in people with HIV compared with no therapy as measured by MR-PDFF.
Reference
1. Pett S, McCabe L, Latifoltojar A, et al. Results of MAVMET, a multi-centre, open-label, 48-week randomised controlled trial of maraviroc with or without metformin for the treatment of non-alcoholic fatty liver disease in HIV-positive virologically suppressed adults. 18th European AIDS Conference, EACS 2021, October 27-30, 2021, London. Abstract BPD3/4.


|
| |
|
 |
 |
|
|