 |
 |
 |
| |
Efficacy of switching to bictegravir/emtricitabine/tenofovir
alafenamide in patients with pre-existing nrti resistances: real world data
|
| |
| |
EACS 2021
R. Micán1, A. de Gea Grela1, C. Busca1, J.I. Bernardino1, R. Montejano1, M. Montes1, J. Cardiñanos1, R. de Miguel1, L. Serrano1, I. Pérez-Valero2, E. Valencia1, V. Moreno1, J.R. Arribas1, J. González-García1, L. Martín-Carbonero1
1Hospital Universitario La Paz/IdiPAZ, Madrid, Spain, 2Hospital Universitario Infanta Sofía, Córdoba, Spain
Purpose: The use of Bictegravir/Emtricitabine/Tenofovir alafenamide (B/F/TAF) has demonstrated to be effective and durable in suppressed PLWH, including those with pre-existing M184V/I or other NRTI resistances. However, these data are from clinical trials. The impact of archive resistance in B/F/TAF switching strategies in real life is unknown.
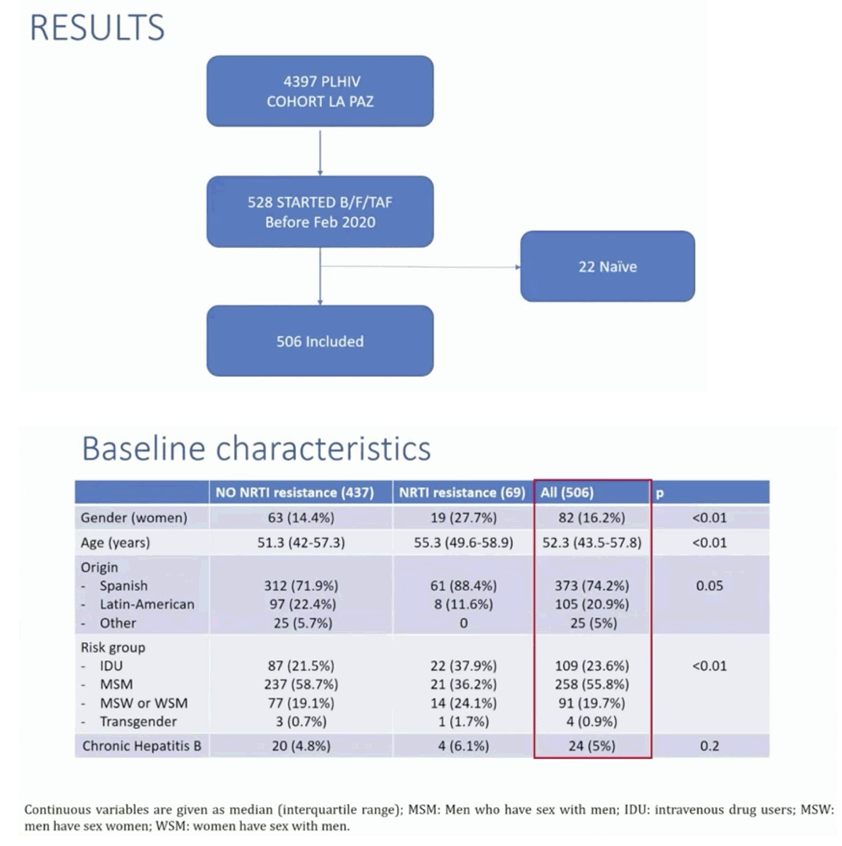
Method: Retrospective review of all patients who switched to B/F/TAF in Hospital La Paz (Madrid) before February 2020. Demographics, clinical, laboratory and historic pre-existing antiretroviral resistance were extracted from our database. Response rates (RR) (VL<50 cp/ml) were analyzed in 48 weeks in an intent to treat (ITT) analysis: missing=failure and on treatment (OTT) analysis: patients who continue with B/F/TAF or virological failure switches. Comparisons on ITT and OTT analysis were performed in patients with and without NRTI archived resistances.
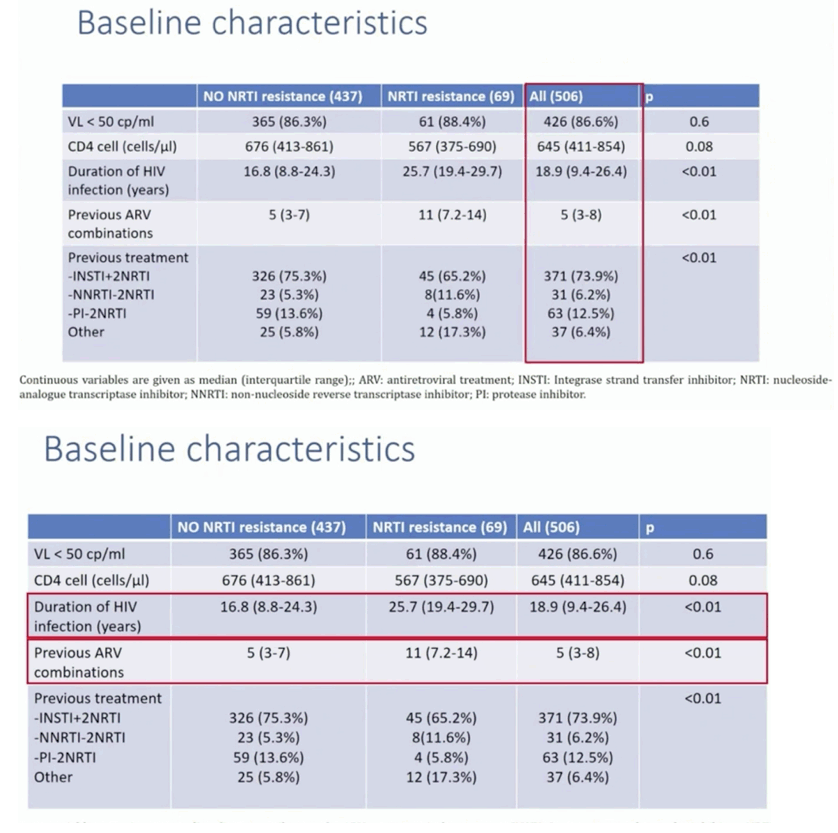
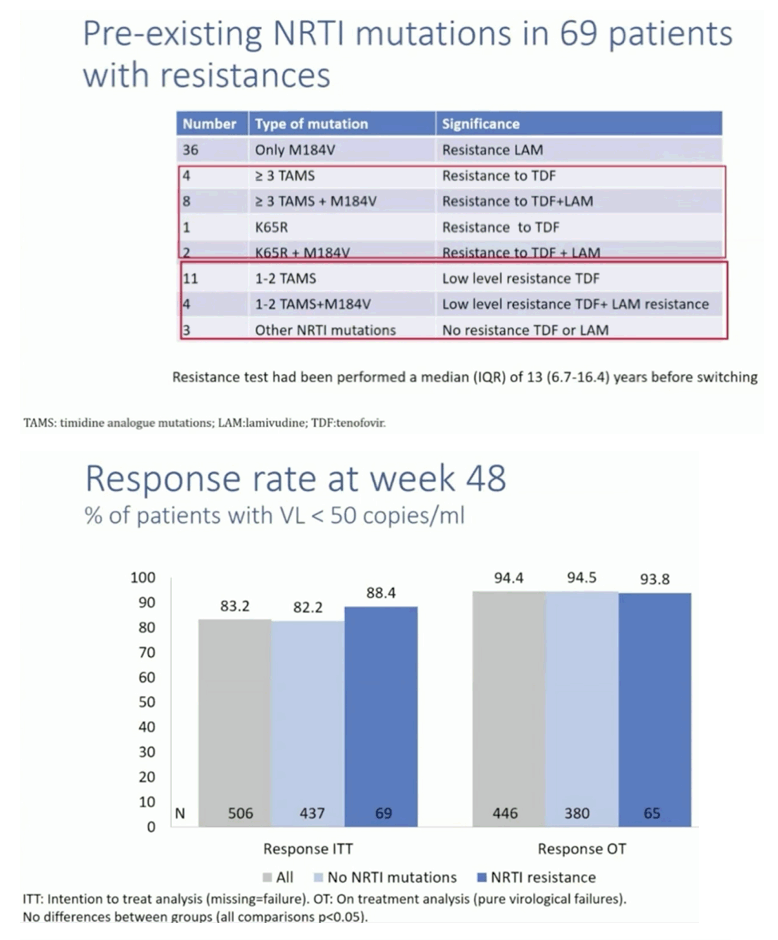
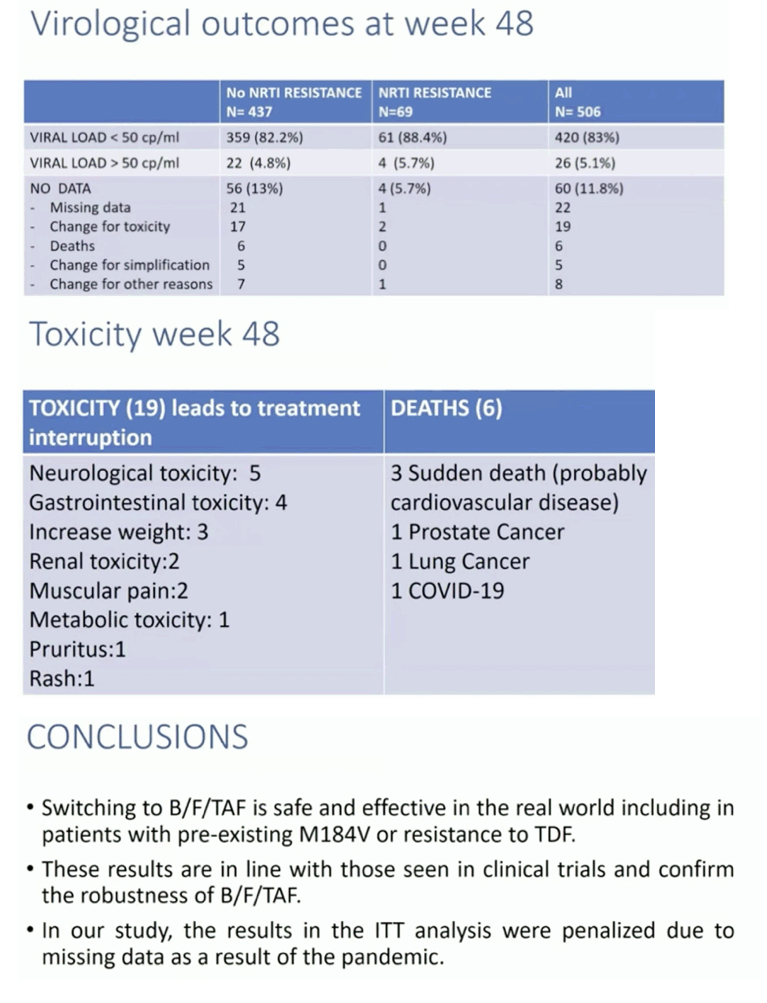
Results: A total of 528 patients were included. Table 1 shows patient characteristics. Overall 84 (15.9%) were women. Median age and median time of HIV infection were 51 and 17.9 years, respectively. Median previous antiretroviral combinations were 5. Overall 69 (13.1%) patients had documented pre-existing NRTI resistance, 57 (10.8%) had M184V/I, 15 (2.8%) had K65R and 15 had changes that confer low-level resistance to TDF. At 48 weeks, 63 patients (11.9%) discontinued B/F/TAF (23 lost visit [4.3%]; 19 toxicities [3.6%]; 6 exitus [1.1%], 5 simplification [0.9%], 2 virological failure [0.3%], 8 other). RR at 48 weeks were 83.3% (440/528) and 94.4% (439/465) in the ITT and OTT, respectively. Patients with pre-existing 184V/I, TDF or other NRTI resistances showed similar RR than those without these mutations (figure 1).
Conclusions: We demonstrate that switching to B/F/TAF is safe and effective in the real-world including patients with pre-existing NRTI mutations like M184V and K65R. These results confirm the robustness of this combination. Results on ITT are penalized for lost visits due to the pandemic.


|
| |
|
 |
 |
|
|