| |
Medicaid Subscription-Based Payment Models
and Implications for Access to Hepatitis C Medications
|
| |
| |
Download the PDF here
In the year preceding SBPM implementation, the mean (SD) rate of quarterly HCV prescription fills per 100 000 Medicaid enrollees was 43.1 (8.6) prescriptions in Louisiana and 50.1 (4.1) in Washington. After SBPM implementation, the mean (SD) rate of quarterly HCV prescription fills per 100 000 enrollees was 206.0 (51.2) prescriptions in Louisiana and 53.9 (11.0) in Washington. In this cross-sectional study, Louisiana experienced substantial increases in HCV medication use among its Medicaid-enrolled population following SBPM implementation, whereas Washington did not. These differences may partially be explained by state-level variation in SBPM implementation, historical restrictions on access to HCV medications, and responses to the COVID-19 pandemic. (or, did Wash State just not provide but withhold adequate resources to support screening & other services required to increase treatment & cure, Jules)
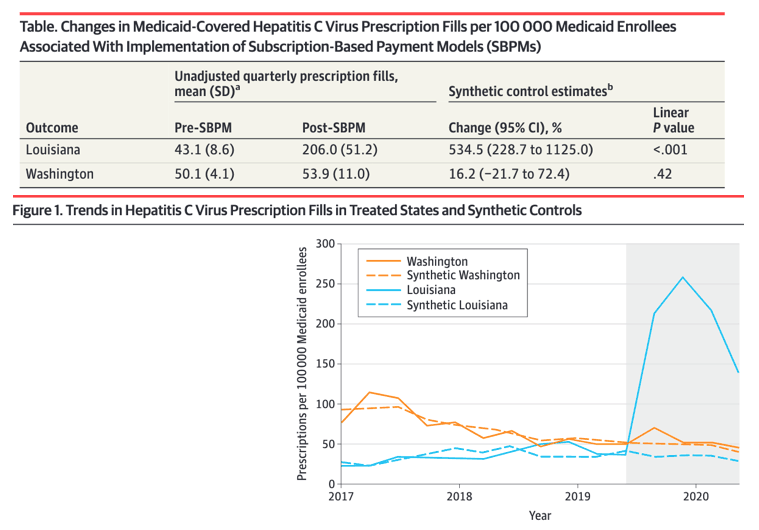
In synthetic control models, SBPM implementation in Louisiana was associated with an increase of 173.5 (95% CI, 74.3-265.3) quarterly prescription fills per 100 000 Medicaid enrollees during the following year, a relative increase of 534.5% (95% CI, 228.7%-1125.0%). Washington did not experience a significant change in prescription fills following SBPM implementation.
It is also plausible that the spending cap of the SBPM enabled the change in access criteria in Louisiana. The spending cap removes states’ incentives to ration access to HCV medications, which may encourage the adoption of policies that facilitate rather than inhibit access to HCV treatment. Nonetheless, the disparate influence of SBPMs in Louisiana and Washington suggests that states with greater pent-up demand for HCV medications may expect to see larger gains in use from a SBPM.
Subscription-based payment model implementation initiatives may have also driven differential state-level changes in use. Although both Louisiana and Washington included plans to increase screening and treatment for HCV, the effectiveness of these initiatives are unknown. It is possible that Louisiana was better able to scale up screening and treatment initiation for HCV, which may have driven the observed changes in use. Notably, reported chronic HCV cases increased 99% from 2017 to 2018 in Louisiana, then declined 42% from 2018 to 2019. The uptick in reported cases of chronic HCV may reflect an effort to screen more individuals for HCV in Louisiana in preparation for SBPM implementation. Moreover, given the access restrictions in place prior to the SBPM, many individuals in Louisiana may have been aware they were infected with HCV but did not previously qualify for treatment. Washington intended to focus on traditionally hard-to-reach populations and planned to offer HCV testing at needle exchanges and substance use disorder treatment facilities, but these initiatives were halted by the onset of the COVID-19 pandemic (personal communication with officials from the Washington State Health Care Authority [Sullivan D, Fliss M, Evaskus L]; March 10, 2020). Other initiatives, such as promotional testing buses that were going to be driven around the state, were also halted because of the pandemic (personal communication with officials from the Washington State Health Care Authority [Sullivan D, Fliss M, Evaskus L]; March 10, 2020). Stalled implementation efforts may have hindered Washington’s ability to identify those with HCV and engage them in treatment. Moreover, Washington experienced a larger decline in mobility than Louisiana in the first months of the pandemic, which may reflect differences in state policies aimed to reduce spread.35 As such, differences in state-level responses to COVID-19 may have also unintentionally hindered HCV screening, disease identification, and medication access.
we cannot make direct comparisons between them given that each state has a unique synthetic control.......we were unable to speak to granular differences in the implementation or contractual details of SBPMs in either state because this information is not publicly available...... the observational nature of our study limits causal conclusions regarding the effects of SBPMs on use of Medicaid-covered HCV medications. Finally, we cannot speak to the effect of SBPMs on spending related to HCV treatment.
Conclusions
In this cross-sectional study, SBPMs were associated with increased Medicaid-covered HCV prescription fills in Louisiana but not in Washington. These results suggest that SBPMs may enable states to facilitate access to effective but costly medications, which may both improve the health of HCV-infected Medicaid enrollees and potentially reduce downstream spending on complications from untreated HCV.
------------
August 27, 2021
Medicaid Subscription-Based Payment Models and Implications for Access to Hepatitis C Medications
JAMA Health Forum. Samantha G. Auty, MS; Paul R. Shafer, PhD; Kevin N. Griffith, PhD
Key Points
Question Did the use of direct-acting antiviral hepatitis C virus (HCV) medications change after implementation of subscription-based payment models for these drugs in Washington and Louisiana?
Findings In this cross-sectional study, Louisiana experienced a 534.5% increase in HCV prescription fills after implementation of a subscription-based payment model, but no significant change in prescription fills was observed in Washington.
Meaning In this study, subscription-based payment models in Louisiana and Washington were differentially associated with use of Medicaid-covered HCV medications, which may reflect state-level differences in implementation, historical restrictions on access to these medications, and responses to the COVID-19 pandemic.
Abstract
Importance Hepatitis C virus (HCV) can be cured with direct-acting antiviral medications, but state Medicaid programs often restrict access to these lifesaving medications owing to their high costs. Subscription-based payment models (SBPMs), wherein states contract with a single manufacturer to supply prescriptions at a reduced price, may offer a solution that increases access. Whether SBPMs are associated with changes in HCV medication use is unknown.
Objective To estimate changes in Medicaid-covered HCV prescription fills after Louisiana and Washington implemented SBPMs on July 1, 2019.
Design, Setting, and Participants This cross-sectional study examined trends in prescription fills of Medicaid-covered direct-acting antiviral HCV medications in Louisiana and Washington after implementation of SBPMs. A synthetic control approach was used to compare changes in HCV prescription fills between states that did and did not implement SBPMs. The unit of analysis was state-quarter. Outpatient direct-acting antiviral HCV prescription fills from the Medicaid State Drug Utilization Data files were obtained from all 50 US states and the District of Columbia from January 1, 2017, to June 30, 2020.
Exposures Implementation of SBPMs for Medicaid-covered direct-acting antiviral HCV medications.
Main Outcomes and Measures Direct-acting antiviral HCV prescriptions filled per 100 000 Medicaid enrollees.
Results In the year preceding SBPM implementation, the mean (SD) rate of quarterly HCV prescription fills per 100 000 Medicaid enrollees was 43.1 (8.6) prescriptions in Louisiana and 50.1 (4.1) in Washington. After SBPM implementation, the mean (SD) rate of quarterly HCV prescription fills per 100 000 enrollees was 206.0 (51.2) prescriptions in Louisiana and 53.9 (11.0) in Washington. In synthetic control models, SBPM implementation in Louisiana was associated with an increase of 173.5 (95% CI, 74.3-265.3) quarterly prescription fills per 100 000 Medicaid enrollees during the following year, a relative increase of 534.5% (95% CI, 228.7%-1125.0%). Washington did not experience a significant change in prescription fills following SBPM implementation.
Conclusions and Relevance In this cross-sectional study, Louisiana experienced substantial increases in HCV medication use among its Medicaid-enrolled population following SBPM implementation, whereas Washington did not. These differences may partially be explained by state-level variation in SBPM implementation, historical restrictions on access to HCV medications, and responses to the COVID-19 pandemic.
| |
| |
| |
|
|
|