| |
Safety and effectiveness of up to 3 years' bulevirtide monotherapy in patients with HDV-related cirrhosis
|
| |
| |
Download the PDF here
Chronic hepatitis Delta (CHD) is a rare but severe form of chronic viral hepatitis that affects approximately 12-72 million patients worldwide.[1] It is sustained by the hepatitis D virus (HDV), a small defective virus that requires the helper function of HBV to replicate and propagate.[2] In the last 30 years, the only therapeutic approach has been the off-label use of a 48-week course of Interferon (IFNα).[3] However, this antiviral strategy is characterized by limited off-therapy responses and an unfavorable safety profile.[3],[4]
To our knowledge this is the first report assessing the safety, effectiveness and clinical response of BLV administered as monotherapy for up to three years in patients with HDV-related compensated cirrhosis in whom Peg-IFN therapy was contraindicated. The long-term administration of BLV monotherapy in these difficult to treat patients was associated with positive virological and clinical results coupled with a favorable safety profile. All patients reported excellent compliance to BLV therapy (2 subcutaneous injections every morning), as confirmed by the increase of bile salts levels, a biomarker of target engagement.
One of the most important findings of the study is the positive virological and biochemical response observed as all three patients achieved and maintained undetectable HDV-RNA, which was tested by a very sensitive assay. These findings are unprecedented for at least three reasons: first, no studies to date have reported the outcome of BLV treatment beyond week 48; second, no data exists to demonstrate that virological response was maintained upon dose reduction; third, the effectiveness of the administration of this drug for such a long-term in difficult to treat patients such as those with advanced compensated cirrhosis, including one case with clinical significant portal hypertension (CSPH), has never been reported so far.
Another important finding was the clinical response that we have observed in these patients. One patient who had compensated cirrhosis with CSPH, showed a clinically relevant improvement of liver function tests, AFP and platelet levels, as well as portal hypertension features, with the regression of EV.
EASL: Safety and Efficacy of Bulevirtide Monotherapy and in Combination with Peginterferon Alfa-2a in Patients with Chronic Hepatitis Delta: 24 Weeks Interim Data of MYR204 Phase 2b Study - (06/27/21)
EASL: Bulevirtide Monotherapy at Low and High Doses in Patients With Chronic Hepatitis Delta: 24-Week Interim Data of the Phase 3 MYR301 Study - (06/24/21)
EASL: Bulevirtide With or Without PEG-IFN Shows 24-Week Activity Against Hep D - Mark Mascolini (06/27/21)
------------------
Published:October 23, 2021 Alessandro Loglio, Peter Ferenci, Sara Colonia Uceda Renteria, Christine Y.L.Tham, Caroline Scholtes, Heidemarie Holzmann, Florian van Bömmel, Marta Borghi,Riccardo Perbellini, Alessandro Rimondi, Elisa Farina, Elena Trombetta, Maria Manunta, Laura Porretti, Daniele Prati, Ferruccio Ceriotti, Fabien Zoulim, Antonio Bertoletti, Pietro Lampertico
Abstract
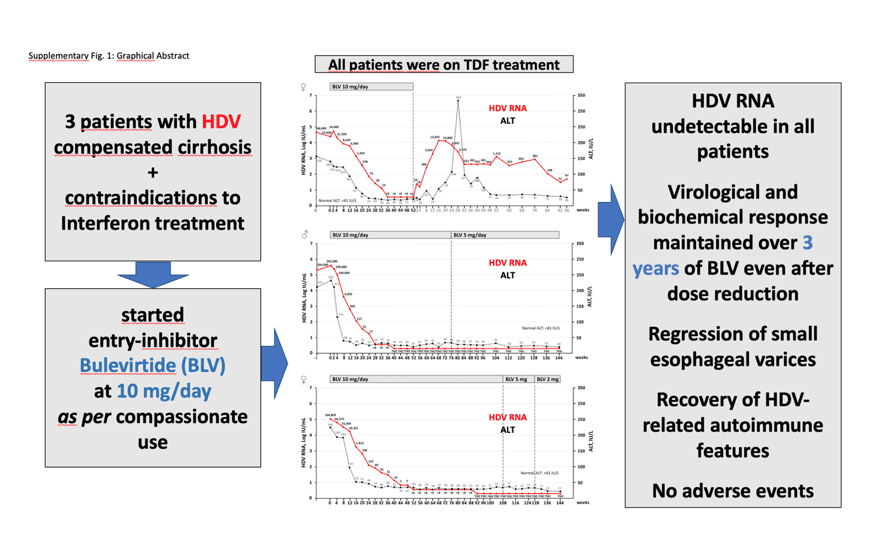
The entry-inhibitor Bulevirtide (BLV) received conditional approval by EMA in July 2020 for the treatment of adult patients with compensated chronic hepatitis Delta. However, the effectiveness and safety of BLV administered as monotherapy beyond 48 weeks in difficult to treat HDV cirrhotic patients is presently unknown. Here we describe the first patients with HDV-related compensated cirrhosis who were treated with BLV (10 mg/day as a starting dose) for up to three years as compassionate use. Patients were also monitored for HBcrAg and HBV-RNA levels and HDV and HBV specific T-cells markers.
In the patient who stopped BLV at week 48 after achieving a virological and biochemical response, the initial virological and biochemical rebound was followed by ALT normalization coupled with low HDV-RNA and HBsAg levels. In the two patients treated continuously for 3 years, virological and biochemical responses were maintained throughout the treatment period even after dose reduction. In a patient with advanced compensated cirrhosis, liver function tests significantly improved, esophageal varices disappeared, and histological/lab features of autoimmune hepatitis resolved. Overall, no safety issues were recorded, as bile salt increase was asymptomatic. While serum HBV-RNA levels remained undetectable in all patients, HBcrAg levels showed a progressive, yet modest decline during long-term BLV-treatment. No HDV-specific Interferon-γ producing T-cells were detected, neither after HDV reactivation (after BLV withdrawn in Patient 1) nor during 3 years of BLV treatment. In conclusion, this report shows that continuous administration of BLV monotherapy for three years provides excellent virological and clinical response in HDV cirrhotic patients who had contraindications to IFN-based therapies.
Lay Summary
HDV-RNA levels became undetectable, and ALT normalized in all three patients treated with Bulevirtide (BLV). Virological and biochemical responses were maintained even after dose reduction.- Improvement of liver function tests, regression of esophageal varices and recovery of HDV-related autoimmune disease were documented in the male cirrhotic patient long-term treated with BLV.- An asymptomatic increase of bile acids was the only drug-related clinical adverse event.
Introduction
Chronic hepatitis Delta (CHD) is a rare but severe form of chronic viral hepatitis that affects approximately 12-72 million patients worldwide.[1] It is sustained by the hepatitis D virus (HDV), a small defective virus that requires the helper function of HBV to replicate and propagate.[2] In the last 30 years, the only therapeutic approach has been the off-label use of a 48-week course of Interferon (IFNα).[3] However, this antiviral strategy is characterized by limited off-therapy responses and an unfavorable safety profile.[3],[4]
The unmet medical need of an effective and safe therapeutic option for CHD patients coupled with the recent identification of the entry receptor for HBV and HDV, has fostered the research in this field. Among the several compounds now being tested in clinical studies, Bulevirtide (BLV) is the only drug that has received a conditional approval from the European Medicine Agency in July 2020, at the dose of 2 mg/day s.c.[4] Previously named Myrcludex-B and now commercialized as Hepcludex® in European Union, BLV is a first in class entry-inhibitor of HBV, a subcutaneously delivered lipopeptide that mimics the Na+-taurocholate co-transporting polypeptide (NTCP) receptor binding domain, blocking the HDV/HBV entry exclusively in liver cells. In Phase II trials, BLV administration induced HDV-RNA reduction and ALT improvement during a 24- and 48-week treatment that was however followed by a relapse after treatment cessation in most patients. When combined with Peg-IFNα, a synergic effect on HDV-RNA and HBsAg decline was demonstrated.[5]
In this brief report we describe for the first time the safety, effectiveness and clinical response to BLV administered for up to 3 years as a monotherapy in patients with HDV-related compensated cirrhosis. In addition, we enriched the study by an integrated analysis of both standard and innovative HBV markers, such as serum HBV core-related Antigen (HBcrAg) and HBV-RNA, coupled with an immunological monitoring of IFN-γ HDV and HBV specific T-cells.
Results
Patient 1
A 69 year-old, Caucasian, HBeAg-negative female with HDV-related compensated cirrhosis complicated by portal hypertension (splenomegaly and thrombocytopenia) that contraindicated IFN therapy. Comorbidities included diabetes mellitus, femoral and lumbar osteopenia, mild arterial hypertension and uterine polyps under assessment. This patient had genotype D of HBV and genotype 1 of HDV; she was treated with TDF.
During 10 mg/day BLV treatment, ALT rapidly normalized, HDV-RNA became undetectable and clinical parameters improved (Figure-Panel A, Supplementary Table). Following the diagnosis of an endometrial carcinoma at week 52, BLV was withdrawn as a precautious measure to avoid any possible drug-to-drug interaction with chemotherapy. After BLV discontinuation, HDV-RNA rapidly rebounded and ALT flared up peaking at week 28 (333 IU/L), but both markers fell within the normal range by week 48 off-therapy (Figure-Panel A, Supplementary Table). HDV reactivation was not associated to any sign of clinical decompensation. HBsAg levels, that had slightly increased during BLV treatment, progressively and significantly declined after BLV was withdrawn, paralleling HDV-RNA rebound and ALT flare (Figure-Panel A). In summary, at 96-week off-therapy, HBsAg and HDV-RNA levels were very low and ALT normal. No significant increases in HDV or HBV specific IFN-γ T-cell response was observed neither during BLV treatment nor during virological and clinical relapse, nor HBcrAg levels that remained always below the LOD except at BLV baseline and at ALT peak secondary to HDV relapse (Figure-Panel A).
Bile acids, that increased during BLV administration, rapidly normalized after drug withdrawal. The asymptomatic increase of serum bile acids levels >160 μmol/l that we observed at week 20 and 24 was likely due to the fact that BLV was self-administered before blood sampling, and not after as usually done.
Patient 2
The second patient was a 51 year-old male, Caucasian, HBeAg-negative with HDV-compensated cirrhosis complicated by thrombocytopenia, small esophageal varices (EV) and autoimmune hepatitis (liver biopsy performed in 2010) that contraindicated IFN-treatment. [6]
He had diabetes on diet therapy (Table). This patient, on TDF therapy, had genotype D of HBV and genotype 1 of HDV. BLV dose: In Patient 2, BLV dose was reduced from 10 to 5 mg/day at week 76; in Patient 3, BLV dose was reduced from 10 to 5 mg/day at week 108 and to 2 mg/day at week 128. Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, gamma-glutamyl transferase; pCHE, pseudo-cholinesterase; CAP, Controlled Attenuation Parameter. Reference values: AST, 10-33 U/L; ALT, 6-41 U/L; ALP, 35-104 U/L; GGT, 5-36 U/L; Total bilirubin, 0.12-1.10 mg/dl; pCHE, 4,200-11,250 U/L; Albumin, 3.4-4.8 g/dl; IgG, 700-1600 mg/dL; INR, 0.84-1.20; White cells, 4,800-10,800/mmc; Hemoglobin, 12-16 g/dl; Platelet, 130-400 x109/L; Creatinine, 0.5-1.0 mg/dl; Glycemia, 70-110 mg/dL; Triglycerides, <150 mg/dL; Total cholesterol, <190 mg/dL; HDL, >45 mg/dL.
Treatment with BLV 10 mg/day induced a rapid biochemical and virological responses that was maintained up to week 144 without any sign of virological or biochemical breakthrough even after BLV dose reduction to 5 mg/day at week 76 (Figure-Panel B): we decided to reduce BLV daily dose only to simplify therapy, i.e. only one injection every morning, given the excellent HDV suppression lasting for >1 year. During BLV treatment, liver function tests as well as alpha-fetoprotein (AFP) and IgG levels rapidly normalized, platelet count improved to almost normal levels, while HBsAg levels and the other HBV markers remained stable over time. Serum HBV-RNA levels remained undetectable while HBcrAg levels showed a progressive, yet modest decline (from 4.5 to 3.4 Log U/mL). In a liver biopsy performed after 72 weeks of BLV treatment, we observed a reduction of plasma cells infiltration compared to what had been observed in the previous liver biopsy performed in 2010, with an improvement of histological features of autoimmune hepatitis.
Notably, platelet count significantly increased, liver stiffness improved, and esophageal varices disappeared after 20 months of BLV treatment (Table). The latter finding was confirmed in two consecutive endoscopies 12-months apart. No recovery of HDV nor HBV specific IFN-γ producing T-cells function was observed at any time point during BLV administration (Figure-Panel B). BLV was well-tolerated as no local or systemic adverse events were reported, the increase of bile acids levels was dose-related and fully asymptomatic.
Patient 3
The third patient was a 58 year-old female, native to Uzbekistan, HBeAg-negative with HDV-related compensated cirrhosis complicated by an autoimmune thrombocytopenia with detectable anti-platelet antibodies for which she was treated in the past with high doses of steroids (Table). This patient, on anti-HBV therapy with TDF, had genotype D of HBV and genotype 1 of HDV.
BLV administration led to a rapid biochemical response that was followed by a virological response with HDV-RNA becoming undetectable at week 52, both responses were maintained up to week 144 despite BLV dose reduction to 5 and 2 mg/day (Figure-Panel C). Liver function tests as well as HBV markers remained stable over time while HBcrAg levels showed a progressive but modest decline (Table, Figure-Panel C). No local or systemic side effects were recorded apart from a dose-related, fully asymptomatic increase of bile acid, that had a decrease from a mean level of 203 to 152 umol/L after BLV dose reduction (10→5 mg/day). Autoimmune thrombocytopenia did not recur.
|
|
| |
| |
|
|
|