| |
Treatment and Renal Outcomes Up to 96 Weeks After Tenofovir Alafenamide
Switch From Tenofovir Disoproxil Fumarate in Routine Practice
|
| |
| |
Download the PDF here
In this large, multicenter, real-world study of patients with CHB on TAF sequential therapy after receiving TDF for close to 5 years, we found continued increase in viral, biochemical, and complete response rates up to 96 weeks after switching from TDF to TAF as well as stable levels of mean eGFR following adjustment for age, sex, baseline eGFR, and diabetes, hypertension, or cirrhosis. These findings of continued improvement in viral suppression and complete response obtained with TAF treatment are in line with other reports, adding strength to the evidence that TAF also provides excellent therapeutic results in routine clinical practice.(10-16)
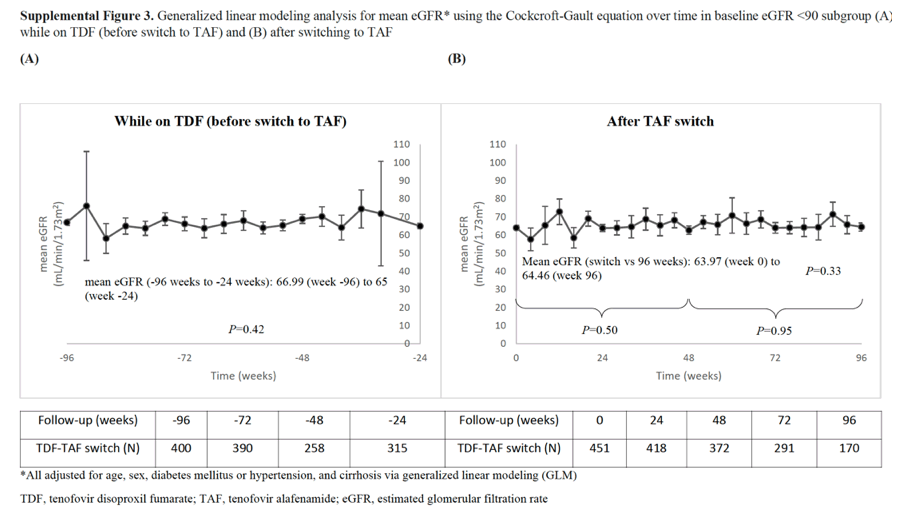
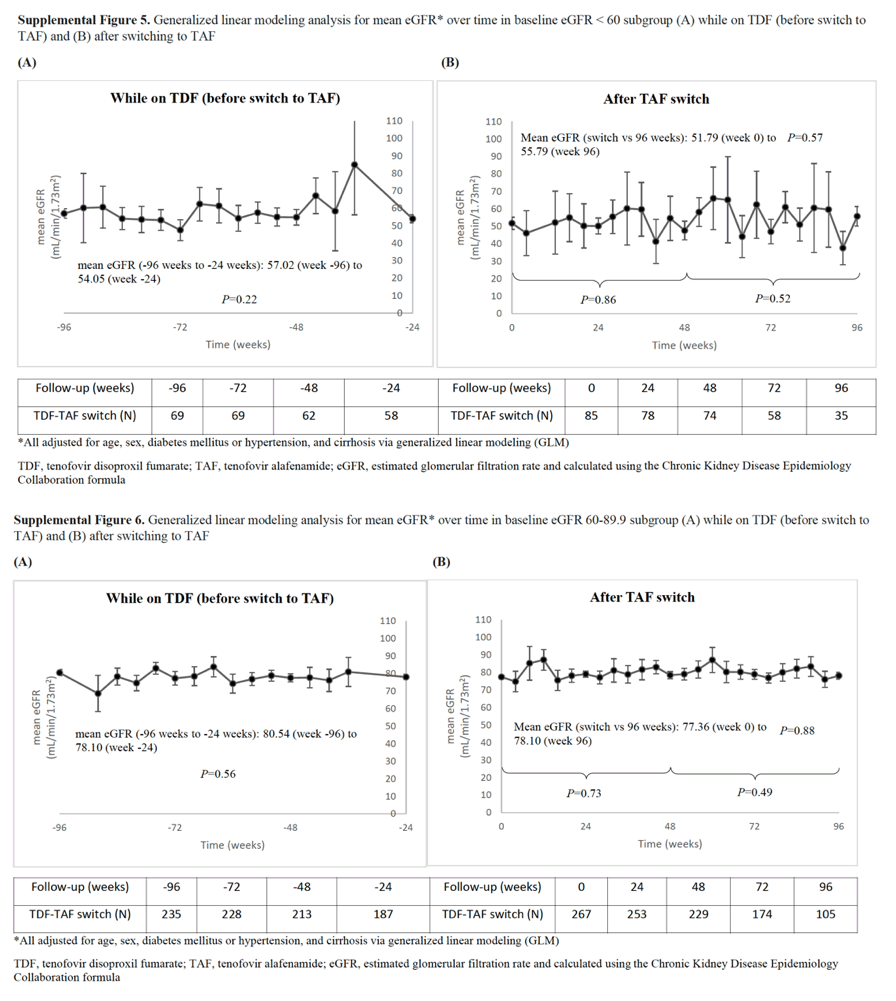
With regard to renal function, we found that the adjusted mean eGFR remained stable while on TDF and after the switch to TAF for patients with normal renal function. However, we noted that among those with eGFR < 90, while the adjusted mean was stable after switch to TAF, it had decreased significantly while on TDF prior to switch. These findings suggest that there may be a renal advantage with TAF compared to TDF; however, further research is needed to validate this point.
Changes in CKD Stages over Time
When we inspected the cohort by CKD stages at the switch point and their evolution over the 96-week follow-up (Fig. 2), we found that 55 of 267 (20.6%) CKD stage 2 (mild renal impairment, eGFR < 90 but ≥60 mL/min/1.73 m2) patients at switch regressed to CKD stage 1, signifying return to normal renal function and that 30 of 85 (35.3%) CKD stage 3-5 (eGFR < 60 mL/min/1.73 m2) patients regressed to CKD stage 2 and 1 (1.2%) to normal renal function. At the same time, among those with stage 1 (normal renal function, eGFR ≥ 90 mL/min/1.73 m2) disease at the switch (n = 463), 52 (11.2%) progressed to CKD stage 2 and 2 (0.4%) progressed to CKD stage 3-5. Patients who improved at least one CKD stage were younger (59.1 ± 12.4 vs. 63.2 ± 12.6 years, P = 0.008). When assessing CKD stage migration at the end of 48 weeks of follow-up, we found similar patterns: 51 of 267 (19.1%) CKD stage 2 patients at switch regressed to stage 1, and 26 of 85 CKD stage 3-5 patients at switch regressed to stage 2 (Supporting Fig. S7A).
We also determined the proportion of patients with <20% change and ≥20% change in eGFR over the study observation up to 96 weeks after switch to TAF in the total cohort (Fig. 3). The largest changes were observed in patients with CKD stage 3-5 at switch, 27.1% (23/85) of whom experienced ≥20% increase in eGFR, while 9.4% (8/85) experienced ≥20% decrease in eGFR.
In the sensitivity analysis of the TDF treatment-naive cohort, over the 96 weeks following TAF switch, we also found that 23% (29/126) of CKD stage 2 patients at switch point regressed to CKD stage 1 (5.6% [7/126] to stage 3-5), 41.5% (17/41) CKD stage 3-5 patients regressed to CKD stage 2, and 1 (2.4%) CKD stage 3-5 patients to stage 1, while among those with CKD stage 1 disease at the switch (n = 238), 27 (11.3%) progressed to stage 2 but none to stages 3-5 (Supporting Fig. S8). At the end of follow-up at 48 weeks, 22.2% (28/126) stage 2 patients at switch regressed to stage 1, and 41.5% (17/41) stage 3-5 patients regressed to stage 2. (Supporting Fig. S7B).
Factors Associated with Changes in eGFR
Using GEE analysis adjusting for age, ethnicity (Asian vs. non-Asian), sex, hypertension or diabetes mellitus or cirrhosis, and baseline eGFR (Table 3), we found that older age (coefficient, -0.43; 95% CI, -0.48 to -0.37; P < 0.001), being male (coefficient, -3.00; 95% CI, -4.32 to -1.67; P < 0.001), having a baseline eGFR of 60-89 (coefficient, -17.88; 95% CI, -19.48 to -16.29; P < 0.001), or a baseline eGFR < 60 (coefficient, -39.30; 95% CI, -41.83 to -36.76; P < 0.001) were associated with decreasing eGFR levels after switching to TAF over the 96 weeks of follow-up after switch (Table 3A) but not ethnicity (coefficient, -1.001; 95% CI, -3.10 to 1.10; P = 0.35), which was also not significant on univariable analysis (Asian vs. non-Asian, coefficient, -0.58; 95% CI, -4.97 to 3.82; P = 0.80). Analysis with the same model up to 48 weeks of follow-up postswitch (Table 3B) and between 48 weeks and 96 weeks postswitch (Table 3C) showed similar results.
These findings were similar for the treatment-naive group as being older (coefficient, -0.44; 95% CI, -0.52 to -0.36; P < 0.001), male (coefficient, -3.30; 95% CI, -5.11 to -1.49; P < 0.001), having a baseline eGFR of 60-89 (coefficient, -18.06; 95% CI, -20.25 to -15.86; P < 0.001), and having a baseline eGFR < 60 (coefficient, -39.75; 95% CI, -39.75 to -32.76; P < 0.001) were all associated with decreasing eGFR.
EASL 2029, Lampertico et al - A Phase 3 Study Comparing Switching From Tenofovir Disoproxil Fumarate to Tenofovir Alafenamide With Continued TDF Treatment in Virologically Suppressed Patients With Chronic Hepatitis B: Week 48 Efficacy and Safety Results
Lampertico P, Buti M, Fung S, Ahn SH, Chuang W-L, Tak WY, et al. Switching from tenofovir disoproxil fumarate to tenofovir alafenamide in virologically suppressed patients with chronic hepatitis B: a randomised, double-blind, phase 3, multicentre non-inferiority study. Lancet Gastroenterol Hepatol 2020; 5: 441- 453.
Participants in this study were enrolled between Dec 29, 2016, and Oct 20, 2017. 541 patients were screened and 490 patients were randomly assigned to switch to tenofovir alafenamide or to stay on tenofovir disoproxil fumarate. Two patients assigned to receive tenofovir alafenamide did not receive treatment; thus the full analysis set for efficacy and safety analyses consisted of 243 patients in the tenofovir alafenamide group and 245 in the tenofovir disoproxil fumarate group. At week 48, one patient from each treatment group (both <1%) had HBV DNA of at least 20 IU/mL (difference in proportion 0·0%, 95% CI -1·9 to 2·0), thereby showing non-inferior efficacy of tenofovir alafenamide to tenofovir disoproxil fumarate. Patients who received tenofovir alafenamide had significantly increased bone mineral density at hip (mean change 0·66% [SD 2·08] vs -0·51% [SD 1·91]; difference in least square means 1·17% [95% CI 0·80 to 1·54; p<0·0001]) and at spine (mean change 1·74% [3·46] vs -0·11% [3·13]; difference in least square means 1·85% [1·24 to 2·46; p<0·0001]), creatinine clearance by Cockcroft-Gault relative to tenofovir disoproxil fumarate (median change 0·94 mL/min [IQR -4·47 to 6·24] vs -2·74 mL/min [-7·89 to 1·88]; p <0·0001), and improved markers of bone turnover and tubular function at week 48. The most common treatment-emergent adverse events were upper respiratory tract infection (18 [7%] of 243 patients in the tenofovir alafenamide group and 16 [7%] of 245 patients in the tenofovir disoproxil fumarate group) and nasopharyngitis (13 [5%] of 243 patients in the tenofovir alafenamide group and 12 [5%] of 245 patients in the tenofovir disoproxil fumarate group). The incidence of grade 3 and above adverse events and serious adverse events was low and similar between groups. No viral resistance was observed in patients who qualified for viral sequencing.
AASLD 2019 - Safety and Efficacy at 48 Weeks After Switching From Tenofovir Disoproxil Fumarate to Tenofovir Alafenamide in Chronic HBV Patients With Risk Factors for TDF Use
Safety and Efficacy of Switching From Tenofovir Disoproxil Fumarate to Tenofovir Alafenamide in People With HIV Aged 50 Years and Older
--------------------------------------
Treatment and Renal Outcomes Up to 96 Weeks After Tenofovir Alafenamide Switch From Tenofovir Disoproxil Fumarate in Routine Practice
11 March 2021 - Hidenori Toyoda,1 Jennifer Leong,2 Charles Landis ,3 Masanori Atsukawa,4 Tsunamasa Watanabe,5 Daniel Q. Huang,6,7 Joanne Liu,3,8 Sabrina Xin Zi Quek,9 Toru Ishikawa,10 Taeang Arai,4 Keisuke Yokohama,11 Makoto Chuma,12 Koichi Takaguchi,13 Haruki Uojima,14 Tomonori Senoo,13 Hansen Dang,15 Mayumi Maeda,15 Joseph Hoang,15 Richard H. Le,15 Satoshi Yasuda,1 Khin N. Thin,15 Sally Tran,15 Nicholas Chien,15 Linda Henry,15 Akira Asai ,11 Shinya Fukunishi,16 Ramsey Cheung,15,17 Seng Gee Lim ,6,7 Huy N. Trinh,18 and Mindie H. Nguyen
Abstract
Background and Aims
Real-world data for treatment effectiveness and renal outcomes in chronic hepatitis B (CHB) patients who were switched to the new and safer prodrug tenofovir alafenamide (TAF) from tenofovir disoproxil fumarate (TDF) are limited. Therefore, we aimed to evaluate treatment and renal outcomes of this population.
Approach and Results
We analyzed 834 patients with CHB previously treated with TDF for ≥12 months who were switched to TAF in routine practice at 13 US and Asian centers for changes in viral (HBV DNA < 20 IU/mL), biochemical (alanine aminotransferase [ALT] < 35/25 U/L for male/female), and complete (viral+biochemical) responses, as well as estimated glomerular filtration rate (eGFR; milliliters per minute per 1.73 square meters) up to 96 weeks after switch.
Viral suppression (P < 0.001) and ALT normalization (P = 0.003) rates increased significantly after switch, with a trend for increasing complete response (Ptrend = 0.004), while the eGFR trend (Ptrend > 0.44) or mean eGFR (P > 0.83, adjusted for age, sex, baseline eGFR, and diabetes, hypertension, or cirrhosis by generalized linear modeling) remained stable.
However, among those with baseline eGFR < 90 (chronic kidney disease [CKD] stage ≥2), mean eGFR decreased significantly while on TDF (P = 0.029) but not after TAF switch (P = 0.90). By week 96, 21% (55/267) of patients with CKD stage 2 at switch improved to stage 1 and 35% (30/85) of CKD stage 3-5 patients improved to stage 2 and 1.2% (1/85) to stage 1.
Conclusions
Overall, we observed continued improvement in virologic response, ALT normalization, and no significant changes in eGFR following switch to TAF from TDF.
HBV continues to be pervasive throughout the world despite the ongoing global initiative to eliminate viral hepatitis by the year 2030.(1-4) Currently, there are no curative treatments for HBV, but there are medications that can suppress the virus and help to slow the progression of chronic hepatitis B (CHB).(5) As such, suppression of HBV remains one of the pillars of CHB management.(6) Tenofovir disoproxil fumarate (TDF) and entecavir (ETV) have been the mainstays of therapy over the past decade; however, each is associated with side effects including renal impairment and bone loss(7, 8) with TDF and potential viral resistance with ETV, especially in those with a history of resistance to the older nucleoside analogue lamivudine.(9) More recently, an HBV viral suppression drug was approved, tenofovir alafenamide (TAF).
TAF is a prodrug of tenofovir (TFV), which has shown potent inhibition of HBV replication at a low dose, with high intracellular concentration and >90% lower systemic TFV concentration than TDF.(10, 11) In two randomized, double-blind, multinational, phase 3, noninferiority trials for HBeAg-positive and HBeAg-negative patients, TAF 25 mg orally once-daily was associated with a significantly higher alanine aminotransferase (ALT) normalization rate (male, ALT ≤ 30 U/L; female, ALT ≤ 19 U/L) than was seen with TDF at 48 weeks without incurring adverse renal and bone events at a longer follow-up period of 96 weeks.(10, 12-14)Regarding outcomes of patients switched to TAF from TDF, one small, prospective, single-arm, open-label clinical trial of 75 patients with CHB previously treated with TDF and with HBV DNA < 21 IU/mL who were switched to TAF found improved bone and renal tubular markers by end of study follow-up at 24 weeks.(13) Since then, a randomized, double-blind, phase 3, multicenter, noninferiority trial comparing virologically suppressed patients with CHB who were switched from TDF to TAF (n = 243) or continued on TDF (n = 245) also found improved bone and renal markers at week 48 without a loss in efficacy.(15) However, real-world data and longer follow-up are necessary to determine the clinical relevance of the differences in response to TAF in comparison with TDF for patients being managed in routine clinical practice, but these data are limited. To the best of our knowledge, there has been only one published real-world study, which included 36 patients switched to TAF from TDF in routine practice who were followed for only 24 weeks after switch to TAF.(16) Therefore, we aimed to examine the effectiveness and renal outcomes of sequential therapy with TAF in a large cohort of patients with CHB from routine practice who had received TDF for at least 12 months at 13 clinical centers in the United States and Asia. Specifically, we determined the viral, biochemical, and complete response rates as well as the changes in estimated glomerular filtration rate (eGFR) and chronic kidney disease (CKD) stages up to 96 weeks after switching to TAF.
|
|
| |
| |
|
|
|